Abstract
Purpose
To test the antimicrobial properties of the extracts of stems and leaves of Ginja cherry plant. Both stems and leaves are waste in the production of the cherry liquor and they could be valorised by extracting valuable compounds, making the process more environmentally sustainable.
Methods
The ethanol extracts from both stems and leaves were analysed by LC-ESI/MS to determine the phenolic composition. They were tested against Gram positive and Gram negative bacteria (Bacillus subtilis, Staphylococcus aureus MSSA, Staphylococcus aureus MRSA, Pseudomonas sp., Pseudomonas aeruginosa, Flavobacterium sp., Escherichia coli, Salmonella), using the disk diffusion technique and the broth dilution technique.
Results
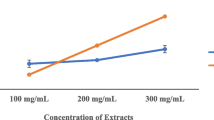
The extracts showed good antibacterial properties towards Gram positive and Gram negative bacteria. The values of the Minimum Inhibitory Concentration (MIC) were lower for Gram positive bacteria (10–15 mg/ml) than for Gram negative ones (10–100 mg/ml). The values of Minimum Bactericidal Concentration (MBC) were between 2 and 4 times higher than the MICs.
Conclusions
The waste from Ginja cherry plants can be successfully employed to extract valuable compounds such as polyphenols, with antibacterial properties.



Similar content being viewed by others
References
European Commission: Study on the feasibility of the establishment of a Waste Implementation Agency. http://ec.europa.eu/environment/waste/index.htm (2010). Accessed 24 Feb 2010
European Commission: Directive 2008/98/EC on waste (Waste Framework Directive). http://ec.europa.eu/environment/waste/framework/index.htm (2010). Accessed 24 Feb 2010
Lule, S.U., Xia, W.: Food phenolics, pros and cons: a review. Food Rev. Int. 21, 367–388 (2005)
Eastwood, M.A.: Interaction of dietary antioxidants in vivo: how fruit and vegetables prevent disease. QJM 92, 527–530 (1999)
Middleton, E.: Effect of plant flavonoids on immune and inflammatory cell function. Adv. Exp. Med. Biol. 439, 175–182 (1998)
Taguri, T., Tanaka, T., Kound, I.: Antibacterial spectrum of plants polyphenols and extracts depending upon hydroxyphenyl structure. Biol. Pharm. Bull. 29(11), 2226–2235 (2006)
Moure, A., Cruz, M.C., Franco, D., Domingues, J.M., Sineiro, J., Dominguez, E., Nuñez, M.J., Parajó, J.C.: Natural antioxidant from residual sources. Food Chem. 72, 145–171 (2001)
Lesage-Meessen, L., Navarro, D., Maunier, S., Sigolloit, J.C., Lorquin, J., Delattre, M., Simon, J.L., Ashter, M., Labat, M.: Simple phenolic content in olive oil residues as a function of the extraction system. Food Chem. 75, 501–507 (2001)
Obied, H.K., Bedgood, D.R. Jr, Prenzler, P.D., Robards, K.: Bioscreening of Australian olive mill waste extracts: biophenol content, antioxidant, antimicrobial and molluscicidial activities. Food Chem. Toxicol. 45, 1238–1248 (2007)
Sudjana, A.N., D’Orazio, C., Ryan, V., Rasool, N., Ng, J., Islam, N., Riley, T.V., Hammer, K.A.: Antimicrobial activity of commercial Olea europea (olive) leaf extract. Int. J. Antimicrob. Agents 33, 461–463 (2009)
Peschel, W., Sanchez-Rabaneda, F., Diekmann, W., Plescher, A., Gartzia, I., Jimenez, D., Lamuela-Raventos, R., Buxaderas, S., Codina, C.: An industrial approach in the search of natural antioxidants from vegetable and fruit wastes. Food Chem. 97(1), 137–150 (2006)
Wijngaard, H.H., Rossle, C., Brunton, N.: A survey of Irish fruit and vegetable waste and by-products as a source of polyphenols antioxidants. Food Chem. 116, 202–207 (2009)
Pereira, J.A., Oliveira, I., Sousa, A., Valenta, P., Andrade, P.B., Ferreira, I.C.F.R., Ferreres, F., Bento, A., Seabra, R., Estevinho, L.: Walnut (Juglans regia L.) leaves: phenolic compounds, antibacterial activity and antioxidant potential of different cultivars. Food Chem. Toxicol. 45, 2287–2295 (2007)
Kumar, P.S., Kumar, A., Sivakumar, R., Kaushik, C.: Experimentation on solvent extracts from natural waste. J. Mater. Sci. 44, 5894–5899 (2009)
Demiray S., Pintado M.M., Castro P.M.L.: High value added compounds with antioxidant activities from Ginja cherries, stems and leaves. EFFOST conference, Budapest, 11–13 Nov 2009
Bauer, A.W., Kirby, W.M., Sherries, J.C., Turck, M.: Antibiotic susceptibility testing by a standardised single method. Am. J. Clin. Pathol. 45, 493–496 (1966)
Moreno, S., Scheyer, T., Romano, C.S., Vojnov, A.A.: Antioxidant and antimicrobial activity of rosemary extracts linked to their polyphenolic composition. Free Radic. Res. 40(2), 223–231 (2006)
Lao, R.C., Shu, Y.Y., Holmes, J., Chiu, C.: Environmental sample cleaning and extraction procedures by microwave-assisted process (MAP) technology. Microchem. J. 53(1), 99–108 (1996)
Herald, P.J., Davidson, P.M.: Antibacterial activity of selected hydrocinnamic acid. J. Food Sci. 48(4), 1378–1379 (2006)
Liu, K., Tsao, S., Yin, M.: In vitro antibacterial activity of roselle calyx and protocatechuic acid. Phytother. Res. 19(11), 942–945 (2006)
Denny, B.J., West, P.W., Mathew, T.C.: Antagonistic interactions between the flavonoids hesperetin and naringenin and beta-lactam antibiotics against Staphylococcus aureus. Br. J. Biomed. Sci. 65(3), 145–147 (2008)
Kajiya, K., Hojo, H., Suzuki, M., Nanjo, F., Kumazawa, S., Nakayama, T.: Relationship between antibacterial activity of (+)-catechin derivatives and their interaction with a model membrane. J. Agric. Food Chem. 52, 1514–1519 (2004)
Nazaruk, J., Czechowska, S.K., Markiewicz, R., Borawska, M.H.: Polyphenolic compounds and in vitro antimicrobial and antioxidant activity of aqueous extracts from leaves of some Cirsium species. Nat. Prod. Res. 22(18), 1583–1588 (2008)
Franco, A.R., Calheiros, C.S.C., Pacheco, C.C., De Marco, P., Manaia, C.M., Castro, P.M.L.: Isolation and characterisation of polymeric galloyl-ester-degrading bacteria from a tannery discharge place. Microb. Ecol. 50, 550–556 (2005)
Chung, K.T., Lu, Z., Chou, M.W.: Mechanism of inhibition of tannin acid and related compounds on the growth of intestinal bacteria. Food Chem. Toxicol. 36(12), 104–1060 (1998)
Munoz, M., Guevara, L., Palop, A., Tabera, J., Fernandez, P.S.: Determination of the effect of plant essential oils obtained by supercritical fluid extraction on the growth and viability of Listeria monocytogenes in broth and food systems using flow cytometry. LWT Food Sci. Technol. 42, 220–227 (2009)
Karanika, M.S., Komaitis, M., Aggelis, G.: Effect of aqueous extracts of some plants of Lamiaceae family on the growth of Yarrowia lipolytica. Int. J. Food Microbiol. 64, 175–181 (2001)
Furiga, A., Lonvaud-Funel, A., Dorigna, G., Badet, C.: In vitro anti-bacterial and anti-adherence effects of natural polyphenolic compounds on oral bacteria. J. Appl. Microbiol. 105, 1470–1476 (2008)
Sasaki, H., Matsumoto, M., Tanaka, T., Maeda, M., Nakai, M., Hamada, S., Ooshima, T.: Antibacterial activity of polyphenol components in oolong tea extracts against Streptococcus mutans. Caries Res. 38(1), 2–8 (2004)
Romano, C.S., Anadi, K., Repetto, V., Vojnov, A.A., Moreno, S.: Synergistic antioxidant and antibacterial activity of rosemary plus butylated derivatives. Food Chem. 115(2), 456–461 (2009)
Acknowledgement
This work was performed within the InSolEx network (Innovative Solutions for Extracting High Value Natural Compounds) funded by the European Union, contract MRT-CT-2006-036053.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Piccirillo, C., Demiray, S., Franco, A.R. et al. High Added-Value Compounds with Antibacterial Properties from Ginja Cherries By-products. Waste Biomass Valor 1, 209–217 (2010). https://doi.org/10.1007/s12649-010-9019-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-010-9019-5