Abstract
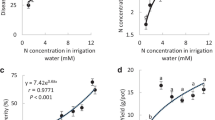
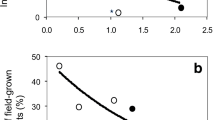
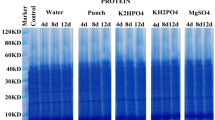
Plant nutrition affects plant diseases and epidemics development. The effects of supplemental nitrogen, potassium and calcium on white mold susceptibility in sweet basil were tested in pots and under commercial conditions. An increased concentration of N in the irrigation solution increased the N content of shoots, which led to higher susceptibility to white mold on cut shoots in a linear manner. Increased levels of K in the irrigation solution and in the sweet basil tissue resulted in an exponential decrease in the severity of white mold on cut shoots. Likewise, foliar application of K also significantly decreased white mold susceptibility. Lower K fertigation under commercial-like conditions significantly increased susceptibility to S. sclerotiorum infection. Calcium decreased disease susceptibility, but there was no additive effect when Ca was added to the K treatment. Combining Ca and K fertigation with foliar-applied KCl and fungicide (boscalid + pyraclostrobin) provided synergistic lower disease on cut shoots. The K spray was not as effective as the fungicide for suppressing disease. In conclusion, proper K fertilization and the application of Ca can significantly reduce the susceptibility of sweet basil shoots to S. sclerotiorum and may be integrated into management programs for proper disease control.




Similar content being viewed by others
References
Abawi, G. S., & Grogan, R. G. (1979). Epidemiology of diseases caused by Sclerotinia species. Phytopathology, 69, 899–904.
Abia, J. A., & Smith, B. N. (1980). Mineral nutritional status of pumpkin and infection by Sclerotinia sclerotiorum. Plant Physiology, 65, 106 (Abstr.).
Arfaoui, A., El Hadrami, A., Adam, L. R., & Daayf, F. (2016). Pre-treatment with calcium enhanced defense-related genes' expression in the soybean's isoflavone pathway in response to Sclerotinia sclerotiorum. Physiological and Molecular Plant Pathology, 93, 12–21.
Arfaoui, A., El Hadrami, A., & Daayf, F. (2018). Pre-treatment of soybean plants with calcium stimulates ROS responses and mitigates infection by Sclerotinia sclerotiorum. Plant Physiology and Biochemistry, 122, 121–128.
Bangerth, F. (1979). Calcium related physiological disorders of plant. Annual Review of Phytopathology, 17, 97–122.
Bar-Tal, A., Baas, R., Ganmore-Neumann, R., Dik, A., Marissen, N., Silber, A., Davidov, S., Hazan, A., Kirshner, B., & Elad, Y. (2001). Rose flower production and quality as affected by Ca concentration in the petal. Agronomie, 21, 393–402.
Ben Yephet, Y. (1988). Control of sclerotia and apothecia of Sclerotinia sclerotiorum by metham sodium, methyl bromide and soil solarization. Crop Protection, 7, 25–27.
Biddle, A. J. (2001). Botrytis gray mold. In J. M. Kraft & F. L. Pfleger (Eds.), Compendium of pea diseases and pests (2nd ed., pp. 31–32). St. Paul: American Phytopathological Society Press.
Datnoff, L. E., Elmer, W. E., & Huber, D. W. (Eds.). (2007). Mineral nutrition and plant disease. St. Paul: The American Phytopathological Society.
Dudai, N., Chaimovitsh, D., Reuveni, R., Ravid, U., Larkov, O., & Putievsky, E. (2002). Breeding of sweet basil (Ocimum basilicum) resistant to Fusarium oxyxsporum f. sp. basilicum. Journal of Herbs Spices and Medicinal. Plants, 9, 45–51.
Elad, Y., & Evensen, K. (1995). Physiological aspects of resistance to Botrytis cinerea. Phytopathology, 85, 637–643.
Elad, Y., Israeli, L., Fogel, M., Rav David, D., Kenigsbuch, D., Chalupowicz, D., Maurer, D., Lichter, A., Silverman, D., Biton, S., Yitzhak, S., Harari, D., Maduel, A., Pivonia, S., & Adler, U. (2014). Conditions influencing the development of sweet basil grey mould and cultural measures for disease management. Crop Protection, 64, 67–77.
Elad, Y., Fogel, M., Silverman, D., Biton, S., Yitzhak, S., Harari, D., & Adler, U. (2015). White mould of sweet basil: Conditions influencing its development in greenhouses and cultural measures for disease management. Plant Pathology, 64, 951–960.
Engelhard, W. (1989). Soilborne plant pathogens: Management of diseases with macro- and microelements. St. Paul: American Phytopathological Society.
Erel, R., Dag, A., Ben-Gal, A., Schwartz, A., & Yermiyahu, U. (2008). Flowering and fruit-set of young olive (Olea europea L. cv. Barnea) trees in response to nitrogen, phosphorus and potassium. Journal of the American Society of Horticultural Sciences, 133, 639–647.
Garibaldi, A., Gullino, M. L., & Minuto, G. (1997). Diseases of basil and their management. Plant Disease, 81, 124–132.
Hartill, W. F. T. (1980). Aerobiology of Sclerotinia sclerotiorum and Botrytis cinerea spores in New Zealand tobacco crops. New Zealand Agricultural Research, 23, 259–262.
Hobbs, E. L., & Waters, W. E. (1964). Influence of nitrogen and potassium on susceptibility of Chrysanthemum morifolium to Botrytis cinerea. Phytopathology, 54, 674–676.
Hochmuth, R. C., Davis, L. L. L., Laughlin, W. L., Simonne, E. H., & Sprenkel, R. K. (2004). Developing a production system for growing organic herbs using soilless culture in a greenhouse. University of Florida Cooperation Extension Service, 22, 37–42.
Holcomb, G. E., & Reed, M. J. (1994). Stem rot of basil caused by Sclerotinia sclerotiorum. Plant Disease, 78, 924.
Kirkby, E. A., & Pilbeam, D. J. (1984). Calcium as a plant nutrient. Plant and Cell Environment, 7, 397–405.
Koike, S. T. (2000). Occurrence of stem rot of basil, caused by Sclerotinia sclerotiorum, in coastal California. Plant Disease, 84, 1342.
Kosman, E., & Cohen, Y. (1996). Procedures for calculating the differentiating synergism and antagonism in action of fungicide mixtures. Phytopathology, 86, 1263–1272.
Leigh, R. A., & Wyn Jones, R. G. (1984). A hypothesis relating critical potassium concentrations for growth to the distribution and functions of this ion in the plant cell. New Phytologist, 97, 1–13.
Levy, Y., Benderly, M., Cohen, Y., Gisi, U., & Bass, D. (1986). The joint action of fungicides in mixtures: Comparison of two methods for synergy calculation. Bulletin EPPO, 16, 651–657.
Liang, X., & Rollins, J. A. (2018). Mechanisms of broad host range necrotrophic pathogenesis in Sclerotinia sclerotiorum. Phytopathology, 108, 1128–1140.
Marschner, H. (1986). Mineral Nutrition of higher plants (pp. 229–448). London: Academic Press.
Mbengue, M., Navaud, O., Peyraud, R., Barascud, M., Badet, T., Vincent, R., Barbacci, A., & Raffaele, S. (2016). Emerging trends in molecular interactions between plants and the broad host range fungal pathogens Botrytis cinerea and Sclerotinia sclerotiorum. Frontiers in Plant Sciences, 31. https://doi.org/10.3389/fpls.2016.00422.
Newton, H. C., & Sequeira, L. (1972). Ascospores of primary infective propagules of Sclerotinia sclerotiorum in Wisconsin. Plant Disease Reporter, 56, 789–802.
Ouhibi, C., Attia, H., Nicot, P., Lecompte, F., Vidal, V., Lachaal, M., Urban, L., & Aarrouf, J. (2015). Effects of nitrogen supply and of UV-C irradiation on the susceptibility of Lactuca sativa L. to Botrytis cinerea and Sclerotinia minor. Plant and Soil, 393, 35–46.
Paula Júnior Trazilbo, J., Vieira, R. F., Teixeira, H., & Carneiro, J. E. S. (2009). Foliar application of calcium chloride and calcium silicate decreases white mold intensity on dry beans. Tropic. Plant Pathology, 34, 171–174.
Paulitz, T. C. (1997). First report of Sclerotinia sclerotiorum on basil in Canada. Plant Disease, 81, 229.
Poovaiah, B. W., Reddy, A. S. N., & McFadden, J. J. (1987). Calcium messenger system: Role of protein phosphorylation and inositol bisphospholipids. Physiologia Plantarum, 69, 569–573.
Prabhu, A. S., Fageria, N. K., Huber, D. M., & Rodrigues, F. A. (2008). Potassium and plant disease. In L. E. Datnoff, W. H. Elmer, & D. M. Huber (Eds.), Mineral nutrition and plant disease (pp. 57–78). St Paul: APS Press.
Schachtman, D. P., & Shin, R. (2006). Nutrient sensing and signaling: NPKS. Annual Review of Plant Biology, 58, 47–69.
Shtienberg, D., Elad, Y., Borenshtein, M., Ziv, G., Grava, A., & Cohen, S. (2010). Polyethylene mulch modulates greenhouse microclimate and reduces infection of Phytophthora infestans in tomato and Pseudoperonospora cubensis in cucumber. Phytopathology, 100, 97–104.
Suelter, C. H. (1970). Enzymes activated by monovalent cations. Science, 168, 789–795.
Sweeny, D. W., Granade, G. V., Eversmeyer, M. G., & Whitney, D. A. (2000). Phosphorus, potassium, chloride, and fungicide effects on wheat yield and leaf rust severity. Journal of Plant Nutrition, 23, 1267–1281.
Volpin, H., & Elad, Y. (1991). Influence of calcium nutrition on susceptibility of rose flowers to gray mold. Phytopathology, 81, 1390–1394.
Yermiyahu, U., Israeli, L., Rav David, D., Faingold, I., & Elad, Y. (2015). Higher potassium concentration in shoots reduces gray mold in sweet basil. Phytopathology, 105, 1059–1068.
Yermiyahu, U., Shamai, I., Peleg, R., Dudai, N., & Shtienberg, D. (2006). Reduction of Botrytis cinerea sporulation in sweet basil by altering the concentrations of nitrogen and calcium in the irrigation solution. Plant Pathology, 55, 544–552.
Youssef, K., & Roberto, S. R. (2014). Applications of salt solutions before and after harvest affect the quality and incidence of postharvest gray mold of 'Italia' table grapes. Postharvest Biology and Technology, 87, 95–102.
Acknowledgements
We acknowledge the assistance of Ahmed Hoshala, Ludmila Yosofov, Shoshana Suriano, Ziva Gilad, Ephraim Tzipilevitz, Ahiam Meir, Shahar Yitzhak, Tzion Deko, Dafna Harari, Shimon Pivonia, Ami Maduel, David Silverman, Shimon Biton, Yoel Hadad, Svetlana Dubrinin, Menahem Borenshtein and Ran Shulhani. This research was funded by the Herb Growers Board and by the Chief Scientist of the Israeli Ministry of Agriculture, project number 132-1408. Publication of the Agricultural Research Organization no. 543/14.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rav David, D., Yermiyahu, U., Fogel, M. et al. Plant nutrition for management of white mold in sweet basil. Phytoparasitica 47, 99–115 (2019). https://doi.org/10.1007/s12600-019-00716-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12600-019-00716-3