Abstract
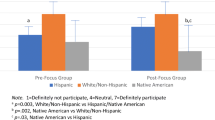
The National Children’s Study (NCS) Harris County, Texas Study Center participated in the NCS Provider Based Sampling (PBS) substudy of the NCS Vanguard Phase pilot. As part of the hospital-based birth cohort component of the PBS substudy, we conducted a secondary data analysis to evaluate the proportion of postpartum women who consented to future biospecimen collection alone and to both future collection and use of residual birth biospecimens. In phase 1, 32 postpartum women at one hospital were asked to consent only to maternal future biospecimen collection. In phase 2, 40 other postpartum women from the same hospital were asked for an additional consent to use residual clinical biospecimens from the birth event that otherwise would be discarded, including cord blood and maternal blood and urine. Among 103 eligible women, a total of 72 participated. They were 28.3 ± 5.9 years old on average; 58 % were Hispanic; 63 % consented in English, and 37 % in Spanish; 39 % had some college education; 42 % were married; 60 % had an annual family income <$30,000; and 51 % were employed. In phase 1, 59 % consented to future biospecimen collection, and in phase 2, 95 % consented to both future collection and use of at least one residual birth biospecimen, with a difference between phases of 36 % [95 % CI 17–54 %]. Demographic characteristics did not differ among those who did and did not consent. Postpartum women were significantly more likely to grant consent for use of future and residual hospital-obtained biospecimens than future biospecimen collection alone.
Similar content being viewed by others
References
Botkin JR, Rothwell E, Anderson R, Stark L, Goldenberg A, Lewis M et al (2012) Public attitudes regarding the use of residual newborn screening specimens for research. Pediatrics 129:231–238
Caenazzo L, Tozzo P, Pegoraro R (2012) Biobanking research on oncological residual material: a framework between the rights of the individual and the interest of society. BMC Med Ethics 14:17
Chan TW, Mackey S, Hegney DG (2012) Patients' experiences on donation of their residual biological samples and the impact of these experiences on the type of consent given for the future research use of the tissue: a systematic review. Int J Evid Based Health 10:9–26
Evans BJ, Meslin EM (2006) Encouraging translational research through harmonization of FDA and common rule informed consent requirements for research with banked specimens. J Leg Med 27:119–166
Fong M, Braun KL, Chang RM (2004) Native Hawaiian preferences for informed consent and disclosure of results from research using stored biological specimens. Pac Health Dialog 11:154–159
Gefenas E, Dranseika V, Serepkaite J, Cekanauskaite A, Caenazzo L, Gordijn B et al (2012) Turning residual human biological materials into research collections: playing with consent. J Med Ethics 38:351–355
Hakimian R, Korn D (2004) Ownership and use of tissue specimens for research. JAMA 292:2500–2505
Hiratsuka VY, Brown JK, Hoeft TJ, Dillard DA (2012) Alaska native people's perceptions, understandings, and expectations for research involving biological specimens. Int J Circumpolar Health 71:18642
Hoeyer K, Olofsson BO, Mjorndal T, Lynoe N (2004) Informed consent and biobanks: a population-based study of attitudes towards tissue donation for genetic research. Scand J Public Health 32:224–229
Hoeyer K, Olofsson BO, Mjorndal T, Lynoe N (2005) The ethics of research using biobanks: reason to question the importance attributed to informed consent. Arch Intern Med 165:97–100
Kaufman D, Bollinger J, Dvoskin R, Scott J (2012) Preferences for opt-in and opt-out enrollment and consent models in biobank research: a national survey of Veterans Administration patients. Genet Med 14:787–794
Landrigan PJ, Trasande L, Thorpe LE, Gwynn C, Lioy PJ, D'Alton ME, Lipkind HS, Swanson J, Wadhwa PD, Clark EB, Rauth VA, Perera FP, Susser E (2006) The National Children's Study: a 21-year prospective study of 100,000 American children. Pediatrics 118:2173–2186
Lewis C, Clotworthy M, Hilton S, Magee C, Robertson MJ, Stubbins LJ, et al. (2013a). Public views on the donation and use of human biological samples in biomedical research: a mixed methods study. BMJ Open 3. doi:10.1136/bmjopen-2013-003056
Lewis C, Clotworthy M, Hilton S, Magee C, Robertson MJ, Stubbins LJ, et al. (2013b). Consent for the use of human biological samples for biomedical research: a mixed methods study exploring the UK public's preferences. BMJ Open 3. doi:10.1136/bmjopen-2013003022
Marsolo K, Corsmo J, Barnes MG, Pollick C, Chalfin J, Nix J et al (2012) Challenges in creating an opt-in biobank with a registrar-based consent process and a commercial EHR. J Am Med Inform Assoc 19:1115–1118
U.S. Department of Health and Human Services (2015) Proposed rules. Fed Regist 80:53993–54061
Vermeulen E, Schmidt MK, Aaronson NK, Kuenen M, Baas-Vrancken Peeters MJ, van der Poel H et al (2009) A trial of consent procedures for future research with clinically derived biological samples. Br J Cancer 101:1505–1512
Acknowledgments
We would like to acknowledge the work of Vivian Driskell, Katherine Addy, Angelique D. Clarke, and Dinah J. Pena in collecting and entering data for this study. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institutes of Health or the U.S. Department of Health and Human Services.
The study was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development contract HHSN275200800020C.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Kozinetz, C.A., Royse, K., Graham, S.C. et al. Consenting postpartum women for use of routinely collected biospecimens and/or future biospecimen collection. J Community Genet 7, 153–158 (2016). https://doi.org/10.1007/s12687-016-0261-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12687-016-0261-9