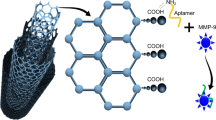
Abstract
The level of clotting protein ‘factor IX’ (FIX) is highly associated with cerebral ischemia, and this research work has developed a sensitive detection of FIX on dielectrode sensor by current-volt measurement. Sensing area was grown with zinc oxide nanowire to attach more probe for FIX interaction. Aptamer was utilized as the detection probe and attached on the sensing electrode surface through amine-aldehyde chemical linkage. In addition, biotin-streptavidin interaction was utilized to attach the higher number aptamers on the electrode surface connected with dual-probe station. FIX detection limit was found as 10 fM in the phosphate buffer saline spiked samples and 1:320 dilution of human serum. The linear ranges were as 10 fM to 100 pM and 1:320 to 1:80, respectively. With a good determination co-efficient [y = 2.6813x − 3.8467; R2 = 0.9479] this biosensing strategy helps to quantify FIX and monitor the condition of cerebral ischemia.





Similar content being viewed by others
References
Albertyn LE (1987) Acute portal vein thrombosis. Clin Radiol 38:645–648. https://doi.org/10.1016/S0009-9260(87)80353-7
Andrews RK, Berndt MC (2004) Platelet physiology and thrombosis. In: Thrombosis Research. In: XIth International Congress on Antiphospholipid Antibodies, 14–18th November 2004, Sydney Australia. Elsevier Ltd. Published by Elsevier Inc 114: 447–453
Appiah D, Fashanu OE, Heckbert SR et al (2017) Relation of coagulation factor XI with incident coronary heart disease and stroke: the cardiovascular health study. Blood Coagul Fibrinolysis 28:389–392. https://doi.org/10.1097/MBC.0000000000000616
Bagoly Z, Szegedi I, Kálmándi R et al (2019) Markers of coagulation and fibrinolysis predicting the outcome of acute ischemic stroke thrombolysis treatment: a review of the literature. Front Neurol 10:1–13. https://doi.org/10.3389/fneur.2019.00513
Beck S, Meer J, Taylor T (2016) Deep vein thrombosis. Emerg Med 48:29–31
Burden G, Factors R (2006) Disease control priorities project. In: Lopez AD, Mathers CD, Ezzati M, Jamison DT, Murray CJL (eds) Global burden of disease and risk factors. Oxford University Press, New York (ISBN-10: 0-8213-6262-3)
Cheen OC, Gopinath SCB, Perumal V et al (2017) Aptamer-based impedimetric determination of the human blood clotting factor IX in serum using an interdigitated electrode modified with a ZnO nanolayer. Microchim Acta 184:117–125. https://doi.org/10.1007/s00604-016-2001-6
Gopinath SCB, Shikamoto Y, Mizuno H, Kumar PKR (2006) A potent anti-coagulant RNA aptamer inhibits blood coagulation by specifically blocking the extrinsic clotting pathway. Thromb Haemost 95:767–771. https://doi.org/10.1160/Th06-01-0047
Gopinath SC, Shikamoto Y, Mizuno H, Kumar PK (2007) Snake-venom-derived Factor IX-binding protein specifically blocks the gamma-carboxyglutamic acid-rich-domain-mediated membrane binding of human Factors IX and X. Biochem J 405:351–357. https://doi.org/10.1042/BJ20061737
Gopinath SCB, Awazu K, Tominaga J, Kumar PKR (2008) Monitoring biomolecular interactions on a digital versatile disk: a BioDVD platform technology. ACS Nano 2:1885–1895. https://doi.org/10.1021/nn800285p
Gopinath SCB, Awazu K, Fujimaki M (2012) Waveguide-mode sensors as aptasensors. Sensors 12:2136–2151. https://doi.org/10.3390/s120202136
Gross PL, Weil P, Rand ML (2018) Hemostasis & thrombosis. In: Rodwell VW, Bender DA, Botham KM, Kennelly PJ, Weil P (eds) Harper’s illustrated biochemistry. McGraw Hill, New York, p 31
Guo S, Lakshmipriya T, Gopinath SCB et al (2019) Complementation of ELISA and an interdigitated electrode surface in gold nanoparticle functionalization for effective detection of human blood clotting defects. Nanoscale Res Lett 14:222. https://doi.org/10.1186/s11671-019-3058-z
Guo X, Wen F, Zheng N et al (2020) Aptamer-based biosensor for detection of mycotoxins. Front Chem 8:195. https://doi.org/10.3389/fchem.2020.00195
Heikal NM, Murphy KK, Crist RA et al (2013) Elevated factor IX activity is associated with an increased odds ratio for both arterial and venous thrombotic events. Am J Clin Pathol 140:680–685. https://doi.org/10.1309/AJCPAGOR4Q2IIKUG
Howard EL, Becker KCD, Rusconi CP, Becker RC (2007) Factor IXa inhibitors as novel anticoagulants. Arterioscler Thromb Vasc Biol 27:722–727
Jickling GC, Sharp FR (2011) Blood biomarkers of ischemic stroke. Neurotherapeutics 8:349–360
Jickling GC, Sharp FR (2015) Biomarker panels in ischemic stroke. Stroke 46:915–920. https://doi.org/10.1161/STROKEAHA.114.005604
Lakshmipriya T, Fujimaki M, Gopinath SCB et al (2013) A high-performance waveguide-mode biosensor for detection of factor IX using PEG-based blocking agents to suppress non-specific binding and improve sensitivity. Analyst 138:2863. https://doi.org/10.1039/c3an00298e
Lakshmipriya T, Horiguchi Y, Nagasaki Y (2014) Co-immobilized poly(ethylene glycol)-block-polyamines promote sensitivity and restrict biofouling on gold sensor surface for detecting factor IX in human plasma. Analyst 139:3977–3985. https://doi.org/10.1039/c4an00168k
Lakshmipriya T, Gopinath SCB, Tang T-H (2016) Biotin-streptavidin competition mediates sensitive detection of biomolecules in enzyme linked immunosorbent assay. PLOS ONE 11:e0151153. https://doi.org/10.1371/journal.pone.0151153
Letchumanan I, Gopinath SCB, Md Arshad MK et al (2019) Gold nano-urchin integrated label-free amperometric aptasensing human blood clotting factor IX: A prognosticative approach for “Royal disease.” Biosens Bioelectron 131:128–135. https://doi.org/10.1016/j.bios.2019.02.006
Nagasaki Y (2011) Construction of a densely poly(ethylene glycol)-chain-tethered surface and its performance. Polym J 43:949–958. https://doi.org/10.1038/pj.2011.93
Nagasaki Y, Kobayashi H, Katsuyama Y et al (2007) Enhanced immunoresponse of antibody/mixed-PEG co-immobilized surface construction of high-performance immunomagnetic ELISA system. J Colloid Interface Sci 309:524–530. https://doi.org/10.1016/j.jcis.2006.12.079
Rusconi CP, Scardino E, Layzer J et al (2002) RNA aptamers as reversible antagonists of coagulation factor IXa. Nature 419:90–94. https://doi.org/10.1038/nature00963
Sharpe EE, Pasternak JJ (2017) Cerebral venous thrombosis. Essentials of neuroanesthesia. Academic Press, Cambridge, pp 681–691
Sullenger B, Woodruff R, Monroe DM (2012) Potent anticoagulant aptamer directed against factor IXa blocks macromolecular substrate interaction. J Biol Chem 287:12779–12786. https://doi.org/10.1074/jbc.M111.300772
Yang DT, Flanders MM, Kim H, Rodgers GM (2006) Elevated factor XI activity levels are associated with an increased odds ratio for cerebrovascular events. Am J Clin Pathol 126:411–415. https://doi.org/10.1309/QC259F09UNMKVP0R
Yao J, Li S, Zhang L et al (2020) Aptamer-antibody dual probes on single-walled carbon nanotube bridged dielectrode: comparative analysis on human blood clotting factor. Int J Biol Macromol 151:1133–1138. https://doi.org/10.1016/j.ijbiomac.2019.10.156
Author information
Authors and Affiliations
Contributions
WG, MS and XL conceived and designed the study. XG, YL, HL and LX performed the literature search and data extraction. WX drafted the manuscript. The authors are contributed to the preparation of the manuscript and discussion. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interest.
Ethical statement
None
Rights and permissions
About this article
Cite this article
Gong, W., Sun, M., Guo, X. et al. Nanowired dual-electrodes surface to monitor cerebral ischemia by current-volt measurements. 3 Biotech 11, 502 (2021). https://doi.org/10.1007/s13205-021-03048-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-03048-4