Abstract
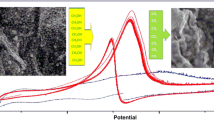
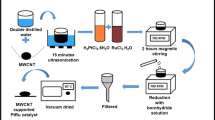
The poisoning of pure platinum electrodes has propelled the improvement of platinum-based alloy catalysts. In this work, we report the application of exfoliated graphite electrode as an anode platform for binary platinum-based electrocatalysts for methanol oxidation in alkaline media. Platinum-based binary electrocatalysts comprising of Pt-Au and Pt-Pd nanoparticles on functionalized multi-walled carbon nanotubes (MWCNTs) support were synthesized via polyol process. The nanocomposite electrocatalysts (Pt-Au/fMWCNTs and Pt-Pd/fMWCNTs) were characterized by transmission electron microscopy (TEM) coupled with energy-dispersive X-ray spectroscopy (EDS) and X-ray diffractometer (XRD). The electrochemical activity of the Pt-Pd/fMWCNTs and the Pt-Au/fMWCNTs electrocatalysts was assessed utilizing cyclic voltammetry (CV), chronoamperometry (CA), and electrochemical impedance spectroscopy (EIS) in the presence of methanol. The nanoparticles were in the size regime of 3 to 5 nm and the XRD affirmed successful functionalization of the MWCNT and its impregnation with the binary Pt nanoparticles. The electrooxidation of methanol in alkaline conditions was seen with a high current density of about 26.1 mA cm−2 on Pt-Au/fMWCNTs and 13.52 mA cm−2 on Pt-Pd/fMWCNTs electrocatalysts deposited on exfoliated graphite working electrodes. The exfoliated graphite platform is a promising electrode for fuel cell application.

.







Similar content being viewed by others
References
J.A. Rodriguez, P. Liu, D.J. Stacchiola, S.D. Senanayake, M.G. White, J.G. Chen, Hydrogenation of CO2to Methanol: Importance of Metal–Oxide and Metal–Carbide Interfaces in the Activation of CO2. ACS Catal. 5(11), 6696–6706 (2015)
A.E. Vilian, S.K. Hwang, C.H. Kwak, S.Y. Oh, C.Y. Kim, G.W. Lee, J.B. Lee, Y.S. Huh, Y.K. Han, Pt-Au bimetallic nanoparticles decorated on reduced graphene oxide as an excellent electrocatalysts for methanol oxidation. Synth. Met. 219, 52–59 (2016)
P. Trop, B. Anicic, D. Goricanec, Production of methanol from a mixture of torrefied biomass and coal. Energy 77, 125–132 (2014)
S.K. Kamarudin, N. Shamsul, J.A. Ghani, S. Chia, H. Liew, A. Samsudin, Production of methanol from biomass waste via pyrolysis. Bioresour. Technol. 129, 463–468 (2013)
Y. Lu, S. Du, R. Steinberger-Wilckens, One-dimensional nanostructured electrocatalysts for polymer electrolyte membrane fuel cells—A review. Appl. Catal. B Environ. 199, 292–314 (2016)
Z.Z. Yijie Lei, T. Li, J. Gu, T. Yu, J. Liu, Electroanalysis 24, 699–706 (2012)
M. Inaba, H. Daimon, Development of highly active and durable platinum core-shell catalysts for polymer electrolyte fuel cells. J. Jpn. Petrol. Inst. 58(2), 55–63 (2015)
E. Antolini, Palladium in fuel cell catalysis. Energy Environ. Sci. 2(9), 915–931 (2009)
E. Papaioannou, A. Siokou, C. Comninellis, A. Katsaounis, Pt–Ir binary electrodes for direct oxidation of methanol in low-temperature fuel cells (DMFCs). Electrocatalysis 4(4), 375–381 (2013)
J. Masa, K. Ozoemena, W. Schuhmann, J.H. Zagal, Oxygen reduction reaction usingN4-metallomacrocyclic catalysts: fundamentals on rational catalyst design. J. Porphyr. Phthalocyanines 16(07n08), 761–784 (2012)
N. Soin, S.S. Roy, T.H. Lim, J.A. McLaughlin, Microstructural and electrochemical properties of vertically aligned few layered graphene (FLG) nanoflakes and their application in methanol oxidation. Mater. Chem. Phys. 129(3), 1051–1057 (2011)
Y.H. Qin, H.H. Yang, X.S. Zhang, P. Li, C.A. Ma, Effect of carbon nanofibers microstructure on electrocatalytic activities of Pd electrocatalysts for ethanol oxidation in alkaline medium. Int. J. Hydrog. Energy 35(15), 7667–7674 (2010)
I. Danaee, M. Jafarian, F. Forouzandeh, F. Gobal, M. Mahjani, Electrocatalytic oxidation of methanol on Ni and NiCu alloy modified glassy carbon electrode. Int. J. Hydrog. Energy 33(16), 4367–4376 (2008)
Y. Li, L. Tang, J. Li, Preparation and electrochemical performance for methanol oxidation of pt/graphene nanocomposites. Electrochem. Commun. 11(4), 846–849 (2009)
Y. Nishijima, K. Ueno, Y. Yokota, K. Murakoshi, H. Misawa, Plasmon-assisted photocurrent generation from visible to near-infrared wavelength using a Au-nanorods/TiO2 electrode. J. Phys. Chem. Lett. 1(13), 2031–2036 (2010)
P.H. Chen, D. Chung, Dynamic mechanical behavior of flexible graphite made from exfoliated graphite. Carbon 50(1), 283–289 (2012)
W. Cai, T. Lai, H. Du, J. Ye, Electrochemical determination of ascorbic acid, dopamine and uric acid based on an exfoliated graphite paper electrode: a high performance flexible sensor. Sensors Actuators B Chem. 193, 492–500 (2014)
T. Ndlovu, O. Arotiba, S. Sampath, R. Krause, B. Mamba, Int. J. Electrochem. Sci. 7, 9441–9453 (2012)
C. Frysz, D.L. Chung, Electrochemical behavior of flexible graphite. Carbon 35(6), 858–860 (1997)
T. Ndlovu, B.B. Mamba, S. Sampath, R.W. Krause, O.A. Arotiba, Voltammetric detection of arsenic on a bismuth modified exfoliated graphite electrode. Electrochim. Acta 128, 48–53 (2014)
T. Ndlovu, O.A. Arotiba, S. Sampath, R.W. Krause, B.B. Mamba, An exfoliated graphite-based bisphenol A electrochemical sensor. Sensors 12(9), 11601–11611 (2012)
M. Zhang, Z. Yan, J. Xie, Core/shell Ni@Pd nanoparticles supported on MWCNTs at improved electrocatalytic performance for alcohol oxidation in alkaline media. Electrochim. Acta 77, 237–243 (2012)
N. Hidayati, K. Scott, Bull. Chem. React. Eng. Catal. 11, 10–20 (2016)
T. Ndlovu, O.A. Arotiba, S. Sampath, R.W. Krause, B.B. Mamba, Electrochemical detection and removal of lead in water using poly(propylene imine) modified re-compressed exfoliated graphite electrodes. J. Appl. Electrochem. 41(12), 1389–1396 (2011)
M. Bonnissel, L. Luo, D. Tondeur, Compacted exfoliated natural graphite as heat conduction medium. Carbon 39(14), 2151–2161 (2001)
C.L. Ngo, Q.T. Le, T.T. Ngo, D.N. Nguyen, M.T. Vu, Adv. Nat. Sci. Nanosci. Nanotechnol. 4, 035017–035028 (2013)
K. Jiang, W.B. Cai, Carbon supported Pd-Pt-Cu nanocatalysts for formic acid electrooxidation: Synthetic screening and componential functions. Appl. Catal. B Environ. 147, 185–192 (2014)
I.A. Rutkowska, A. Wadas, P.J. Kulesza, Mixed layered WO 3 /ZrO 2 films (with and without rhodium) as active supports for PtRu nanoparticles: enhancement of oxidation of ethanol. Electrochim. Acta 210, 575–587 (2016)
S. Jingyu, H. Jianshu, C. Yanxia, Z. Xiaogang, Int. J. Electrochem. Sci. 2, 64–71 (2007)
S.S. Gwebu, P.N. Nomngongo, P.N. Mashazi, T. Nyokong, N.W. Maxakato, Int. J. Electrochem. Sci. 12, 6365–6378 (2017)
K. Honda, M. Yoshimura, T.N. Rao, D. Tryk, A. Fujishima, K. Yasui, Y. Sakamoto, K. Nishio, H. Masuda, Electrochemical properties of Pt-modified nano-honeycomb diamond electrodes. J. Electroanal. Chem. 514(1-2), 35–50 (2001)
Y. Wang, Z.M. Sheng, H. Yang, S.P. Jiang, C.M. Li, Electrocatalysis of carbon black- or activated carbon nanotubes-supported Pd–Ag towards methanol oxidation in alkaline media. Int. J. Hydrog. Energy 35(19), 10087–10093 (2010)
Y. Xu, Y. Dong, J. Shi, M. Xu, Z. Zhang, X. Yang, Au@Pt core-shell nanoparticles supported on multiwalled carbon nanotubes for methanol oxidation. Catal. Commun. 13(1), 54–58 (2011)
Acknowledgements
The authors also thank Professor Ndungu and Dr. Modibedi for their scientific contributions and also to Mr. Thabo Matthews who looked at some of the research work and did corrections to manuscript especially interpretation of data and graphics.
Funding
The authors are grateful to the University of Johannesburg, Faculty of Science, University Research Council, and Centre for Nanomaterials Science Research and National Research Foundation (TTK-15071-0125-019) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mpeta, L.S., Gwebu, S.S., Arotiba, O.A. et al. Methanol Oxidation in Alkaline Media with Pt-Au/fMWCNTs and Pt-Pd/fMWCNTs Electrocatalysts on an Exfoliated Graphite Electrode. Electrocatalysis 10, 672–679 (2019). https://doi.org/10.1007/s12678-019-00555-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12678-019-00555-0