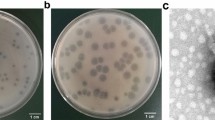
Abstract
Serratia marcescens is a Gram-negative bacterium from the Enterobacteriaceae family. As an opportunistic human pathogen and a causative agent of a range of nosocomial infections, S. marcescens represents a growing public health problem. Little is known about S. marcescens pathogenicity factors, in part due to the lack of well-developed functional genomic tools. Bacteriophage transduction is one of the main mechanisms of horizontal transfer of DNA in bacteria. S. marcescens bacteriophages previously used in transduction experiments are strain-specific, and no comprehensive studies have previously been done to evaluate the efficiency of transduction across different phages and host strains. In this study, we directly compared the ability of bacteriophages ΦOT8 and ΦIF3 to infect two S. marcescens strains, SM6 and SR41-8000. We successfully used bacteriophage ΦOT8 to transfer a mutation in tolC locus between two S. marcescens strains.

Similar content being viewed by others
References
Mahlen, S. D. (2011). Serratia infections: from military experiments to current practice. Clinical Microbiology Reviews, 24(4), 755–791. doi:10.1128/CMR.00017-11.
Hines, D. A., Saurugger, P. N., Ihler, G. M., Benedik, M. J. (1988). Genetic analysis of extracellular proteins of Serratia marcescens. Journal of Bacteriology, 170(9), 4141–4146.
Matsumoto, H., Tazaki, T., Hosogaya, S. (1973). A generalized transducing phage of Serratia marcescens. Japanese Journal of Microbiology, 17(6), 473–479.
Petty, N. K., Foulds, I. J., Pradel, E., Ewbank, J. J., Salmond, G. P. (2006). A generalized transducing phage (phiIF3) for the genomically sequenced Serratia marcescens strain Db11: a tool for functional genomics of an opportunistic human pathogen. Microbiology, 152(Pt 6), 1701–1708. doi:10.1099/mic.0.28712-0.
Regue, M., Fabregat, C., Vinas, M. (1991). A generalized transducing bacteriophage for Serratia marcescens. Research in Microbiology, 142(1), 23–27.
Evans, T. J., Crow, M. A., Williamson, N. R., Orme, W., Thomson, N. R., Komitopoulou, E., et al. (2010). Characterization of a broad-host-range flagellum-dependent phage that mediates high-efficiency generalized transduction in, and between, Serratia and Pantoea. Microbiology, 156(Pt 1), 240–247. doi:10.1099/mic.0.032797-0.
Iguchi, A., Nagaya, Y., Pradel, E., Ooka, T., Ogura, Y., Katsura, K., et al. (2014). Genome evolution and plasticity of Serratia marcescens, an important multidrug-resistant nosocomial pathogen. Genome Biology and Evolution, 6(8), 2096–2110. doi:10.1093/gbe/evu160.
Takagi, T., & Kisumi, M. (1985). Isolation of a versatile Serratia marcescens mutant as a host and molecular cloning of the aspartase gene. Journal of Bacteriology, 161(1), 1–6.
Uzzau, S., Figueroa-Bossi, N., Rubino, S., Bossi, L. (2001). Epitope tagging of chromosomal genes in Salmonella. Proceedings of the National Academy of Sciences of the United States of America, 98(26), 15264–15269. doi:10.1073/pnas.261348198.
Bogomolnaya, L. M., Santiviago, C. A., Yang, H. J., Baumler, A. J., Andrews-Polymenis, H. L. (2008). ‘Form variation’ of the O12 antigen is critical for persistence of Salmonella Typhimurium in the murine intestine. Molecular Microbiology, 70(5), 1105–1119. doi:10.1111/j.1365-2958.2008.06461.x.
Acknowledgments
This work was supported by the Russian Science Foundation project 16-14-10200 (L.K., T.S., and L.B.) and performed in accordance with the Russian Government Program of Competitive Growth of Kazan Federal University.
Author information
Authors and Affiliations
Corresponding author
Additional information
Tatiana V. Shirshikova and Olga V. Morozova contributed equally to this work.
Rights and permissions
About this article
Cite this article
Shirshikova, T.V., Morozova, O.V., Kamaletdinova, L.K. et al. Generalized Bacteriophage Transduction in Serratia marcescens . BioNanoSci. 6, 487–489 (2016). https://doi.org/10.1007/s12668-016-0268-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12668-016-0268-z