Abstract
Three types of honeys viz. Robinia pseudoacacia (RSA), Plectranthus rugosus (PR) and multifloral honey (MF) from Kashmir Himalayas of India were characterized on the basis of physicochemical, antioxidant and FTIR-ATR spectroscopic features. The physicochemical parameters indicated that ash content, electrical conductivity, redness (a*) and yellowness (b*) were higher in MF honey, moisture content, Hydroxymethylfurfural (HMF) and lightness (L*) in PR honey and Water activity (aw) and proline content in RSA honey (P ≤ 0.05). Significantly higher values of total sugar, total reducing sugar and sucrose content was observed in MF honey (P ≤ 0.05). The total phenolic content (TPC), total flavonoids, ferric reducing antioxidant power and 1, 1-diphenyl-2-picrylhydrazy-IC50 (DPPH- IC50) free radical scavenging activity showed that MF honey had significantly higher antioxidant activity (P ≤ 0.05). ATR-FTIR analysis demonstrated that the absence of peaks at broad band around 1653.454–1637.656 cm−1 in MF honey, 1185.989–1153.072 cm−1 and 918.570–916.544 cm−1 wavenumbers in PR honey thus clearly discriminating different types of honeys. The results revealed that physico-chemical, antioxidant and spectroscopic characteristics of honey types vary with geographic origin and their floral sources and MF honey have a high therapeutic potential.

Similar content being viewed by others
References
Abramovic H, Jamnik M, Burkan L, Kac M (2008) Water activity and water content in Slovenian honeys. Food Cont 19:1086–1090
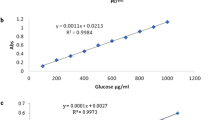
AOAC (2012) Official method of analysis, 19th edn. Association of official analytical chemists, Washington DC
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: the FRAP assay. Analy Biochem 239:70–76
Blasa M, Candiracci M, Accorsi A, Piacentini MP, Piatti E (2007) Honey flavonoids as protection agents against oxidative damage to human red blood cells. Food Chem 104:1635–1640
CAC (Codex Alimentarius Commission) (2001) Revised codex standard for honey. Codex Alimen 12–1981:1–7
Can Z, Yildiz O, Sahin H, Turumtay EA, Silici S, Kolayli S (2015) An investigation of Turkish honeys: their physico-chemical properties, antioxidant capacities and phenolic profiles. Food Chem 180:133–141
Chirife J, Zamora MC, Motto A (2006) The correlation between water activity and moisture in honey. Fundamental aspects and application to argentine honeys. J Food Engi 72:287–292
Chen HY, Lin YC, Hsieh CL (2007) Evaluation of antioxidant activity of aqueous extract of some selected nutraceutical herbs. Food Chem 104(4):1418–1424
Codex Alimentarius Commission Standards (2001) CODEX STAN 12-1981, Rev.1 (1987), Rev.2.
Di Marco G, Gismondi A, Canuti L, Scimeca M, Volpe A, Canini A (2014) Tetracycline accumulates in Iberis sempervirens L. through apoplastic transport inducing oxidative stress and growth inhibition. Plant Biol 16:792–800
do Nascimentoa KS, Sattler JAG, Macedo LFL, González CVS, de Melo ILP, da Silva Araújo E, Granato D, Sattler A, de Almeida-Muradiana LB, (2018) Phenolic compounds, antioxidant capacity and physicochemical properties of Brazilian Apis mellifera honeys. LWT Food Sci Tech 91:85–94
Escriche I, Tanleque-Alberto F, Visquert M, Mircea Oroian M (2017) Physicochemical and rheological characterization of honey from Mozambique. LWT Food Sci Tech 86:108–115
Estevinho L, Pereira AL, Moreira L, Dias LG, Pereira E (2008) Antioxidant and antimicrobial effects of phenolic compounds extracts of Northeast Portugal honey. Food Chem Toxicol 46:3774–3779
European Directive (2002) Council Directive 2001/110/CE of 20 December 2001. Off J Eur Commun, 12 January2002; L10/47.
Fechner DC, Hidalgo MJ, Diaz JDR, Gil RA, Pellerano RG (2020) Geographical origin authentication of honey produced in Argentina. Food Biosci 33:100483
Finola MS, Lasagno MC, Marioli JM (2007) Microbiological and chemical characterization of honeys from central Argentina. Food Chem 100:1649–1653
Fukumoto LR, Mazza G (2000) Assessing antioxidant and prooxidant activities of phenolic compounds. J Agric Food Chem 48:3597–3604
Geana EI, Ciucure CT (2020) Establishing authenticity of honey via comprehensive Romanian honey analysis. Food Chem 306:125595
Gilbert HZ, Silici J, Charlton S, Dal A, Gürel C (2009) Profiling Turkish honeys to determine authenticity using physical and chemical characteristics. J Agric Food Chem 57:3911–3919
Gok S, Severcan M, Goormaghtigh E, Kandemir I, Severcan F (2015) Differentiation of Anatolian honey samples from different botanical origins by ATR-FTIR spectroscopy using multivariate analysis. Food Chem 170:234–240
Gonzalez-Miret ML, Terrab A, Hernanz D, Fernandez-Recamales MA, Heredia FJ (2005) Multivariate correlation between colour and mineral composition of honeys and by their botanical origin. J Agric Food Chem 53:2575–2580
IRAM. Instituto de Racionalizacion Argentino de Materiales (1996) Argentina. Retrieved from https://www.iram.org.ar/seccion.php?ID=3&IDS=42.
Jandrić Z, Haughey SA, Frew RD, McComb K, Galvin-King P, Elliott CT (2015) Discrimination of honey of different floral origins by a combination of various chemical parameters. Food Chem 189:52–59
Kamboj R, Bera MB, Nanda V (2013) Evaluation of physico-chemical properties, trace metal content and antioxidant activity of Indian honeys. Inter J Food Sci Tech 48:578–587
Kaškoniene V, Venskutonis PR, Ceksteryt V (2010) Carbohydrate composition and electrical conductivity of different origin honeys from Lithuania. LWT Food Sci Tech 43:801–807.
Kasprzyk I, Depciuch J, Grabek-Lejko D, Parlinska-Wojtan M (2018) FTIR-ATR spectroscopy of pollen and honey as a tool for unifloral honey authentication. The case study of rape honey. Food Cont 84:33–40
Kucuk M, Kolayl S, Karaoglu S, Ulusoy E, Baltac C, Candan F (2007) Biological activities and chemical composition of three honeys of different types from Anatolia. Food Chem 100:526–534
Lebuhn G, Droege S, Connor EF, Gemmill-Herren B, Potts SG, Minckley RT (2013) Detecting insect pollinator declines on regional and global scales. Conserv Biol 27:113–120
Machado De-Melo AA, de Almeida-Muradian LB, Sancho MT, Pascual-Maté A (2017). Composition and properties of Apis mellifera honey: a review. J Apicul Res. https://doi.org/10.1080/00218839.2017.1338444
Masek A, Chrzescijanska E, Kosmalska A, Zaborski M (2014) Characteristics of compounds in hops using cyclic voltammetry, UV–VIS, FTIR and GC–MS analysis. Food Chem 156:353–361
Marco GD, Manfredini A, Leonardi D, Canuti L, Impei S, Gismondi A, Canini A (2017) Geographical, botanical and chemical profile of monofloral Italian honeys as food quality guarantee and territory brand. Plant Biosyst Inter J Dealing Aspects Plant Biol 151(3):450–463
Mossel B, Bhandari BD, Arcy B, Caffin N (2003) Determination of viscosity of some Australian Honeys based on composition. Int J Food Prop 6:87–97
Nayik GA, Dar BN, Nanda V (2015) Physicochemical, rheological and sugar profile of different unifloral honeys from Kashmir valley of India. Arab J Chem. https://doi.org/10.1016/j.arabjc.2015.08.017
Ozcan MM, Olmez C (2014) Some qualitative properties of different monofloral honeys. Food Chem 163:212–218
Rodriguez I, Camara-Martos F, Flores JM, Serrano S (2019) Spanish avocado (Persea americana Mill.) honey: authentication based on its composition criteria, mineral content and sensory attributes. LWT Food Sci Tech 111:561–572
Saxena S, Gautam S, Sharma A (2010) Physical, biochemical and antioxidant properties of some Indian honeys. Food Chem 118:391–397
Sharma D, Abrol DP, Ahmad H, Bandral RS, Ishar A (2014) Traditional Beekeeping In Jammu And Kashmir. India Bee World 91(3):1–4
Siddiqui AJ, Musharraf SG, Choudhary MI, Rahman A-u (2017) Application of analytical methods in authentication and adulteration of honey. Food Chem 217:687–698
Singleton VL, Orthofer R, Lamuela-Raventós RM (1999) Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol 299:152–178
Stagos D, Soulitsiotis N, Tsadila C, Papaeconomou S, ArvanitisC NA, Karkanta F, Adamou-Androulak S, Spetrotos K, Spandidos DA, Kouretas D, Mossialos D (2018) Antibacterial and antioxidant activity of different types of honey derived from Mount Olympus in Greece. Inter J Mol Med 42:726–734
Tahir HE, Xiaobo Z, Zhihua L, Jiyong S, Zhai X, Wang S, Mariod AA (2017) Rapid prediction of phenolic compounds and antioxidant activity of Sudanese honey using Raman and Fourier transform infrared (FT-IR) spectroscopy. Food Chem 226:202–211
Tewari J, Irudayaraj J (2005) Quantification of saccharides in multiple floral honey using Fourier transform infrared microattenuated total reflectance spectroscopy. J Agricul Food Chem 52:3237–3243
Umegaki T, Hui Sz M, Kojima Y (2017) Fabrication of hollow silicaenickel particles for the hydrolytic dehydrogenation of ammonia borane using rape pollen templates. New J Chem 41:992–996
Yap SK, Chin NL, Yusof YA, Chong KY (2019) Quality characteristics of dehydrated raw Kelulut honey. International J Food Prop 22(1):556–571
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ganaie, T.A., Masoodi, F.A., Rather, S.A. et al. Physicochemical, antioxidant and FTIR-ATR spectroscopy evaluation of Kashmiri honeys as food quality traceability and Himalayan brand. J Food Sci Technol 58, 4139–4148 (2021). https://doi.org/10.1007/s13197-020-04878-5
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-020-04878-5