Abstract
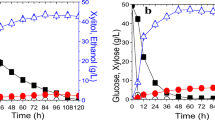
In a previously engineered Saccharomyces cerevisiae recombinant, the cellobiose fermentation rate was significantly lower than the glucose fermentation rate. Thus, we implemented a genome-wide perturbation library to find gene targets for improving the cellobiose fermentation capability of the yeast strain. Unexpectedly, we discovered a transformant that contained an additional β-glucosidase gene (gh1-1), possibly through homologous recombination between the plasmids. The additional β-glucosidase led to the fastest cellobiose fermentation activity among all the transformants evaluated, and the strain demonstrated significantly higher β-glucosidase activity than the control strain, especially during the initial exponential growth phase. The present work revealed the benefit of the extra gh1-1 copy for efficient cellobiose fermentation in the engineered S. cerevisiae strain.






Similar content being viewed by others
References
Al-Ahmad H (2018) Biotechnology for bioenergy dedicated trees: meeting future energy demands. Z Naturforsch C 73:15–32. https://doi.org/10.1515/znc-2016-0185
Alper H, Stephanopoulos G (2009) Engineering for biofuels: exploiting innate microbial capacity or importing biosynthetic potential? Nat Rev Microbiol 7:715–723. https://doi.org/10.1038/nrmicro2186
Amorim HV, Lopes ML, de Castro Oliveira JV, Buckeridge MS, Goldman GH (2011) Scientific challenges of bioethanol production in Brazil. Appl Microbiol Biotechnol 91:1267–1275. https://doi.org/10.1007/s00253-011-3437-6
Bohlin C, Olsen SN, Morant MD, Patkar S, Borch K, Westh P (2010) A comparative study of activity and apparent inhibition of fungal beta-glucosidases. Biotechnol Bioeng 107:943–952. https://doi.org/10.1002/bit.22885
Bohlin C, Praestgaard E, Baumann MJ, Borch K, Praestgaard J, Monrad RN, Westh P (2013) A comparative study of hydrolysis and transglycosylation activities of fungal beta-glucosidases. Appl Microbiol Biotechnol 97:159–169. https://doi.org/10.1007/s00253-012-3875-9
Borin GP, Sanchez CC, de Santana ES, Zanini GK, Dos Santos RAC, de Oliveira Pontes A, de Souza AT, Dal’Mas R, Riano-Pachon DM, Goldman GH, Oliveira JVC (2017) Comparative transcriptome analysis reveals different strategies for degradation of steam-exploded sugarcane bagasse by Aspergillus niger and Trichoderma reesei. BMC Genomics 18:501. https://doi.org/10.1186/s12864-017-3857-5
Carroll A, Somerville C (2009) Cellulosic biofuels. Annu Rev Plant Biol 60:165–182. https://doi.org/10.1146/annurev.arplant.043008.092125
Christianson TW, Sikorski RS, Dante M, Shero JH, Hieter P (1992) Multifunctional yeast high-copy-number shuttle vectors. Gene 110:119–122. https://doi.org/10.1016/0378-1119(92)90454-w
Clikeman JA, Khalsa GJ, Barton SL, Nickoloff JA (2001) Homologous recombinational repair of double-strand breaks in yeast is enhanced by MAT heterozygosity through yKU-dependent and -independent mechanisms. Genetics 157:579–589
David F, Siewers V (2015) Advances in yeast genome engineering. FEMS Yeast Res 15:1–14. https://doi.org/10.1111/1567-1364.12200
Florindo RN, Souza VP, Mutti HS, Camilo C, Manzine LR, Marana SR, Polikarpov I, Nascimento AS (2018) Structural insights into beta-glucosidase transglycosylation based on biochemical, structural and computational analysis of two GH1 enzymes from Trichoderma harzianum. N Biotechnol 40:218–227. https://doi.org/10.1016/j.nbt.2017.08.012
Galazka JM, Tian C, Beeson WT, Martinez B, Glass NL, Cate JH (2010) Cellodextrin transport in yeast for improved biofuel production. Science 330:84–86. https://doi.org/10.1126/science.1192838
Giebel BM, Swart PK, Riemer DD (2011) New insights to the use of ethanol in automotive fuels: a stable isotopic tracer for fossil- and bio-fuel combustion inputs to the atmosphere. Environ Sci Technol 45:6661–6669. https://doi.org/10.1021/es200982t
Ha SJ, Galazka JM, Kim SR, Choi JH, Yang X, Seo JH, Glass NL, Cate JH, Jin YS (2011) Engineered Saccharomyces cerevisiae capable of simultaneous cellobiose and xylose fermentation. Proc Natl Acad Sci USA 108:504–509. https://doi.org/10.1073/pnas.1010456108
Hiom K (2000) Homologous recombination. Curr Biol 10:R359–R361
Hong KK, Nielsen J (2012) Metabolic engineering of Saccharomyces cerevisiae: a key cell factory platform for future biorefineries. Cell Mol Life Sci 69:2671–2690. https://doi.org/10.1007/s00018-012-0945-1
Jeng WY, Wang NC, Lin MH, Lin CT, Liaw YC, Chang WJ, Liu CI, Liang PH, Wang AH (2011) Structural and functional analysis of three beta-glucosidases from bacterium Clostridium cellulovorans, fungus Trichoderma reesei and termite Neotermes koshunensis. J Struct Biol 173:46–56. https://doi.org/10.1016/j.jsb.2010.07.008
Jin YS, Alper H, Yang YT, Stephanopoulos G (2005) Improvement of xylose uptake and ethanol production in recombinant Saccharomyces cerevisiae through an inverse metabolic engineering approach. Appl Environ Microbiol 71:8249–8256. https://doi.org/10.1128/AEM.71.12.8249-8256.2005
Jones KL, Kim SW, Keasling JD (2000) Low-copy plasmids can perform as well as or better than high-copy plasmids for metabolic engineering of bacteria. Metabol Eng 2:328–338. https://doi.org/10.1006/mben.2000.0161
Kim SR, Ha SJ, Kong II, Jin YS (2012a) High expression of XYL2 coding for xylitol dehydrogenase is necessary for efficient xylose fermentation by engineered Saccharomyces cerevisiae. Metabol Eng 14:336–343. https://doi.org/10.1016/j.ymben.2012.04.001
Kim SR, Ha SJ, Wei N, Oh EJ, Jin YS (2012b) Simultaneous co-fermentation of mixed sugars: a promising strategy for producing cellulosic ethanol. Trends Biotechnol 30:274–282. https://doi.org/10.1016/j.tibtech.2012.01.005
Kim HJ, Turner TL, Jin YS (2013a) Combinatorial genetic perturbation to refine metabolic circuits for producing biofuels and biochemicals. Biotechnol Adv 31:976–985. https://doi.org/10.1016/j.biotechadv.2013.03.010
Kim SR, Park YC, Jin YS, Seo JH (2013b) Strain engineering of Saccharomyces cerevisiae for enhanced xylose metabolism. Biotechnol Adv 31:851–861. https://doi.org/10.1016/j.biotechadv.2013.03.004
Kim SJ, Kim JW, Lee YG, Park YC, Seo JH (2017) Metabolic engineering of Saccharomyces cerevisiae for 2,3-butanediol production. Appl Microbiol Biotechnol 101:2241–2250. https://doi.org/10.1007/s00253-017-8172-1
Kwak S, Jin YS (2017) Production of fuels and chemicals from xylose by engineered Saccharomyces cerevisiae: a review and perspective. Microb Cell Fact 16:82. https://doi.org/10.1186/s12934-017-0694-9
Lee KS, Hong ME, Jung SC, Ha SJ, Yu BJ, Koo HM, Park SM, Seo JH, Kweon DH, Park JC, Jin YS (2011) Improved galactose fermentation of Saccharomyces cerevisiae through inverse metabolic engineering. Biotechnol Bioeng 108:621–631. https://doi.org/10.1002/bit.22988
Lynd LR, Laser MS, Bransby D, Dale BE, Davison B, Hamilton R, Himmel M, Keller M, McMillan JD, Sheehan J, Wyman CE (2008) How biotech can transform biofuels. Nat Biotechnol 26:169–172. https://doi.org/10.1038/nbt0208-169
Martin MA (2010) First generation biofuels compete. N Biotechnol 27:596–608. https://doi.org/10.1016/j.nbt.2010.06.010
McClintock MK, Wang J, Zhang K (2017) Application of nonphosphorylative metabolism as an alternative for utilization of lignocellulosic biomass. Front Microbiol 8:2310. https://doi.org/10.3389/fmicb.2017.02310
Sasano Y, Nagasawa K, Kaboli S, Sugiyama M, Harashima S (2016) CRISPR-PCS: a powerful new approach to inducing multiple chromosome splitting in Saccharomyces cerevisiae. Sci Rep 6:30278. https://doi.org/10.1038/srep30278
Schuller D, Casal M (2005) The use of genetically modified Saccharomyces cerevisiae strains in the wine industry. Appl Microbiol Biotechnol 68:292–304. https://doi.org/10.1007/s00253-005-1994-2
Son HF, Lee SM, Kim KJ (2018) Crystal structure and biochemical characterization of xylose isomerase from Piromyces sp. E2. J Microbiol Biotechnol 28:571–578. https://doi.org/10.4014/jmb.1711.11026
Tang Y, Xia L, Ding X, Luo Y, Huang F, Jiang Y (2011) Duplication of partial spinosyn biosynthetic gene cluster in Saccharopolyspora spinosa enhances spinosyn production. FEMS Microbiol Lett 325:22–29. https://doi.org/10.1111/j.1574-6968.2011.02405.x
Tian C, Beeson WT, Iavarone AT, Sun J, Marletta MA, Cate JH, Glass NL (2009) Systems analysis of plant cell wall degradation by the model filamentous fungus Neurospora crassa. Proc Natl Acad Sci USA 106:22157–22162. https://doi.org/10.1073/pnas.0906810106
van Dijken JP, Bauer J, Brambilla L, Duboc P, Francois JM, Gancedo C, Giuseppin ML, Heijnen JJ, Hoare M, Lange HC, Madden EA, Niederberger P, Nielsen J, Parrou JL, Petit T, Porro D, Reuss M, van Riel N, Rizzi M, Steensma HY, Verrips CT, Vindelov J, Pronk JT (2000) An interlaboratory comparison of physiological and genetic properties of four Saccharomyces cerevisiae strains. Enzyme Microb Technol 26:706–714. https://doi.org/10.1016/S0141-0229(00)00162-9
Vigentini I, Gebbia M, Belotti A, Foschino R, Roth FP (2017) CRISPR/Cas9 system as a valuable genome editing tool for wine yeasts with application to decrease urea production. Front Microbiol 8:2194. https://doi.org/10.3389/fmicb.2017.02194
Wyman CE (2007) What is (and is not) vital to advancing cellulosic ethanol. Trends Biotechnol 25:153–157. https://doi.org/10.1016/j.tibtech.2007.02.009
Zhanga Z, Lohrb L, Escalanteb C, Wetzstein M (2010) Food versus fuel: what do prices tell us? Energy Policy 38:445–451
Acknowledgements
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2018R1D1A1B07051143) and by a fund from the Energy Biosciences Institute.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Rights and permissions
About this article
Cite this article
Kim, H.J., Lee, WH., Turner, T.L. et al. An extra copy of the β-glucosidase gene improved the cellobiose fermentation capability of an engineered Saccharomyces cerevisiae strain. 3 Biotech 9, 367 (2019). https://doi.org/10.1007/s13205-019-1899-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-019-1899-x