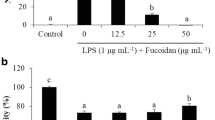
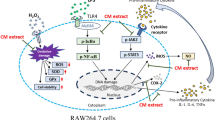
Abstract
Fucoidan is linked to a variety of biological processes. Differences in algae species, extraction, seasons, and locations generate structural variability in fucoidan, affecting its bioactivities. Nothing is known about fucoidan from the brown alga Dictyota bartayresiana, its anti-inflammatory properties, or its inherent mechanism. This study aimed to investigate the anti-inflammatory properties of fucoidan isolated from D. bartayresiana against LPS-induced RAW 264.7 macrophages and to explore potential molecular pathways associated with this anti-inflammatory effects. Fucoidan was first isolated and purified from D. bartayresiana, and then, MTT assay was used to determine the effect of fucoidan on cell viability. Its effects on reactive oxygen species (ROS) formation and apoptosis were also studied using the ROS assay and acridine orange/ethidium bromide fluorescence labelling, respectively. Molecular docking and molecular dynamics simulation studies were performed on target proteins NF-κB and TNF-α to identify the route implicated in these inflammatory events. It was observed that fucoidan reduced LPS-induced inflammation in RAW 264.7 cells. Fucoidan also decreased the LPS-stimulated ROS surge and was found to induce apoptosis in the cells. Molecular docking and molecular dynamics simulation studies revealed that fucoidan’s potent anti-inflammatory action was achieved by obstructing the NF-κB signalling pathway. These findings were particularly noteworthy and novel because fucoidan isolated from D. bartayresiana had not previously been shown to have anti-inflammatory properties in RAW 264.7 cells or to exert its activity by obstructing the NF-κB signalling pathway. Conclusively, these findings proposed fucoidan as a potential pharmaceutical drug for inflammation-related diseases.










Similar content being viewed by others
Data availability
The data analyzed during the present study are available from the corresponding author upon reasonable request.
References
Abdel-Fattah AF, Hussein MMD, Fouad ST (1978) Carbohydrates of the brown seaweed Dictyota dichotoma. Phytochemistry 17:741–743. https://doi.org/10.1016/S0031-9422(00)94218-3
Aisha K, Maryam S (2019) Genus Dictyota (Dictyophyceae, Phaeophycota) from the coastal waters of Pakistan. Int J Biol Res 7(1):1–23
Apostolova E, Lukova P, Baldzhieva A et al (2020) Polymers immunomodulatory and anti-inflammatory effects of fucoidan: a review. Polymers (basel) 12:2338. https://doi.org/10.3390/polym12102338
Bouzidi N, Daghbouche Y, El Hattab M et al (2008) Determination of total sterols in brown algae by Fourier transform infrared spectroscopy. Anal Chim Acta 616:185–189. https://doi.org/10.1016/J.ACA.2008.04.028
Chang KC, Ko YS, Kim HJ et al (2016) 13-Methylberberine reduces HMGB1 release in LPS-activated RAW264.7 cells and increases the survival of septic mice through AMPK/P38 MAPK activation. Int Immunopharmacol 40:269–276. https://doi.org/10.1016/J.INTIMP.2016.08.022
Chen P-H, Chiang P-C, Lo W-C et al (2021) NC-ND license A novel fucoidan complex-based functional beverage attenuates oral cancer through inducing apoptosis, G2/M cell cycle arrest and retarding cell migration/invasion. J Funct Foods 85:1756–4646. https://doi.org/10.1016/j.jff.2021.104665
Chuang CC, McIntosh MK (2011) Potential mechanisms by which polyphenol-rich grapes prevent obesity-mediated inflammation and metabolic diseases. Annu Rev Nutr 31:155–176. https://doi.org/10.1146/annurev-nutr-072610-145149
Citkowska A, Szekalska M, Winnicka K (2019) Possibilities of fucoidan utilization in the development of pharmaceutical dosage forms. Mar Drugs 17:458. https://doi.org/10.3390/MD17080458
Costa LS, Fidelis GP, Cordeiro SL et al (2010) Biological activities of sulfated polysaccharides from tropical seaweeds. Biomed Pharmacother 64:21–28. https://doi.org/10.1016/J.BIOPHA.2009.03.005
Delma CR, Thirugnanasambandan S, Srinivasan GP et al (2019) Fucoidan from marine brown algae attenuates pancreatic cancer progression by regulating p53–NFκB crosstalk. Phytochemistry 167:112078. https://doi.org/10.1016/J.PHYTOCHEM.2019.112078
dos Santos Nunes RG, Pereira PS, Elekofehinti OO et al (2019) Possible involvement of transcriptional activation of nuclear factor erythroid 2-related factor 2 (Nrf2)in the protective effect of caffeic acid on paraquat-induced oxidative damage in Drosophila melanogaster. Pestic Biochem Physiol 157:161–168. https://doi.org/10.1016/j.pestbp.2019.03.017
Dubey A, Sivaraman J (2022) Evaluation of the cytotoxicity, apoptosis and autophagy induction by fucoidan in human hepatic adenocarcinoma cells. Indian J Pharm Educ Res 56:756–764. https://doi.org/10.5530/ijper.56.3.125
Fitton JH, Stringer DN, Park Y, Karpiniec SS (2019) Therapies from fucoidan: new developments. Mar Drugs 17(10):571. https://doi.org/10.3390/md17100571
Hariram Nile S, Won Park S (2013) Optimized methods for in vitro and in vivo anti-inflammatory assays and its applications in herbal and synthetic drug analysis. Mini-Rev Med Chem 13:95–100. https://doi.org/10.2174/1389557511307010095
Iwalewa EO, Mcgaw LJ, Naidoo V, Eloff JN (2010) Inflammation: the foundation of diseases and disorders. A review of phytomedicines of South African origin used to treat pain and inflammatory conditions. Afr J Biotechnol 6:2868–2885. https://doi.org/10.4314/ajb.v6i25.58240
Jayawardena TU, Fernando IPS, Lee WW et al (2019) Isolation and purification of fucoidan fraction in Turbinaria ornata from the Maldives; inflammation inhibitory potential under LPS stimulated conditions in in-vitro and in-vivo models. Int J Biol Macromol 131:614–623. https://doi.org/10.1016/J.IJBIOMAC.2019.03.105
Kauppinen A, Suuronen T, Ojala J et al (2013) Antagonistic crosstalk between NF-κB and SIRT1 in the regulation of inflammation and metabolic disorders. Cell Signal 25:1939–1948. https://doi.org/10.1016/J.CELLSIG.2013.06.007
Kiemer AK, Müller C, Vollmar AM (2002) Inhibition of LPS-induced nitric oxide and TNF-α production by α-lipoic acid in rat Kupffer cells and in RAW 264.7 murine macrophages. Immunol Cell Biol 80:550–557. https://doi.org/10.1046/J.1440-1711.2002.01124.X
Kim JW, Zhou Z, Yun H et al (2020) Cigarette smoking differentially regulates inflammatory responses in a mouse model of nonalcoholic steatohepatitis depending on exposure time point. Food Chem Toxicol 135:110930. https://doi.org/10.1016/J.FCT.2019.110930
Kopplin G, Rokstad AM, Mélida H et al (2018) Structural characterization of fucoidan from Laminaria hyperborea: assessment of coagulation and inflammatory properties and their structure-function relationship. ACS Appl Bio Mater 1:1880–1892. https://doi.org/10.1021/ACSABM.8B00436
Lee H-G, Nagahawatta DP, Liyanage NM et al (2022) Structural characterization and anti-inflammatory activity of fucoidan isolated from Ecklonia maxima stipe. Algae 37(3):239–247. https://doi.org/10.4490/algae.2022.37.9.12
Li C, Gao Y, Xing Y et al (2011) Fucoidan, a sulfated polysaccharide from brown algae, against myocardial ischemia-reperfusion injury in rats via regulating the inflammation response. Food Chem Toxicol 49:2090–2095. https://doi.org/10.1016/J.FCT.2011.05.022
Li S, Li J, Mao G et al (2018) A fucoidan from sea cucumber Pearsonothuria graeffei with well-repeated structure alleviates gut microbiota dysbiosis and metabolic syndromes in HFD-fed mice. Food Funct 9:5371–5380. https://doi.org/10.1039/C8FO01174E
Li R, Zhou Q-L, Yang R-Y et al (2023) Determining the potent immunostimulation potential arising from the heteropolysaccharide structure of a novel fucoidan, derived from Sargassum Zhangii. Food Chem 18:1–10. https://doi.org/10.1016/j.fochx.2023.100712
Liu D, Cao G, Han L et al (2016) Flavonoids from Radix Tetrastigmae inhibit TLR4/MD-2 mediated JNK and NF-κB pathway with anti-inflammatory properties. Cytokine 84:29–36. https://doi.org/10.1016/J.CYTO.2015.08.003
Liu Q, Xiao XH, Hu LB et al (2017) Anhuienoside C ameliorates collagen-induced arthritis through inhibition of MAPK and NF-κB signaling pathways. Front Pharmacol 8:1–12. https://doi.org/10.3389/FPHAR.2017.00299
Manggau M, Kasim S, Fitri N et al (2022) Antioxidant, anti-inflammatory and anticoagulant activities of sulfate polysaccharide isolate from brown alga Sargassum policystum. IOP Conf Ser Earth Environ Sci 967:1–12. https://doi.org/10.1088/1755-1315/967/1/012029
Martins AP, Yokoya NS, Colepicolo P (2016) Biochemical modulation by carbon and nitrogen addition in cultures of Dictyota menstrualis (Dictyotales, Phaeophyceae) to generate oil-based bioproducts. Mar Biotechnol 18:314–326. https://doi.org/10.1007/S10126-016-9693-9
Mauriz JL, Collado PS, Veneroso C et al (2013) A review of the molecular aspects of melatonin’s anti-inflammatory actions: recent insights and new perspectives. J Pineal Res 54:1–14. https://doi.org/10.1111/j.1600-079x.2012.01014.x
Na YS, Kim WJ, Kim SM et al (2010) Purification, characterization and immunostimulating activity of water-soluble polysaccharide isolated from Capsosiphon fulvescens. Int Immunopharmacol 10:364–370. https://doi.org/10.1016/J.INTIMP.2009.12.011
Ni L, Wang L, Fu X et al (2020) In vitro and in vivo anti-inflammatory activities of a fucose-rich fucoidan isolated from Saccharina japonica. Int J Biol Macromol 156:717–729. https://doi.org/10.1016/J.IJBIOMAC.2020.04.012
Obluchinskaya ED, Pozharitskaya ON, Shikov AN (2022) In vitro anti-inflammatory activities of fucoidans from five species of brown seaweeds. Mar Drugs 20:1–16. https://doi.org/10.3390/md20100606
Phull AR, Kim SJ (2017) Fucoidan as bio-functional molecule: Insights into the anti-inflammatory potential and associated molecular mechanisms. J Funct Foods 38:415–426. https://doi.org/10.1016/J.JFF.2017.09.051
Priyan I, Fernando S, Kapuge K et al (2020) Fucoidan purified from Sargassum polycystum induces apoptosis through mitochondria-mediated pathway in HL-60 and MCF-7 cells. Mar Drugs 18(196):1–13. https://doi.org/10.3390/md18040196
Rabanal M, Ponce NMA, Navarro DA et al (2014) The system of fucoidans from the brown seaweed Dictyota dichotoma: chemical analysis and antiviral activity. Carbohydr Polym 101:804–811. https://doi.org/10.1016/J.CARBPOL.2013.10.019
Ragan MA, Jensen A (1977) Quantitative studies on brown algal phenols. I. Estimation of absolute polyphenol content of Ascophyllum nodosum (L.) Le Jol. and Fucus vesiculosus (L.). J Exp Mar Bio Ecol 30:209–221. https://doi.org/10.1016/0022-0981(77)90013-2
Subbaiya R, Saravanan M, Priya AR et al (2017) Biomimetic synthesis of silver nanoparticles from Streptomyces atrovirens and their potential anticancer activity against human breast cancer cells; Biomimetic synthesis of silver nanoparticles from Streptomyces atrovirens and their potential anticancer activity against human breast cancer cells. IET Nanobiotechnol 11(8):965–972. https://doi.org/10.1049/iet-nbt.2016.0222
Sun Y, Liu J, Jiang X et al (2015) One-step synthesis of chiral oxindole-type analogues with potent anti-inflammatory and analgesic activities. Sci Rep 5:1–7. https://doi.org/10.1038/srep13699
Taylor RB, Lindquist N, Kubanek J, Hay ME (2003) Intraspecific variation in palatability and defensive chemistry of brown seaweeds: effects on herbivore fitness. Oecologia 136:412–423. https://doi.org/10.1007/S00442-003-1280-X
Toker G, Küpeli E, Memisoǧlu M, Yesilada E (2004) Flavonoids with antinociceptive and anti-inflammatory activities from the leaves of Tilia argentea (silver linden). J Ethnopharmacol 95:393–397. https://doi.org/10.1016/J.JEP.2004.08.008
Wang Y, Chen P, Tang C et al (2014) Antinociceptive and anti-inflammatory activities of extract and two isolated flavonoids of Carthamus tinctorius L. J Ethnopharmacol 151:944–950. https://doi.org/10.1016/J.JEP.2013.12.003
Wang Y, Xu Y, Zhang P et al (2018) Smiglaside A ameliorates LPS-induced acute lung injury by modulating macrophage polarization via AMPK-PPARγ pathway. Biochem Pharmacol 156:385–395. https://doi.org/10.1016/J.BCP.2018.09.002
Wang L, Oh JY, Hwang J et al (2019) In vitro and in vivo antioxidant activities of polysaccharides isolated from celluclast-assisted extract of an edible brown seaweed, Sargassum fulvellum. Antioxidants 8(493):1–12. https://doi.org/10.3390/antiox8100493
Wang Z, Guan Y, Yang R et al (2020) Anti-inflammatory activity of 3-cinnamoyltribuloside and its metabolomic analysis in LPS-activated RAW 264.7 cells. BMC Complement Med Ther 20(329):1–12. https://doi.org/10.1186/S12906-020-03115-Y
Wu L, Sun J, Su X et al (2016) A review about the development of fucoidan in antitumor activity: progress and challenges. Carbohydr Polym 154:96–111. https://doi.org/10.1016/J.CARBPOL.2016.08.005
Wu J, Liu Z, Su J et al (2017) Anti-inflammatory activity of 3β-hydroxycholest-5-en-7-one isolated from Hippocampus trimaculatus leach via inhibiting iNOS, TNF-α, and 1L–1β of LPS induced RAW 264.7 macrophage cells. Food Funct 8:788–795. https://doi.org/10.1039/C6FO01154C
Xu Y, Chen F (2020) Antioxidant, anti-inflammatory and anti-apoptotic activities of nesfatin-1: a review. J Inflamm Res 13:607–617. https://doi.org/10.2147/JIR.S273446
Ye J, Chen D, Ye Z et al (2020) Fucoidan isolated from Saccharina japonica inhibits LPS-induced inflammation in macrophages via blocking NF-κB, MAPK and JAK-STAT pathways. Mar Drugs 18:1–17. https://doi.org/10.3390/md18060328
Zhang H, Tsao R (2016) Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr Opin Food Sci 8:33–42. https://doi.org/10.1016/J.COFS.2016.02.002
Zhang A, Sun H, Wang X (2013) Recent advances in natural products from plants for treatment of liver diseases. Eur J Med Chem 63:570–577. https://doi.org/10.1016/J.EJMECH.2012.12.062
Zhang TT, Wang M, Yang L et al (2015) Flavonoid glycosides from Rubus chingii Hu fruits display anti-inflammatory activity through suppressing MAPKs activation in macrophages. J Funct Foods 18:235–243. https://doi.org/10.1016/J.JFF.2015.07.006
Zhou ZL, Yang YX, Ding J et al (2012) Triptolide: structural modifications, structure–activity relationships, bioactivities, clinical development and mechanisms. Nat Prod Rep 29:457–475. https://doi.org/10.1039/C2NP00088A
Acknowledgements
The authors gratefully acknowledge Vellore Institute of Technology, Vellore, for the support for Research.
Author information
Authors and Affiliations
Contributions
AD and JS conceptualized the research, AD did isolation of compound, investigation, data curation, and original draft preparations, TD performed the laboratory experiments and TR conceptualized the laboratory experiments, VD performed the computational experiments, and JS finally reviewed and supervised the entire study.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest in the publication.
Ethics declarations
The authors have no competing interests to declare that are relevant to the content of this article. There are no human or animal subjects involved in this study, and informed consent is not applicable.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dubey, A., Dasgupta, T., Devaraji, V. et al. Investigating anti-inflammatory and apoptotic actions of fucoidan concentrating on computational and therapeutic applications. 3 Biotech 13, 355 (2023). https://doi.org/10.1007/s13205-023-03771-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-023-03771-0