Abstract
Background
The pathophysiology of neurodegenerative diseases (NDDs) is closely associated with cellular oxidative stress which can result in the accumulation of toxic proteins in the endoplasmic reticulum (ER) leading to ER stress and subsequent unfolded protein response (UPR) signaling, a mechanism that aggravate these disorders. Vitamin D has been suggested to have important neuroprotective role and its administration has been shown to reduce neuronal injury, neurotoxicity and oxidative stress in various animal systems.
Objective
The current study was undertaken to examine the effect of vitamin D3 on UPR in ER stress induced Mus musculus neuronal cells.
Methods
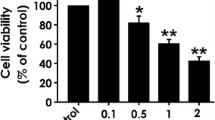
Mus musculus cortical and hippocampal primary neuronal cultures were pretreated with 1,25-dihydroxyvitamin D3 (1, 25-(OH)2D3), the active form of vitamin D, followed by ER stress induction with a chemical ER stress inducer thapsigargin and with an advanced glycated protein, AGE-BSA. The UPR genes and related microRNAs (miRNA) expressions were analyzed mainly using real-time PCR.
Results
The experiment resulted in the suppression of ER stress marker BiP and UPR pathway genes such as Perk, Ire1α, Chop and Puma which mediate cellular apoptosis indicating the protective effect of 1, 25-(OH)2D3 against neuronal ER stress. Further studies into the molecular aspects showed that ER stress mediated down-regulated expression of microRNAs (miRNAs) such as mmu-miR-24, 27b, 124, 224, 290, 351 and 488 which are known to regulate the UPR pathway genes were also reduced with vitamin pretreatment, of which the miRNAs miR-24 and 27b which shares the same cluster are potentially involved in various human diseases.
Conclusion
This study emphasizes the therapeutic role of vitamin D in reducing neuronal ER stress and the need for maintaining sufficient amount of this vitamin in our diet.









Similar content being viewed by others
References
Abate G, Delbarba A, Marziano M, Memo M, Uberti D (2015) Advanced glycation end products (ages) in food: focusing on mediterranean pasta. J Nutr Food Sci 05(06):440
Annweiler C (2016) Vitamin D in dementia prevention. Ann N Y Acad Sci 1367(1):57–63
Bang C, Fiedler J, Thum T (2012) Cardiovascular importance of the MicroRNA-23/27/24 family. Microcirculation 19(3):208–214
Bartel B (2005) MicroRNAs directing siRNA biogenesis. Nat Struct Mol Biol 12(7):569–571
Bartoszewska S, Kochan K, Madanecki P, Piotrowski A, Ochocka R, Collawn J, Bartoszewski R (2013) Regulation of the unfolded protein response by microRNAs. Cell Mol Biol Lett 18(4):555–578
Beaudoin GM, Lee S, Singh D, Yuan Y, Ng Y, Reichardt LF, Arikkath J (2012) Culturing pyramidal neurons from the early postnatal mouse hippocampus and cortex. Nat Protoc 7(9):1741–1754
Besirli CG, Wagner EF, Johnson EM (2005) The limited role of NH2-terminal c-Jun phosphorylation in neuronal apoptosis. J Cell Biol 170:401–411
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL, Vandesompele J, Wittwer CT (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55(4):611–622
Cheng J, Xia X, Rui Y, Zhang Z, Qin L, Han S, Wan Z (2016) The combination of 1α,25dihydroxyvitaminD3 with resveratrol improves neuronal degeneration by regulating endoplasmic reticulum stress, insulin signaling and inhibiting tau hyperphosphorylation in SH-SY5Y cells. Food Chem Toxicol 93:32–40
Chhabra R, Dubey R, Saini N (2010) Cooperative and individualistic functions of the microRNAs in the miR-23a~27a~24-2 cluster and its implication in human diseases. Mol Cancer 9(1):232
Cho S, Wu C, Yasuda T, Cruz LO, Khan AA, Lin L, Nguyen DT, Miller M, Lee HM, Kuo ML, Broide DH, Rajewsky K, Rudensky AY, Lu L (2016) miR-23∼27∼24 clusters control effector T cell differentiation and function. J Cell Biol 212(4):2124OIA22
Ciechanover A, Brundin P (2003) The ubiquitin proteasome system in neurodegenerative diseases. Neuron 40(2):427–446
Galehdar Z, Swan P, Fuerth B, Callaghan SM, Park DS, Cregan SP (2010) Neuronal apoptosis induced by endoplasmic reticulum stress is regulated by ATF4-CHOP-mediated induction of the Bcl-2 homology 3-only member PUMA. J Neurosci 30(50):16938–16948
Gascon E, Gao FB (2012) Cause or effect: misregulation of microRNA pathways in neurodegeneration. Front Neurosci 6:48
Haas MJ, Jafri M, Wehmeier KR, Onstead-Haas LM, Mooradian AD (2016) Inhibition of endoplasmic reticulum stress and oxidative stress by vitamin D in endothelial cells. Free Radic Biol Med 99:1–10
Haddur E, Ozkaya AB, Ak H, Aydin HH (2015) The effect of calcitriol on endoplasmic reticulum stress response. Biochem Cell Biol 93(3):268–271
Hikisz PC, Kiliańska Z (2012) Puma, a critical mediator of cell death—one decade on from its discovery. Cell Mol Biol Lett 17:646–669
Ibi M, Sawada H, Nakanishi M, Kume T, Katsuki H, Kaneko S, Shimohama S, Akaike A (2001) Protective effects of 1α,25-(OH)2D3 against the neurotoxicity of glutamate and reactive oxygen species in mesencephalic culture. Neuropharmacology 40(6):761–771
Jayachandran P, Koshy L, Sudhakaran PR, Nair GM, Gangaprasad A, Nair AJ (2018) Differential expression of microRNAs that target genes involved in apoptotic UPR signaling pathway in neurons. Res J Life Sci Bioinform Pharm Chem Sci 4(3):187–201
Jayachandran P, Koshy L, Sudhakaran P, Nair G, Gangaprasad A, Nair A (2020) Dysregulation of miRNA and its reversal with l-ascorbic acid during AGE-BSA induced ER stress in Musmusculus neuronal cells. Gene Rep 21:100841
Kang K, Zhang X, Liu H, Wang Z, Zhong J, Huang Z, Peng X, Zeng Y, Wang Y, Yang Y, Luo J, Gou D (2012) A novel real-time PCR assay of microRNAs using S-Poly(T), a specific oligo(dT) reverse transcription primer with excellent sensitivity and specificity. PLoS ONE 7(11):e48536
Korhonen L, Lindholm D (2004) The ubiquitin proteasome system in synaptic and axonal degeneration: figure 1. J Cell Biol 165(1):27–30
Kura B, Parikh M, Slezak J, Pierce GN (2019) The influence of diet on MicroRNAs that impact cardiovascular disease. Molecules 24(8):1509
Lauer AA, Janitschke D, Hartmann T, Grimm HS, Grimm MO (2020) The effects of vitamin D deficiency on neurodegenerative diseases. Vitamin D Defic. https://doi.org/10.5772/intechopen.89160
Lisse TS, Adams JS, Hewison M (2013) Vitamin D and MicroRNAs in Bone. Crit Rev Eukaryot Gene Expr 23(3):195–214
Maly DJ, Papa FR (2014) Druggable sensors of the unfolded protein response. Nat Chem Biol 10:892–901
Reimertz C, Kögel D, Rami A, Chittenden T, Prehn JH (2003) Gene expression during ER stress–induced apoptosis in neurons. Eur J Cell Biol 162:587–597
Salahuddin P, Rabbani G, Khan R (2014) The role of advanced glycation end products in various types of neurodegenerative disease: a therapeutic approach. Cell Mol Biol Lett 19(3):407–437
Soto C (2003) Unfolding the role of protein misfolding in neurodegenerative diseases. Nat Rev Neurosci 4(1):49–60
Szegezdi E, Logue SE, Gorman AM, Samali A (2006) Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep 7:880–885
Trochoutsou A, Kloukina V, Samitas K, Xanthou G (2015) Vitamin-D in the immune system: genomic and non-genomic actions. Mini-Rev Med Chem 15(11):953–963
Urano F (2000) Coupling of stress in the ER to activation of JNK protein kinases by transmembrane protein kinase IRE1Α. Science 287:664–666
Wen G, Eder K, Ringseis R (2020) 1,25-hydroxyvitamin D3 decreases endoplasmic reticulum stress-induced inflammatory response in mammary epithelial cells. PLoS ONE 15(2):e0228945
Yoneda T, Imaizumi K, Oono K, Yui D, Gomi F, Katayama T, Tohyama M (2001) Activation of caspase-12, an endoplastic reticulum (ER) resident caspase, through tumor necrosis factor receptor-associated factor 2-dependent mechanism in response to the ER stress. J Biol Chem 276:13935–13940
Yoshida H (2007) ER stress and diseases. FEBS J 274(3):630–658
Zhang K, Kaufman RJ (2008) Chapter twenty identification and characterization of endoplasmic reticulum stress-induced apoptosis in vivo. Program Cell Death Gen Princ Stud Cell Death Part A 442:395–419
Zhou Y, Dong B, Kim K, Choi S, Sun Z, Wu N et al (2020) Vitamin D receptor activation in liver macrophages protects against hepatic endoplasmic reticulum stress in mice. Hepatology 71(4):1453–1466
Acknowledgements
The authors acknowledge the Department of Computational Biology and Bioinformatics, University of Kerala, Trivandrum, India for technical support.
Funding
The work was financially supported by the Department of Higher Education Board, Kerala, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no potential conflict of interest.
Ethical approval
The procedures performed in the studies using animals followed the ethical standards of Institutional Animal Ethics Committee, University of Kerala. [AEC-KU-22/2016-17-BT-JKNC2].
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jayachandran, P., Koshy, L., Sudhakaran, P.R. et al. 1, 25-(OH)2D3 protects against ER stress and miRNA dysregulation in Mus musculus neurons. Genes Genom 44, 1565–1576 (2022). https://doi.org/10.1007/s13258-022-01256-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13258-022-01256-7