Abstract
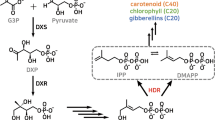
2-C-Methyl-d-erythrol-4-phosphate (MEP) pathway in plant supplies isoprene building blocks for carotenoids and chlorophylls essential in photosynthesis as well as plant hormones such as gibberellin and abscisic acid. To assess the effect of overexpression of the terminal enzyme of the MEP pathway, 1-hydroxy-2-methyl-2-(E)-butenyl-4-diphosphate reductase (HDR), transgenic Nicotiana tabacum overexpressing class 2 HDR from Ginkgo biloba (GbHDR2) under the control of 35S promoter was constructed. Contents of chlorophylls a and b in transgenic tobacco were enhanced by 19 and 7%, respectively, compared to those of the wild type. The carotenoid level was also 18% higher than that in the control plant. As a result, photosynthetic rate of the transgenic tobacco was increased by up to 51%. Diterepenoid duvatrienediol content of transgenic tobacco was also elevated by at least sixfold. To explore the molecular basis of the enhanced isoprenoid accumulation, transcript levels of the key genes involved in the isoprenoid biosynthesis were measured. Transcript levels of geranylgeranyl diphosphate synthase (GGPP), kaurene synthase (KS), gibberellic acid 20 oxidase (GA20ox), and phytoene desaturase (PD) genes in the transgenic tobacco leaves were about twofold higher compared to the wild type. Therefore, upregulation of down-stream genes involved in biosynthesis of di- and tetraterpenoids due to GbHDR2 overexpression was responsible for elevated production of isoprenoids and enhanced photosynthetic rate.






Similar content being viewed by others
Abbreviations
- ABA:
-
Abscisic acid
- CMK,4:
-
(Cytidine 5′-diphospho)-2-C-methyl-d-erythritol kinase
- DMAPP:
-
Dimethylallyl diphosphate
- DVT:
-
Duvatrienediol
- DXS:
-
1-Deoxyxylulose 5-phosphate synthase
- G3P:
-
Glyceraldehydes 3-phosphate
- GA:
-
Gibberellic acid
- GA20ox:
-
Gibberellic acid 20 oxidase
- GbHDR2:
-
Ginkgo biloba Class 2 1-hydroxy-2 methyl-2-(E)-butenyl 4-diphosphate reductase
- GGPPS:
-
Geranylgeranyl diphosphate synthase
- HDR:
-
1-Hydroxy-2-methyl-2-(E)-butenyl-4-diphosphate reductase
- IPP:
-
Isopentenyl diphosphate
- KS:
-
Kaurene synthase
- MEP:
-
2-C-Methyl-d-erythritol-4-phosphate
- ORF:
-
Open reading frame
- PDS:
-
Phytoene desaturase
- qRT-PCR:
-
Quantitative real-time PCR
References
Banerjee A, Wu Y, Banerjee R, Li Y, Yan H, Sharkey TD (2013) Feedback inhibition of deoxy-d-xylulose-5-phosphate synthase regulates the methylerythritol 4-phosphate pathway. J Biol Chem 288(23):16926–16936. https://doi.org/10.1074/jbc.M113.464636
Besumbes Ó, Sauret-Güeto S, Phillips MA, Imperial S, Rodríguez-Concepción M, Boronat A (2004) Metabolic engineering of isoprenoid biosynthesis in Arabidopsis for the production of taxadiene, the first committed precursor of Taxol. Biotechnol Bioeng 88(2):168–175. https://doi.org/10.1002/bit.20237
Botella-Pavía P, Besumbes Ó, Phillips MA, Carretero-Paulet L, Boronat A, Rodríguez-Concepción M (2004) Regulation of carotenoid biosynthesis in plants: evidence for a key role of hydroxymethylbutenyl diphosphate reductase in controlling the supply of plastidial isoprenoid precursors. Plant J 40(2):188–199. https://doi.org/10.1111/j.1365-313X.2004.02198.x
Bouwmeester HJ, Roux C, Lopez-Raez JA, Becard G (2007) Rhizosphere communication of plants, parasitic plants and AM fungi. Trends Plant Sci 12(5):224–230. https://doi.org/10.1016/j.tplants.2007.03.009
Brown AC, Eberl M, Crick DC, Jomaa H, Parish T (2010) The nonmevalonate pathway of isoprenoid biosynthesis in Mycobacterium tuberculosis is essential and transcriptionally regulated by Dxs. J Bacteriol 192(9):2424–2433. https://doi.org/10.1128/JB.01402-09
Chiang SY, Grunwald C (1976) Duvatrienediol, alkanes, and fatty acids in cuticular wax of tobacco leaves of various physiological maturity. Phytochemistry 15(6):961–963. https://doi.org/10.1016/S0031-9422(00)84380-0
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162(1):156–159. https://doi.org/10.1006/abio.1987.9999
Eisenreich W, Bacher A, Arigoni D, Rohdich F (2004) Biosynthesis of isoprenoids via the non-mevalonate pathway. Cell Mol Life Sci 61(12):1401–1426. https://doi.org/10.1007/s00018-004-3381-z
Estévez JM, Cantero A, Reindl A, Reichler S, León P (2001) 1-Deoxy-d-xylulose-5-phosphate synthase, a limiting enzyme for plastidic isoprenoid biosynthesis in plants. J Biol Chem 276(25):22901–22909. https://doi.org/10.1074/jbc.M100854200
Farmer WR, Liao JC (2001) Precursor balancing for metabolic engineering of lycopene production in Escherichia coli. Biotechnol Prog 17(1):57–61. https://doi.org/10.1021/bp000137t
Gallois P, Marinho P (1995) Leaf disk transformation using Agrobacterium tumefaciens-expression of heterologous genes in tobacco. In: Jones H (ed) Plant gene transfer and expression protocols: methods in molecular biology, vol 39. Springer, Totowa. https://doi.org/10.1385/0-89603-321-X:39
Goldstein JL, Brown MS (1990) Regulation of the mevalonate pathway. Nature 343(6257):425–430. https://doi.org/10.1038/343425a0
Hori T, Ridge RW, Tulecke W, Del Tredici P, Trémouillaux-Guiller J, Tobe H (2012) Ginkgo biloba a global treasure: from biology to medicine. Springer, p 427
Kang M (2013) Studies on Ginkgo biloba (E)-4-hydroxy-3-methylbut-(2)-enyl diphosphate reductase gene and promoter. Dissertation, Seoul National University
Kang M-K, Nargis S, Kim S-M, Kim S-U (2013) Distinct expression patterns of two Ginkgo biloba 1-hydroxy-2-methyl-2-(E)-butenyl-4-diphosphate reductase/isopentenyl diphospahte synthase (HDR/IDS) promoters in Arabidopsis model. Plant Physiol Biochem 62:47–53. https://doi.org/10.1016/j.plaphy.2012.10.011
Keeling CI, Weisshaar S, Lin RP, Bohlmann J (2008) Functional plasticity of paralogous diterpene synthases involved in conifer defense. PNAS 105(3):1085–1090. https://doi.org/10.1073/pnas.0709466105
Kim SW, Keasling J (2001) Metabolic engineering of the nonmevalonate isopentenyl diphosphate synthesis pathway in Escherichia coli enhances lycopene production. Biotechnol Bioeng 72(4):408–415. https://doi.org/10.1002/1097-0290(20000220)72:4%3c408::aid-bit1003%3e3.0.co;2-h
Kim S-M, Kuzuyama T, Chang Y-J, Kim S-U (2005) Functional identification of Ginkgo biloba 1-deoxy-d-xylulose 5-phosphate synthase (DXS) gene by using Escherichia coli disruptants defective in DXS gene. J Appl Biol Chem 48(2):101–104
Kim S-M, Kuzuyama T, Chang Y-J, Kim S-U (2006a) Cloning and characterization of 2-C-methyl-d-erythritol 2, 4-cyclodiphosphate synthase (MECS) gene from Ginkgo biloba. Plant Cell Rep 25(8):829–835. https://doi.org/10.1007/s00299-006-0136-3
Kim S-M, Kuzuyama T, Chang Y-J, Kwon H-J, Kim S-U (2006b) Cloning and functional characterization of 2-C-methyl-d-erythritol 4-phosphate cytidyltransferase (GbMECT) gene from Ginkgo biloba. Phytochemistry 67(14):1435–1441. https://doi.org/10.1016/j.phytochem.2006.05.034
Kim S-M, Kuzuyama T, Chang Y-J, Song K-S, Kim S-U (2006c) Identification of class 2 1-deoxy-d-xylulose 5-phosphate synthase and 1-deoxy-d-xylulose 5-phosphate reductoisomerase genes from Ginkgo biloba and their transcription in embryo culture with respect to ginkgolide biosynthesis. Planta Med 72(3):234–240. https://doi.org/10.1055/s-2005-916180
Kim S-M, Kim Y-B, Kuzuyama T, Kim S-U (2008a) Two copies of 4-(cytidine 5′-diphospho)-2-C-methyl-d-erythritol kinase (CMK) gene in Ginkgo biloba: molecular cloning and functional characterization. Planta 228(6):941–950. https://doi.org/10.1007/s00425-008-0794-1
Kim S-M, Kuzuyama T, Kobayashi A, Sando T, Chang Y-J, Kim S-U (2008b) 1-Hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate reductase (IDS) is encoded by multicopy genes in gymnosperms Ginkgo biloba and Pinus taeda. Planta 227(2):287–298. https://doi.org/10.1007/s00425-007-0616-x
Kim Y-B, Kim S-M, Kang M-K, Kuzuyama T, Lee JK, Park S-C, Shin S-c, Kim S-U (2009) Regulation of resin acid synthesis in Pinus densiflora by differential transcription of genes encoding multiple 1-deoxy-d-xylulose 5-phosphate synthase and 1-hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate reductase genes. Tree Physiol 29(5):737–749. https://doi.org/10.1093/treephys/tpp002
Lichtenthaler HK, Buschmann C (2001) Chlorophylls and carotenoids: measurement and characterization by UV–VIS spectroscopy. CPFAC 1(1):F4.3.1-F4.3.8. https://doi.org/10.1002/0471142913.faf0403s01
Maass D, Arango J, Wüst F, Beyer P, Welsch R (2009) Carotenoid crystal formation in Arabidopsis and carrot roots caused by increased phytoene synthase protein levels. PLoS ONE 4(7):e6373. https://doi.org/10.1371/journal.pone.0006373
McGarvey DJ, Croteau R (1995) Terpenoid metabolism. Plant Cell 7(7):1015–1026. https://doi.org/10.1105/tpc.7.7.1015
Morris WL, Ducreux LJ, Hedden P, Millam S, Taylor MA (2006) Overexpression of a bacterial 1-deoxy-d-xylulose 5-phosphate synthase gene in potato tubers perturbs the isoprenoid metabolic network: implications for the control of the tuber life cycle. J Exp Bot 57(12):3007–3018. https://doi.org/10.1093/jxb/erl061
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Murray M, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8(19):4321–4326. https://doi.org/10.1093/nar/8.19.4321
Olofsson L, Engstrom A, Lundgren A, Brodelius PE (2011) Relative expression of genes of terpene metabolism in different tissues of Artemisia annua L. BMC Plant Biol 11:45. https://doi.org/10.1186/1471-2229-11-45
Rieu I, Eriksson S, Powers SJ, Gong F, Griffiths J, Woolley L, Benlloch R, Nilsson O, Thomas SG, Hedden P (2008) Genetic analysis reveals that C19-GA 2-oxidation is a major gibberellin inactivation pathway in Arabidopsis. Plant Cell 20(9):2420–2436. https://doi.org/10.1105/tpc.108.058818
Rodríguez-Concepción M, Campos N, Ferrer A, Boronat A (2012) Biosynthesis of isoprenoid precursors in Arabidopsis. In: Bach T, Rohmer M (eds) Isoprenoid synthesis in plants and microorganisms. Springer, New York, pp 439–456. https://doi.org/10.1007/978-1-4614-4063-5_30
Rohmer M (1999) The discovery of a mevalonate-independent pathway for isoprenoid biosynthesis in bacteria, algae and higher plants. Nat Prod Rep 16(5):565–574. https://doi.org/10.1039/a709175c
Ronen G, Cohen M, Zamir D, Hirschberg J (1999) Regulation of carotenoid biosynthesis during tomato fruit development: expression of the gene for lycopene epsilon-cyclase is down-regulated during ripening and is elevated in the mutant Delta. Plant J 17(4):341–351. https://doi.org/10.1046/j.1365-313x.1999.00381.x
Sapir-Mir M, Mett A, Belausov E, Tal-Meshulam S, Frydman A, Gidoni D, Eyal Y (2008) Peroxisomal localization of Arabidopsis isopentenyl diphosphate isomerases suggests that part of the plant isoprenoid mevalonic acid pathway is compartmentalized to peroxisomes. Plant Physiol 148(3):1219–1228. https://doi.org/10.1104/pp.108.127951
Sathasivam R, Radhakrishnan R, Kim JK, Park SU (2020) An update on biosynthesis and regulation of carotenoids in plants. S Afr J Bot. https://doi.org/10.1016/j.sajb.2020.05.015
Seo M, Koshiba T (2002) Complex regulation of ABA biosynthesis in plants. Trends Plant Sci 7(1):41–48. https://doi.org/10.1016/s1360-1385(01)02187-2
Simkin AJ, Guirimand G, Papon N, Courdavault V, Thabet I, Ginis O, Bouzid S, Giglioli-Guivarc’h N, Clastre M (2011) Peroxisomal localisation of the final steps of the mevalonic acid pathway in planta. Planta 234(5):903–914. https://doi.org/10.1007/s00425-011-1444-6
Tissier A, Sallaud C, Rontein D (2012) Tobacco trichomes as a platform for terpenoid biosynthesis engineering. In: Bach T, Rohmer M (eds) Isoprenoid synthesis in plants and microorganisms. Springer, New York, pp 271–283. https://doi.org/10.1007/978-1-4614-4063-5_18
Von Lintig J, Welsch R, Bonk M, Giuliano G, Batschauer A, Kleinig H (1997) Light-dependent regulation of carotenoid biosynthesis occurs at the level of phytoene synthase expression and is mediated by phytochrome in Sinapis alba and Arabidopsis thaliana seedlings. Plant J 12(3):625–634. https://doi.org/10.1046/j.1365-313x.1997.00625.x
Walter MH, Fester T, Strack D (2000) Arbuscular mycorrhizal fungi induce the non-mevalonate methylerythritol phosphate pathway of isoprenoid biosynthesis correlated with accumulation of the ‘yellow pigment’and other apocarotenoids. Plant J 21(6):571–578. https://doi.org/10.1046/j.1365-313x.2000.00708.x
Wang Y, Fu B, Pan L, Chen L, Fu X, Li K (2013) Overexpression of Arabidopsis Dof1, GS1 and GS2 enhanced nitrogen assimilation in transgenic tobacco grown under low-nitrogen conditions. Plant Mol Biol Rep 31(4):886–900. https://doi.org/10.1007/s11105-013-0561-8
Wille A, Zimmermann P, Vranová E, Fürholz A, Laule O, Bleuler S, Hennig L, Prelić A, von Rohr P, Thiele L (2004) Sparse graphical Gaussian modeling of the isoprenoid gene network in Arabidopsis thaliana. Genome Biol 5(11):1–13. https://doi.org/10.1186/gb-2004-5-11-r92
Yin JL, Shackel NA, Zekry A, McGuinness PH, Richards C, Van Der Putten K, McCaughan GW, Eris JM, Bishop GA (2001) Real-time reverse transcriptase–polymerase chain reaction (RT–PCR) for measurement of cytokine and growth factor mRNA expression with fluorogenic probes or SYBR Green I. Immunol Cell Biol 79(3):213–221. https://doi.org/10.1046/j.1440-1711.2001.01002.x
Acknowledgements
This work was supported by Next Generation BioGreen 21 Program administered by Rural Development Administration, Republic of Korea (Grant PJ009549032014), through Systems and Synthetic Agrobiotech Center.
Author information
Authors and Affiliations
Contributions
SUP and S-UK conceived the study YBK, S-MK, RS, and Y-KK performed the experiments and analyzed the data. YBK, SUP, S-UK writing—review and editing. All of the authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any original research involving human participants or animals were harmed for this study by any of the authors.
Informed consent
This article does not contain any original research involving human participants or animals.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, Y.B., Kim, SM., Sathasivam, R. et al. Overexpression of Ginkgo biloba Hydroxy-2-methyl-2-(E)-butenyl 4-diphosphate reductase 2 gene (GbHDR2) in Nicotiana tabacum cv. Xanthi. 3 Biotech 11, 337 (2021). https://doi.org/10.1007/s13205-021-02887-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-021-02887-5