Abstract
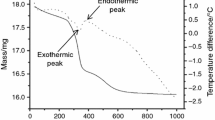
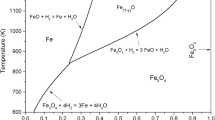
The kinetics of removal of loss on ignition (LOI) by thermal decomposition of hydrated minerals present in natural iron ores (i.e., kaolinite, gibbsite, and goethite) was investigated in a laboratory-scale vertical fluidized bed reactor (FBR) using isothermal methods of kinetic analysis. Experiments in the FBR in batch processes were carried out at different temperatures (300 to 1200°C) and residence time (1 to 30 min) for four different iron ore samples with various LOIs (2.34wt% to 9.83wt%). The operating velocity was maintained in the range from 1.2 to 1.4 times the minimum fluidization velocity (U mf). We observed that, below a certain critical temperature, the FBR did not effectively reduce the LOI to a desired level even with increased residence time. The results of this study indicate that the LOI level could be reduced by 90% within 1 min of residence time at 1100°C. The kinetics for low-LOI samples (<6wt%) indicates two different reaction mechanisms in two temperature regimes. At lower temperatures (300 to 700°C), the kinetics is characterized by a lower activation energy (diffusion-controlled physical moisture removal), followed by a higher activation energy (chemically controlled removal of LOI). In the case of high-LOI samples, three different kinetics mechanisms prevail at different temperature regimes. At temperature up to 450°C, diffusion kinetics prevails (removal of physical moisture); at temperature from 450 to 650°C, chemical kinetics dominates during removal of matrix moisture. At temperatures greater than 650°C, nucleation and growth begins to influence the rate of removal of LOI.
Similar content being viewed by others
References
S.K. Das, B. Das, R. Saktivel, and B.K. Mishra, Mineralogy, microstructure, and chemical compositions of goethites in some iron ore deposits of Orissa, India, Miner. Process. Extr. Metall. Rev., 31(2010), No. 2, p. 97.
B.K. Mishra, B. Das, S. Prakash, S.K. Das, S. K. Biswal, and P.S.R. Reddy, Issues relating characterization and beneficiation of low grade iron ore, Steelworld, November 2007, p. 34.
S.K. Biswal, Utilization of low grade iron ore fines, slimes and tailings by physical beneficiation to minimize the waste generation, J. Sustainable Planet, 1(2010), p. 46.
U.C. Chung, I.O. Lee, H.G. Kim, V. Sahajwalla, and W.B. Chung, Degradation characteristics of iron ore fines of a wide size distribution in fluidized-bed reduction, ISJI Int., 38(1998), No. 9, p. 943.
N.S. Sundarmurti and V. Rao, Thermal conductivity and diffusivity of iron ore pellet having low porosity, ISJI Int., 42(2002), No. 7, p. 800.
L.D. Santos and P.R.G. Brandao, Morphological varieties of goethite in iron ore from Minas Gerais, Brazil, Miner. Eng., 16(2003), No. 11, p. 1285.
R.K. Singh, and G.K. Roy, Prediction of minimum bubbling velocity, fluidization index and range of particulate fluidization for gas–solid fluidization in cylindrical and non-cylindrical beds, Powder Technol., 159(2005), No. 3, p. 168.
S. Gialanella, F. Girardi, G. Ischia, I. Lonardelli, M. Mattarelli, and M. Montagna, On the goethite to hematite phase transformation, J. Therm. Anal. Calorim., (102)2010, No. 3, p. 867.
M. Inoue, K. Kitamura, H. Tanino, H. Nakayama, and T. Inui, Alcohothermal treatments of gibbsite: mechanism for the transformation of boehmite, Clays Clay Miner., 37(1989), p. 71. Clays and Clay Minerals
C.H. Bamford and C.F.H. Tipper, eds., Comprehensive Chemical Kinetics, Elsevier Scientific Publishing Corporation, Amsterdam, New York, 1980.
I. Halikia, L. Zoumpoulakis, E. Christodoulou, and D. Prattis, Kinetic study of the thermal decomposition of calcium carbonate by isothermal method of analysis, Eur. J. Miner. Process. Environ. Prot., 1(2001), No. 2, p. 1303.
I. Halikia, P. Neou-Syngouna, and D. Kolitsa, Isothermal kinetic analysis of the thermal decomposition of magnesium hydroxide using thermo gravimetric data, Thermochim. Acta, 320(1998), No. 1-2, p. 75.
C.J. Goss, The kinetics and reaction mechanism of the goethite to hematite transformation, Mineral. Mag., 51(1987), p. 437.
E. Wolska, Relation between the existence of hydroxyl ions in the anionic substance of hematite and its infrared and X-ray characteristics, Solid State Ionics, 28-30(1988), p. 1349.
Ö. Özdemir and D.J. Dunlop, Intermediate magnetite formation during dehydration of goethite, Earth Planet. Sci. Lett., 177(2000), No. 1-2, p. 59.
K. Przepiera and A. Przepiera, Kinetic of thermal transformations of precipitated magnetite and goethite, J. Therm. Anal. Calorim., 65(2001), No. 2, p. 497.
J. Lima-De-Faria, Dehydration of goethite and diaspore, Z. Kristallogr., 119(1963), No. 1-6, p. 176.
P.S.R. Prasad, K.S. Prasad, V.K. Chaitanya, E.V.S.S.K. Babu, B. Sreedhar, and S.R. Murthy, In situ FTIR study on the dehydration of natural goethite, J. Asian Earth Sci., 27(2006), No. 4, p. 503
Acknowledgement
The authors are thankful to the Ministry of Steel, New Delhi for sponsoring the research work undertaken in the present study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Beuria, P.C., Biswal, S.K., Mishra, B.K. et al. Kinetics of thermal decomposition of hydrated minerals associated with hematite ore in a fluidized bed reactor. Int J Miner Metall Mater 24, 229–239 (2017). https://doi.org/10.1007/s12613-017-1400-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12613-017-1400-y