Abstract
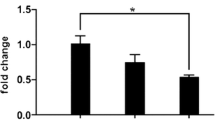
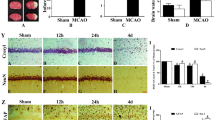
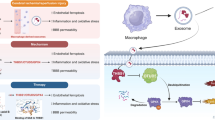
Cardiovascular diseases are approximately three times higher in patients with neurological deficits than in patients without neurological deficits. MicroRNA-126 (MiR-126) facilitates vascular remodeling and decreases fibrosis and is emerging as an important factor in the pathogenesis of cardiovascular diseases and cerebral stroke. In this study, we tested the hypothesis that decreased miR-126 after ischemic stroke may play an important role in regulating cardiac function. Wild-type (WT), specific conditional-knockout endothelial cell miR-126 (miR-126EC−/−), and miR-126 knockout control (miR-126fl/fl) mice were subjected to distal middle cerebral artery occlusion (dMCAo) (n = 10/group). Cardiac hemodynamics and function were measured using transthoracic Doppler echocardiography. Mice were sacrificed at 28 days after dMCAo. WT mice subjected to stroke exhibited significantly decreased cardiac ejection fraction and increased myocyte hypertrophy, fibrosis as well as increased heart inflammation, infiltrating macrophages, and oxidative stress compared to non-stroke animals. Stroke significantly decreased serum and heart miR-126 expression and increased miR-126 target genes, vascular cell adhesion protein-1, and monocyte chemotactic protein-1 gene, and protein expression in the heart compared to non-stroke mice. MiR-126EC−/− mice exhibited significantly decreased cardiac function and increased cardiomyocyte hypertrophy, fibrosis, and inflammatory factor expression after stroke compared to miR-126fl/fl stroke mice. Exosomes derived from endothelial cells of miR-126EC−/− (miR-126EC−/−EC-Exo) mice exhibited significantly decreased miR-126 expression than exosomes derived from miR-126fl/fl (miR-126fl/fl-EC-Exo) mice. Treatment of cardiomyocytes subjected to oxygen glucose deprivation with miR-126fl/fl-EC-Exo exhibited significantly decreased hypertrophy than with miR-126EC−/−EC-Exo treatment. Ischemic stroke directly induces cardiac dysfunction. Decreasing miR-126 expression may contribute to cardiac dysfunction after stroke in mice.






Similar content being viewed by others
Explore related subjects
Discover the latest articles and news from researchers in related subjects, suggested using machine learning.References
Koh SH, Park HH. Neurogenesis in stroke recovery. Transl Stroke Res. 2016; doi:10.1007/s12975-016-0460-z.
Cassidy JM, Cramer SC. Spontaneous and therapeutic-induced mechanisms of functional recovery after stroke. Transl Stroke Res. 2016; doi:10.1007/s12975-016-0467-5.
Wira 3rd CR, Rivers E, Martinez-Capolino C, Silver B, Iyer G, Sherwin R, et al. Cardiac complications in acute ischemic stroke. The Western Journal of Emergency Medicine. 2011;12(4):414–20. doi:10.5811/westjem.2011.2.1765.
Imam YZ, D’Souza A, Malik RA, Shuaib A. Secondary stroke prevention: improving diagnosis and management with newer technologies. Transl Stroke Res. 2016;7(6):458–77. doi:10.1007/s12975-016-0494-2.
Ergul A, Hafez S, Fouda A, Fagan SC. Impact of comorbidities on acute injury and recovery in preclinical stroke research: focus on hypertension and diabetes. Transl Stroke Res. 2016;7(4):248–60. doi:10.1007/s12975-016-0464-8.
Ay H, Koroshetz WJ, Benner T, Vangel MG, Melinosky C, Arsava EM, et al. Neuroanatomic correlates of stroke-related myocardial injury. Neurology. 2006;66(9):1325–9. doi:10.1212/01.wnl.0000206077.13705.6d.
Oppenheimer SM. Neurogenic cardiac effects of cerebrovascular disease. Curr Opin Neurol. 1994;7(1):20–4.
Ishikawa H, Tajiri N, Vasconcellos J, Kaneko Y, Mimura O, Dezawa M, et al. Ischemic stroke brain sends indirect cell death signals to the heart. Stroke. 2013;44(11):3175–82. doi:10.1161/STROKEAHA.113.001714.
Gongora-Rivera F, Labreuche J, Jaramillo A, Steg PG, Hauw JJ, Amarenco P. Autopsy prevalence of coronary atherosclerosis in patients with fatal stroke. Stroke. 2007;38(4):1203–10. doi:10.1161/01.str.0000260091.13729.96.
Tokgozoglu SL, Batur MK, Topcuoglu MA, Saribas O, Kes S, Oto A. Effects of stroke localization on cardiac autonomic balance and sudden death. Stroke. 1999;30(7):1307–11.
Swerdel JN, Janevic TM, Kostis WJ, Faiz A, Cosgrove NM, Kostis JB. Association between dehydration and short-term risk of ischemic stroke in patients with atrial fibrillation. Transl Stroke Res. 2016; doi:10.1007/s12975-016-0471-9.
Oppenheimer SM, Gelb A, Girvin JP, Hachinski VC. Cardiovascular effects of human insular cortex stimulation. Neurology. 1992;42(9):1727–32.
Rosenzweig S, Carmichael ST. Age-dependent exacerbation of white matter stroke outcomes: a role for oxidative damage and inflammatory mediators. Stroke. 2013;44(9):2579–86. doi:10.1161/STROKEAHA.113.001796.
Mishra PK, Tyagi N, Kumar M, Tyagi SC. MicroRNAs as a therapeutic target for cardiovascular diseases. J Cell Mol Med. 2009;13(4):778–89. doi:10.1111/j.1582-4934.2009.00744.x.
Schober A, Nazari-Jahantigh M, Wei Y, Bidzhekov K, Gremse F, Grommes J, et al. MicroRNA-126-5p promotes endothelial proliferation and limits atherosclerosis by suppressing Dlk1. Nat Med. 2014;20(4):368–76. doi:10.1038/nm.3487.
Hsu A, Chen SJ, Chang YS, Chen HC, Chu PH. Systemic approach to identify serum microRNAs as potential biomarkers for acute myocardial infarction. Biomed Res Int. 2014;2014:418628. doi:10.1155/2014/418628.
Long G, Wang F, Li H, Yin Z, Sandip C, Lou Y, et al. Circulating miR-30a, miR-126 and let-7b as biomarker for ischemic stroke in humans. BMC Neurol. 2013;13:178. doi:10.1186/1471-2377-13-178.
Wei XJ, Han M, Yang FY, Wei GC, Liang ZG, Yao H, et al. Biological significance of miR-126 expression in atrial fibrillation and heart failure. Braz J Med Biol Res. 2015;48(11):983–9. doi:10.1590/1414-431X20154590.
Kuraoka M, Furuta T, Matsuwaki T, Omatsu T, Ishii Y, Kyuwa S, et al. Direct experimental occlusion of the distal middle cerebral artery induces high reproducibility of brain ischemia in mice. Experimental animals/Japanese Association for Laboratory Animal Science. 2009;58(1):19–29.
Rosell A, Agin V, Rahman M, Morancho A, Ali C, Koistinaho J, et al. Distal occlusion of the middle cerebral artery in mice: are we ready to assess long-term functional outcome? Transl Stroke Res. 2013;4(3):297–307. doi:10.1007/s12975-012-0234-1.
Claxton S, Kostourou V, Jadeja S, Chambon P, Hodivala-Dilke K, Fruttiger M. Efficient, inducible Cre-recombinase activation in vascular endothelium. Genesis. 2008;46(2):74–80. doi:10.1002/dvg.20367.
Kuhnert F, Mancuso MR, Hampton J, Stankunas K, Asano T, Chen CZ, et al. Attribution of vascular phenotypes of the murine Egfl7 locus to the microRNA miR-126. Development. 2008;135(24):3989–93. doi:10.1242/dev.029736.
Yang XP, Liu YH, Rhaleb NE, Kurihara N, Kim HE, Carretero OA. Echocardiographic assessment of cardiac function in conscious and anesthetized mice. Am J Phys. 1999;277(5 Pt 2):H1967–74.
Swanson RA, Morton MT, Tsao-Wu G, Savalos RA, Davidson C, Sharp FR. A semiautomated method for measuring brain infarct volume. J Cereb Blood Flow Metab. 1990;10(2):290–3.
Xu J, Carretero OA, Liao TD, Peng H, Shesely EG, Xu J, et al. Local angiotensin II aggravates cardiac remodeling in hypertension. Am J Physiol Heart Circ Physiol. 2010;299(5):H1328–38. doi:10.1152/ajpheart.00538.2010.
Mito S, Ozono R, Oshima T, Yano Y, Watari Y, Yamamoto Y, et al. Myocardial protection against pressure overload in mice lacking Bach1, a transcriptional repressor of heme oxygenase-1. Hypertension. 2008;51(6):1570–7. doi:10.1161/HYPERTENSIONAHA.107.102566.
Belostotskaya GB, Golovanova TA. Characterization of contracting cardiomyocyte colonies in the primary culture of neonatal rat myocardial cells: a model of in vitro cardiomyogenesis. Cell Cycle. 2014;13(6):910–8. doi:10.4161/cc.27768.
Cui X, Chopp M, Zacharek A, Ye X, Roberts C, Chen J. Angiopoietin-Tie2 pathway mediates type 2 diabetes induced vascular damage after cerebral stroke. Neurobiol Dis. 2011;43(1):285–92. doi:10.1016/j.nbd.2011.04.005.
Xu J, Sun Y, Carretero OA, Zhu L, Harding P, Shesely EG, et al. Effects of cardiac overexpression of the angiotensin II type 2 receptor on remodeling and dysfunction in mice post-myocardial infarction. Hypertension. 2014;63(6):1251–9.
Chen J, Ning R, Zacharek A, Cui C, Cui X, Yan T, et al. MiR-126 contributes to human umbilical cord blood cell induced Neurorestorative effects after stroke in type-2 diabetic mice. Stem Cells. 2015; doi:10.1002/stem.2193.
Arner E, Mejhert N, Kulyte A, Balwierz PJ, Pachkov M, Cormont M, et al. Adipose tissue microRNAs as regulators of CCL2 production in human obesity. Diabetes. 2012;61(8):1986–93. doi:10.2337/db11-1508.
Witkowski M, Weithauser A, Tabaraie T, Steffens D, Krankel N, Witkowski M, et al. Micro-RNA-126 reduces the blood thrombogenicity in diabetes mellitus via targeting of tissue factor. Arterioscler Thromb Vasc Biol. 2016; doi:10.1161/ATVBAHA.115.306094.
Harris TA, Yamakuchi M, Ferlito M, Mendell JT, Lowenstein CJ. MicroRNA-126 regulates endothelial expression of vascular cell adhesion molecule 1. Proc Natl Acad Sci U S A. 2008;105(5):1516–21. doi:10.1073/pnas.0707493105.
Rauh R, Fischereder M, Spengel FA. Transesophageal echocardiography in patients with focal cerebral ischemia of unknown cause. Stroke. 1996;27(4):691–4.
Kim JM, Jung KH, Chu K, Lee ST, Ban J, Moon J, et al. Atherosclerosis-related circulating MicroRNAs as a predictor of stroke recurrence. Transl Stroke Res. 2015;6(3):191–7. doi:10.1007/s12975-015-0390-1.
Turchinovich A, Samatov TR, Tonevitsky AG, Burwinkel B. Circulating miRNAs: cell-cell communication function? Front Genet. 2013;4:119. doi:10.3389/fgene.2013.00119.
Peng G, Yuan Y, Wu S, He F, Hu Y, Luo B. MicroRNA let-7e is a potential circulating biomarker of acute stage ischemic stroke. Transl Stroke Res. 2015;6(6):437–45. doi:10.1007/s12975-015-0422-x.
Small EM, Frost RJA, Olson EN. MicroRNAs add a new dimension to cardiovascular disease. Circulation. 2010;121(8):1022–32. doi:10.1161/CIRCULATIONAHA.109.889048.
Fei L, Zhang J, Niu H, Yuan C, Ma X. Effects of rosuvastatin and MiR-126 on myocardial injury induced by acute myocardial infarction in rats: role of vascular endothelial growth factor a (VEGF-A). Medical Science Monitor: International Medical Journal of Experimental and Clinical Research. 2016;22:2324–34. doi:10.12659/MSM.896983.
Olivieri F, Spazzafumo L, Bonafe M, Recchioni R, Prattichizzo F, Marcheselli F, et al. MiR-21-5p and miR-126a-3p levels in plasma and circulating angiogenic cells: relationship with type 2 diabetes complications. Oncotarget. 2015;6(34):35372–82. doi:10.18632/oncotarget.6164.
Chen F, Du Y, Esposito E, Liu Y, Guo S, Wang X, et al. Effects of focal cerebral ischemia on exosomal versus serum miR126. Transl Stroke Res. 2015;6(6):478–84. doi:10.1007/s12975-015-0429-3.
Long G, Wang F, Duan Q, Chen F, Yang S, Gong W, et al. Human circulating microRNA-1 and microRNA-126 as potential novel indicators for acute myocardial infarction. Int J Biol Sci. 2012;8(6):811–8. doi:10.7150/ijbs.4439.
Potus F, Ruffenach G, Dahou A, Thebault C, Breuils-Bonnet S, Tremblay E, et al. Downregulation of microRNA-126 contributes to the failing right ventricle in pulmonary arterial hypertension. Circulation. 2015;132(10):932–43. doi:10.1161/CIRCULATIONAHA.115.016382.
Silvani A, Calandra-Buonaura G, Dampney RA, Cortelli P. Brain-heart interactions: physiology and clinical implications. Philos Trans A Math Phys Eng Sci. 2016;374(2067) doi:10.1098/rsta.2015.0181.
Daniele O, Caravaglios G, Fierro B, Natale E. Stroke and cardiac arrhythmias. J Stroke Cerebrovasc Dis. 2002;11(1):28–33. doi:10.1053/jscd.2002.123972.
Liesz A, Kleinschnitz C. Regulatory T cells in post-stroke immune homeostasis. Transl Stroke Res. 2016;7(4):313–21. doi:10.1007/s12975-016-0465-7.
Atangana E, Schneider UC, Blecharz K, Magrini S, Wagner J, Nieminen-Kelha M, et al. Intravascular inflammation triggers intracerebral activated microglia and contributes to secondary brain injury after experimental subarachnoid hemorrhage (eSAH). Transl Stroke Res. 2016; doi:10.1007/s12975-016-0485-3.
Ahmad M, Graham SH. Inflammation after stroke: mechanisms and therapeutic approaches. Transl Stroke Res. 2010;1(2):74–84. doi:10.1007/s12975-010-0023-7.
Yao Y, Tsirka SE. Chemokines and their receptors in intracerebral hemorrhage. Transl Stroke Res. 2012;3(1):70–9. doi:10.1007/s12975-012-0155-z.
Lambert JM, Lopez EF, Lindsey ML. Macrophage roles following myocardial infarction. Int J Cardiol. 2008;130(2):147–58. doi:10.1016/j.ijcard.2008.04.059.
Mewhort HE, Lipon BD, Svystonyuk DA, Teng G, Guzzardi DG, Silva C, et al. Monocytes increase human cardiac myofibroblast-mediated extracellular matrix remodeling through TGF-beta1. Am J Physiol Heart Circ Physiol. 2016;310(6):H716–24. doi:10.1152/ajpheart.00309.2015.
Bujak M, Frangogiannis NG. The role of TGF-beta signaling in myocardial infarction and cardiac remodeling. Cardiovasc Res. 2007;74(2):184–95. doi:10.1016/j.cardiores.2006.10.002.
Liu Y, Zhang J. Nox2 contributes to cardiac fibrosis in diabetic cardiomyopathy in a transforming growth factor-beta dependent manner. Int J Clin Exp Pathol. 2015;8(9):10908–14.
Acknowledgements
The authors wish to thank Cynthia Roberts, Qinge Lu, and Sutapa Santra for the technical assistance. This work was supported by National Institute of Neurological Disorders and Stroke R01 NS083078-01A1 (JC) and R01 NS099030-01 (JC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Jieli Chen declares that she has no conflict of interest. Chengcheng Cui declares that she has no conflict of interest. Xiaoping Yang declares that she has no conflict of interest. Jiang Xu declares that he has no conflict of interest. Poornima Venkat declares that she has no conflict of interest. Alex Zacharek declares that he has no conflict of interest. Peng Yu declares that he has no conflict of interest. Michael Chopp declares that he has no conflict of interest.
Rights and permissions
About this article
Cite this article
Chen, J., Cui, C., Yang, X. et al. MiR-126 Affects Brain-Heart Interaction after Cerebral Ischemic Stroke. Transl. Stroke Res. 8, 374–385 (2017). https://doi.org/10.1007/s12975-017-0520-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12975-017-0520-z