Abstract
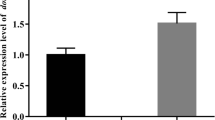
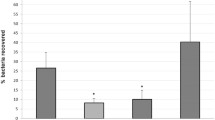
Chemotactic motility is involved in the virulence of many pathogenic bacteria. In order to understand the role of chemotactic motility of Vibrio harveyi in cellular processes and virulence, mini-Tn10/Kan transposon-induced mutants with deficient chemotactic motility were constructed, screened, and identified. Sequence analysis revealed that the 465-bp fragment (Gen Bank accession number HM630274) flanking the transposon insertion site in mutant TS-CM1 had the highest identity (96.9%) with a hypothetical protein gene of V. harveyi ATCC BAA-1116 and the second-highest identity (91.8%) with the pgk gene of V. parahaemolyticus RIMD 2210633. In another mutant, TS-CM2, 356 bp of transposon-flanking sequence (Gen Bank accession number HM630275) also showed the highest identity (94.6%) with a hypothetical protein gene of V. harveyi ATCC BAA-1116 and the second-highest identity (92.4%) with the flaB gene of V. alginolyticus HY9901. Studies on virulence-related biological characteristics such as growth, motility, adhesion, and infectivity of themutants showed that disruption of either the flagellin gene or energy metabolism gene led to subsequent loss of chemotactic motility and changes in growth, motility, adhesion, and virulence of the pathogenic V. harveyi. Hence, the flagellin gene and crucial energy metabolism gene played an important role in the chemotactic motility of V. harveyi.
Similar content being viewed by others
References
Austin B, Zhang Xiaohua. 2006. Vibrio harveyi: a significant pathogen of marine vertebrates and invertebrates. Lett Appl Microbiol, 43: 119–124
Balebona MC, MoriNigo MA, Faris A, et al. 1995. Influence of salinity and pH on the adhesion of pathogenic Vibrio strains to Sparus aurata skin mucus. Aquaculture, 132: 113–120
Banks R D, Blake C C, Evans P R, et al. 1979. Sequence, structure and activity of phosphoglycerate kinase: a possible hinge-bending enzyme. Nature, 279: 773–777
Boin M A, Austin M J, Hāse C C. 2004. Chemotaxis in Vibrio cholerae. FEMS Micro Lett, 239: 1–8
Bordas MA, Balebona MC, Rodriguez-Maroto JM, et al. 1998. Chemotaxis of pathogenic Vibrio strains towards mucus surfaces of gilt-head sea bream(Sparus aurata L.). Appl Environ Microbiol, 64: 1573–1575
Butler S M, Camilli A. 2004. Both chemotaxis and net motility greatly influence the infectivity of Vibrio cholerae. PNAS, 101: 5018–5023
Celani A, Vergassola M. 2010. Bacterial strategies for chemotaxis response. PANS, 107(4): 1391–1396
Deeraksa A, Moonmangmee S, Toyama H, et al. 2005. Characterization and spontaneous mutation of a novel gene, polE, involved in pellicle formation in Acetobacter tropicalis SKU1100. Microbiology, 151: 4111–4120
Haniford D B. 2006. Transpososome dynamics and regulation in Tn10 transposition. Crit Rev Biochem Mol Biol, 42: 407–424
Herrero M, Lorenzo V, Timmis K N. 1990. Transposon vectors containing non-antibiotic resistance selection markers for cloning and stable chromosomal insertion of foreign genes in gram-negative bacteria. J Bacteriol, 172: 6557–6567
Josenhans C, Labigne A, Suerbaum S. 1995. Comparative ultrastructural and functional studies of Helicobacter pylory and Helicobacter mustelae flagellin mutants: both flagellin subunits, FlaA and FlaB, are necessary for full motility in Helicobacter species. J Bacteriol, 177: 3010–3020
Josenhans C, Suerbaum S. 2002. The role of motility as a virulence factor in bacteria. Int J Med Microbiol, 291: 605–614
Khanna M R, Bhavsar S P, Kapadnis B P. 2006. Effect of temperature on growth and chemotactic behaviour of Campylobacter jejuni. Lett Appl Microbiol, 43: 84–90
Kirby J R. 2009. Chemotaxis-like regulatory systems: unique roles in diverse bacteria. Annual Review of Microbiology, 63: 45–59
Kleckner N. 1989. Transposon Tn10. In: Berg D E, Howe M M, eds. Mobile DNA. Washington D C: American Society for Microbiology Press
Klesius P H, Shoemaker C A, Evans J J. 2008. Flavobacterium columnare chemotaxis to channel catfish mucus. FEMS Microbiology Letters, 288(2): 216–220
Lacal J, Muñoz-Martínez F, Reyes-Darías J, et al. 2011. Bacterial chemotaxis towards aromatic hydrocarbons in Pseudomonas. Environmental Microbiology, 13(7): 1733–1744
Larsen M H, Larsen J L, Olsen J F. 2001. Chemotaxis of Vibrio anguillarum to fish mucus: role of the origin of the fish mucus, the fish species and the serogroup of the pathogen. FEMS Microbiol Ecol, 38: 77–80
Lee S E, Kim S Y, Jeong B C, et al. 2006. A bacterial flagellin, Vibrio vulnificus FlaB, has a strong mucosal adjuvant activity to induce protective immunity. Infec Immun, 74: 694–702
Liu Yaoguang, Mitsukawa N, Oosumi T, et al. 1995. Efficient isolation and mapping of Arabidopsis thaliana T-DNA insert junctions by thermal asymmetric interlaced PCR. Plant J, 8: 457–463
Lux R, Moter A, Shi Wenyuan. 2000. Chemotaxis in pathogenic spirochetes: directed movement toward targeting tissues. J Mol Microbiol Biotechnol, 2: 355–364
Lux R, Shi Wenyuan. 2004. Chemotaxis-guided movements in bacteria. Crit Rev Oral Biol Med, 15: 207–220
Macnab R M. 1996. Flagella and motility. In: Neidhardt F C, ed. Escherichia coli and Salmonella: Cellular and Molecular Biology. Washington D C: ASM Press
Merrell D S, Butler S M, Qadri F, et al. 2002. Host-induced epidemic spread of the cholera bacterium. Nature, 417: 642–645
McCarter L L. 2001. Polar flagellarmotility of the Vibrionaceae. Microbiol Mol Biol Rev, 65: 445–462
O’Toole R, Miotom D L, Wolf-Watz H. 1996. Chemotactic motility is required for invasion of the host by the fish pathogen Vibrio anguillarum. Mol Microbiol, 19: 625–637
Porter S L, Wadhams G H, Armitage J P. 2011. Signal processing in complex chemotaxis pathways. Nature Reviews Microbiology, 9: 153–165
Qin Yingxue, Wang Jun, Su Yongquan, et al. 2006. Studies on the pathogenic bacterium of ulcer disease in Epinephelus awoara. Acta Oceanol Sin, 25: 147–153
Rock J L, Nelson D R. 2006. Identification and characterization of a hemolysin gene cluster in Vibrio anguillarum. Infec Immun, 74: 2777–2786
Salvetti S, Celandroni F, Ceragioli M, et al. 2009. Identification of non-flagellar genes involved in swarm cell differentiation using a Bacillus thuringiensis mini-Tn10 mutant library. Microbiology, 155: 912–921
Sambrook J, Fritsch E F, Maniatis T. 1989. Molecular Cloning: A LaboratoryManual. NewYork: Cold Spring Harbor Laboratory Press
Sawabe T, Inoue S, Fukui Y, et al. 2007. Mass mortality of Japanese abalone Haliotis discus hannai caused by Vibrio harveyi infection. Microbes Environ, 22: 300–308
Spohn G, Scarlato V. 2001. Motility, Chemotaxis, and Flagella. In: Mobley H L T, Mendz G L, Haze U S L, eds. Helicobacter pylory: Physiology and Genetics. Washington D C: ASM Press
Valenzuela M, Cerda O, Toledo H. 2003. Overview on chemotaxis and acid resistance in Helicobacter pylori. Biol Res, 36: 429–436
Vine NG, Leukes WD, Kaiser H. 2004. In vitro growth characteristics of five candidates aquaculture probiotics and two fish pathogens grown in fish intestinal mucus. FEMS Microbiol Lett, 231: 145–152
Vladimirov N, Sourjik V. 2009. Chemotaxis: how bacteria usememory. Biological Chemistry, 390(11): 1097–1104
Xie L, Altindal T, Chattopadhyay S, et al. 2011. Bacterial flagellum as a propeller and as a rudder for efficient chemotaxis. PNAS, 108(6): 2246–2251
Yan Qingpi, Zhao Minhui, Wang Xiaolu, et al. 2010. Adhesion mechanisms of Vibrio fluvialis to skin mucus of Epinephelus awoara. Chinese Journal of Oceanology and Limnology, 28(2): 260–266
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: The National Natural Science Foundation of China under contract Nos 31272699 and 41176115; National Department Public Benefit Research Foundation of China under contract No. 200903029; the Natural Science Foundation of Fujian Province under contract No. 2011J06014; the National Hi-Tech Research and Development Programof China (863 Program) under contract No. 2007AA09Z115.
Rights and permissions
About this article
Cite this article
Qin, Y., Yan, Q., Su, Y. et al. Disruption of chemotaxis-related genes affects multiple cellular processes and the virulence of pathogenic Vibrio harveyi . Acta Oceanol. Sin. 32, 55–60 (2013). https://doi.org/10.1007/s13131-013-0341-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13131-013-0341-1