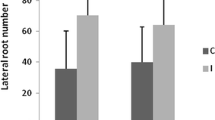
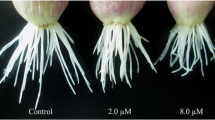
Abstract
Plant growth-promoting rhizobacteria (PGPR) are considered to have a beneficial effect on host plants. Azospirillum brasilense is a diazotroph associated with important agricultural crops such as maize and wheat, and has been used as model organism to investigate associative plant growth promotion. In this study, we have employed scanning electronic microscopic to observe the presence of bacterium on the root surface. Two-dimensional gel electrophoresis and mass spectrometry were used to analyze the changes in protein profile of maize roots in response to A. brasilense strain FP2 7 days after inoculation and we observed 46 differently expressed protein spots. We report the identification of three of these proteins.
Similar content being viewed by others
Abbreviations
- PGPR:
-
plant growth-promoting rhizobacteria
- DAI:
-
days after inoculation
References
Alberton D, Mueller-Santos M, Campos Brusamarello-Santos LC, Valdameri G, Cordeiro FA, Yates MG, Pedrosa FdO, de Souza EM. 2013. Comparative proteomics analysis of the rice roots colonized by Herbaspirillum seropedicaestrain SmR1 reveals induction of the methionine recycling in the plant host. J. Proteome Res. 12: 4757–4768
Arruda L, Beneduzi A, Martins A, Lisboa B, Lopes C, Bertolo F, Passaglia L, Vargas L. 2013. Screening of rhizobacteria isolated from maize (Zea mays L.) in Rio Grande do Sul State (South Brazil) and analysis of their potential to improve plant growth. Appl. Soil Ecol. 63: 15–22
Arruebarrena Di Palma A, Pereyra CM, Moreno Ramirez L, Xiqui Vazquez ML, Baca BE, Pereyra MA, Lamattina L, Creus CM. 2013. Denitrification-derived nitric oxide modulates biofilm formation in Azospirillum brasilense. FEMS Microbiol. Lett. 338: 77–85
Arzanesh MH, Alikhani HA, Khavazi K, Rahimian HA, Miransari M. 2011 Wheat (Triticum aestivum L.) growth enhancement by Azospirillum sp under drought stress World J. Microbiol. Biotechnol. 27: 197–205
Balsanelli E, Serrato RV, de Baura VA, Sassaki G, Yates MG, Rigo LU, Pedrosa FO, de Souza EM, Monteiro RA. 2010. Herbaspirillum seropedicae rfbB and rfbC genes are required for maize colonization. Environ. Microbiol. 12: 2233–2244
Bashan Y, de-Bashan L, Sparks D. 2010.How the plant growth-promoting bacterium Azospirillum promotes plant growth-A critical assessment. Adv. Agron. 108: 77–136
Bashan Y, Holguin G, de-Bashan LE. 2004. Azospirillumplant relationships: physiological, molecular, agricultural, and environmental advances (1997-2003). Can. J. Microbiol. 50: 521–577
Bashan Y, Levanony H, Klein E. 1986. Evidence for aweak active external adsorption of Azospirillumbrasilense Cd to wheat roots. J. Gen. Microbiol. 132: 3069–3073
Bhattacharjee RB, Singh A, Mukhopadhyay SN. 2008. Use of nitrogen-fixing bacteria as biofertiliser for non-legumes: prospects and challenges. Appl. Microbiol. Biotechnol. 80: 199–209
Bhattacharyya PN, Jha DK. 2012. Plant growth-promoting rhizobacteria (PGPR): emergence in agriculture. World J. Microbiol. Biotechnol. 28: 1327–1350.
Brusamarello-Santos LCC, Pacheco F, Aljanabi SMM, Monteiro RA, Cruz LM, Baura VA, Pedrosa FO, Souza EM, Wassem R. 2012. Differential gene expression of rice roots inoculated with the diazotroph Herbaspirillum seropedicae. Plant Soi. 356: 113–125
Cangahuala-Inocente GC, do Amaral FP, Faleirov AC, Huergo LF, Maisonnave Arisi AC. 2013. Identification of six differentially accumulated proteins of Zea mays seedlings (DKB240 variety) inoculated with Azospirillum brasilense strain FP2. Eur. J. Soil Biol. 58: 45–50
Cheng Z, McConkey BJ, Glick BR. 2010. Proteomic studies of plant-bacterial interactions. Soil Biol. Biochem. 42: 1673–1684
Cordeiro FA, Tadra-Sfeir MZ, Huergo LF, Pedrosa FdO, Monteiro RA, de Souza EM. 2013. Proteomic analysis of Herbaspirillum seropedicaecultivated in the presence of sugar cane extract. J. Proteome Res. 12: 1142–1150
Creus CM, Graziano M, Casanovas EM, Pereyra MA, Simontacchi M, Puntarulo S, Barassi CA, Lamattina L. 2005. Nitric oxide is involved in the Azospirillum brasilense-induced lateral root formation in tomato. Plant. 221: 297–303
Dillon S, Bateman A. 2004. The Hotdog fold: wrapping up a superfamily of thioesterases and dehydratases. BMC Bioinformatic. 5: 109–122
Dobbelaere S, Vanderleyden J, Okon Y. 2003. Plant growthpromoting effects of diazotrophs in the rhizosphere. Crit. Rev. Plant Sci. 22: 107–149
Egener T, Hurek T, Reinhold-Hurek B. 1999. Endophytic expression of nif genes of Azoarcus sp strain BH72 in rice roots. Mol. Plant-Microbe Interact. 12: 813–819
El Zemrany H, Cortet J, Lutz MP, Chabert A, Baudoin E, Haurat J, Maughan N, Felix D, Defago G, Bally R, Moenne-Loccoz Y. 2006. Field survival of the phytostimulator Azospirillum lipoferum CRT1 and functional impact on maize crop, biodegradation of crop residues, and soil faunal indicators in a context of decreasing nitrogen fertilisation. Soil Biol. Biochem. 38: 1712–1726
Fadel-Picheth C M, Souza E M, Rigo LU, Funayama S, Pedrosa FO. 1999. Regulation of the nifa gene of Azospirillum brasilense by ammonium and oxygen. FEMS Microbiology Letter. 179: 281–288
Faleiro AC, Pereira TP, Espindula E, Angonesi Brod FC, Maisonnave Arisi AC. 2013. Real time PCR detection targeting nifA gene of plant growth promoting bacteria Azospirillum brasilense strain FP2 in maize roots. Symbiosi. 61: 125–133
Fibach-Paldi S, Burdman S, Okon Y. 2012. Key physiological properties contributing to rhizosphere adaptation and plant growth promotion abilities of Azospirillum brasilense. FEMS Microbiol. Lett. 326: 99–108
Guerrero-Molina M, Winik B, Pedraza R. 2012. More than rhizosphere colonization of strawberry plants by Azospirillum brasilense. Appl. Soil Ecol.61: 205–212
Holguin G, Patten C, Glick B. 1999. Genetics and molecular biology of Azospirillum. Biol. Fert. Soil. 29: 10–23
Hungria M, Campo RJ, Souza EM, Pedrosa FO. 2010. Inoculation with selected strains of Azospirillum brasilense and A. lipoferum improves yields of maize and wheat in Brazil. Plant Soi. 331: 413–425
Ikeda A, Bassani L, Adamoski D, Stringari D, Cordeiro V, Glienke C, Steffens M, Hungria M, Galli-Terasawa L. 2013. Morphological and genetic characterization of endophytic bacteria isolated from roots of different maize genotypes. Microb. Ecol. 65: 154–160
Katsy EI, Prilipov AG. 2009. Mobile elements of an Azospirillum brasilense Sp245 85-MDa plasmid involved in replicon fusions. Plasmi. 62: 22–29
Klassen G, Pedrosa FO, Souza EM, Funayama S, Rigo LU. 1997. Effect of nitrogen compounds on nitrogenase activity in Herbaspirillum seropedicae SMR1. Can. J. Microbiol. 43: 887–891
Mercado-Blanco J, Prieto P. 2012. Bacterial endophytes and root hairs. Plant Soi. 361: 301–306
Molina-Favero C, Creus CM, Simontacchi M, Puntarulo S, Lamattina L. 2008. Aerobic nitric oxide production by Azospirillum brasilense Sp245 and its influence on root architecture in tomato. Mol. Plant-Microbe In. 21: 1001–1009
Neal A, Ahmad S, Gordon-Weeks R, Ton J. 2012. Benzoxazinoids in Root Exudates of Maize Attract Pseudomonas putida to the Rhizosphere. Plos One 7: e35498
Okon Y, Labandera-Gonzalez CA. 1994. Agronomic applications of Azospirillum-an evaluation of 20 years world wide field inoculation. Soil Biology & Biochemistry 26: 11601
Parales R, Harwood C. 2002. Bacterial chemotaxis to pollutants and plant-derived aromatic molecules. Curr. Opin. Microbiol. 5: 266–273
Pedraza RO, Motok J, Salazar SM, Ragout AL, Mentel MI, Tortora ML, Guerrero-Molina MF, Winik BC, Diaz-Ricci JC. 2010. Growth-promotion of strawberry plants inoculated with Azospirillum brasilense. World J.Microbiol. Biotechnol. 26: 265–272
Prinsi B, Negri AS, Pesaresi P, Cocucci M, Espen L. 2009. Evaluation of protein pattern changes in roots and leaves of Zea mays plants in response to nitrate availability by two-dimensional gel electrophoresis analysis. BMC Plant Biol. 9: 113–130
Rajendran C, Gerhardt ECM, Bjelic S, Gasperina A, Scarduelli M, Pedrosa FO, Chubatsu LS, Merrick M, Souza EM, Winkler FK, Huergo LF, Li X-D. 2011. Crystal structure of the GlnZ-DraG complex reveals a different form of P-II-target interaction. Proc. Natl.Acad. Sci. USA 108: 18972–18976
Roesch LFW, de Quadros PD, Camargo FAO, Triplett EW. 2007. Screening of diazotrophic bacteria Azopirillum spp. for nitrogen fixation and auxin production in multiple field sites in southern Brazil. World J.Microbiol. Biotechnol. 23: 1377–1383.
Roesch LFW, Olivares FL, Passaglia LMP, Selbach PA, de Sa ELS, de Camargo FAO. 2006. Characterization of diazotrophic bacteria associated with maize: effect of plant genotype, ontogeny and nitrogen-supply. World J. Microbiol. Biotechnol. 22: 967–974
Ruebelt MC, Leimgruber NK, Lipp M, Reynolds TL, Nemeth MA, Astwood JD, Engel KH, Jany KD. 2006. Application of two-dimensional gel electrophoresis to interrogate alterations in the proteome of genetically modified crops. 1. Assessing analytical validation. J. Agric. Food Chem. 54: 2154–2161
Saharan BS, Nehra V. 2011.Plant growth promoting rhizobacteria: A critical review. Life Sci. Med. Res. 2011: 1–30
Sujeeth N, Kini RK, Shailasree S, Wallaart E, Shetty SH, Hille J. 2012. Characterization of a hydroxyproline-rich glycoprotein in pearl millet and its differential expression in response to the downy mildew pathogen Sclerospora graminicola. Acta Physiol. Plan. 34: 779–791
Tameling W, Vossen J, Albrecht M, Lengauer T, Berden J, Haring M, Cornelissen B, Takken F. 2006. Mutations in the NB-ARC domain of I-2 that impair ATP hydrolysis cause autoactivation. Plant Physiol. 140: 1233–1245
Van Ooijen G, Mayr G, Kasiem M, Albrecht M, Cornelissen B, Takken F. 2008. Structure-function analysis of the NBARC domain of plant disease resistance proteins. J. Exper. Bot. 59: 1383–1397
Vargas L, Gurjao de Carvalho TL, Gomes Ferreira PC, Divan Baldani VL, Baldani JI, Hemerly AS. 2012. Early responses of rice (Oryza sativa L.) seedlings to inoculation with beneficial diazotrophic bacteria are dependent on plant and bacterial genotypes. Plant Soi. 356: 127–137
Verma JP, Yadav J, Tiwari KN, Lavakush, Singh V. 2010. Impact of plant growth promoting rhizobacteria on crop production. Intl. J. Agric. Res. 5: 954–983
Walker V, Couillerot O, Von Felten A, Bellvert F, Jansa J, Maurhofer M, Bally R, Moenne-Loccoz Y, Comte G. 2012. Variation of secondary metabolite levels in maize seedling roots induced by inoculation with Azospirillum, Pseudomonas, and Glomus consortium under field conditions. Plant Soi. 356: 151–163
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Faleiro, A.C., Neto, P.A.V., de Souza, T.V. et al. Microscopic and proteomic analysis of Zea mays roots (P30F53 variety) inoculated with Azospirillum brasilense strain FP2. J. Crop Sci. Biotechnol. 18, 63–71 (2015). https://doi.org/10.1007/s12892-014-0061-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12892-014-0061-x