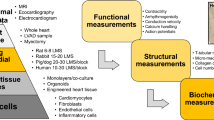
Abstract
The use of enzymatically isolated cardiac myocytes is ubiquitous in modern cardiovascular research. Parallels established between cardiomyocyte shortening responses and those of intact tissue make the cardiomyocyte an invaluable experimental model of cardiac function. Much of our understanding regarding the fundamental processes underlying heart function is owed to our increasing capabilities in single-cell stimulation and direct or indirect observation, as well as quantitative analysis of such cells. Of the many important mechanisms and functions that can be readily assessed in cardiomyocytes at all stages of development, contractility is the most representative and one of the most revealing. The purpose of this review is to provide a survey of various methodological approaches in the literature used to assess adult and neonatal cardiomyocyte contractility. The various methods employed to evaluate the contractile behavior of enzymatically isolated mammalian cardiac myocytes can be conveniently divided into two general categories—those employing optical (image)-based systems and those that use transducer-based technologies. This survey is by no means complete, but we have made an effort to include the most popular methods in terms of reliability and accessibility. These techniques are in constant evolution and hold great promise for the next generation of breakthrough studies in cell biology for the prevention, treatment, and cure of cardiovascular diseases.










Similar content being viewed by others
References
Addae-Mensah K, Wikswo J (2008) Measurement techniques for cellular biomechanics in vitro. Exp Biol Med 233(7):792–809
Azeloglu E, Costa KD (2010) Cross-bridge cycling gives rise to spatiotemporal heterogeneity of dynamic subcellular mechanics in cardiac myocytes probed with atomic force microscopy. Am J Physiol Heart Circ Physiol 298(3):H853–H860
Balaban N, Schwarz U, Riveline D, Goichberg P, Tzur G, Sabanay I, Mahalu D, Safran S, Bershadsky A, Addadi L, Geiger B (2001) Force and focal adhesion assembly: a close relationship studied using elastic micropatterned substrates. Nat Cell Biol 3(5):466–472
Banyasz T, Lozinskiy H, Payne C, Edelmann S, Norton B, Chen B, Chen-Izu Y, Izu L, Balke C (2007) Transformation of adult rat cardiac myocytes in primary culture. Exp Physiol 93(3):370–382
Bao G, Suresh S (2003) Cell and molecular mechanics of biological materials. Nat Mater 2:715–725
Baskin R, Lieber R, Oba T, Yeh Y (1981) Intensity of light diffraction from striated muscle as a function of incident angle. Biophys J 36:759–773
Bazan C, Torres D, Paolini P, Blomgren P (2009) Image processing techniques for assessing contractility in isolated adult cardiac myocytes. I J Biomed Imaging Vol. 2009(Article ID 352954):11 pages, doi:10.1155/2009/352954
Bazan C, Torres D, Paolini P, Blomgren P (2011) Image processing techniques for the assessment of contractile responses in neonatal cardiac myocytes. I J Biomed Imaging Vol. 2011(Article ID 729732):9 pages, doi:10.1155/2011/729732
Best P (1983) Cardiac muscle function: results from skinned fiber preparations. Am J Physiol 244(2):H167–H177
Binnig G, Quate C, Gerber C (1986) Atomic force microscope. Phys Rev Lett 56(9):930–933
Borejdo J, Mason P (1976) Sarcomere length changes during stimulation of frog semitendinosus muscle. J Mechanochem Cell Mot 3(3):155–161
Brady J (1991) Mechanical properties of isolated cardiac myocytes. Physiol Rev 71(2):413–428
Brady A, Tan S, Ricchiuti N (1979) Contractile force measured in unskinned isolated rat heart fibers. Nature 282(5740):728–729
Brenner S (2012) Sydney brenner quotes. Retrieved April 26, 2012, from http://todayinsci.com/B/Brenner_Sydney/BrennerSydney-Quotations.htm
Burke P (1999) Method and apparatus for measuring critical dimensions on a semiconductor surface. U.S. Patent 6,000,281, issued December 14, 1999
Buxboim A, Ivanovska I, Discher D (2010) Matrix elasticity, cytoskeletal forces and physics of the nucleus: how deeply do cells ‘feel’ outside and in? J Cell Sci 123:297–308
Cleworth D, Edman K (1972) Changes in sarcomere length during isometric tension development in frog skeletal muscle. J Physiol (Lond) 227(1):1–17
Copelas L, Briggs M, Grossman W, Morgan J (1987) A method for recording isometric tension development by isolated cardiac myocytes: transducer attachment with fibrin glue. Pflugers Arch 408(3):315–317
Crick F, Hughes A (1950) The physical properties of the cytoplasm. A study by means of the magnetic particle method. Part 1. Exp Cell Res 1(4):37–80
Curtis M, Russell B (2011) Micromechanical regulation in cardiac myocytes and fibroblasts: implications for tissue remodeling. Pflugers Arch, EJP 462(1):105–117
Delbridge L, Roos K (1997) Optical methods to evaluate the contractile function of unloaded isolated cardiac myocytes. J Mol Cell Cardiol 29(1):11–15
Dembo M, Wang Y (1999) Stresses at the cell-to-substrate interface during locomotion of fibroblasts. Biophys J 76(4):2307–2316
Diguet N, Mallat Y, Ladouce R, Clodic G, Prola A, Tritsch E, Blanc J, Larcher JC, Delcayre C, Samuel JL, Friguet B, Bolbach G, Li Z, Mericskay M (2011) Muscle creatine kinase deficiency triggers both actin depolymerization and desmin disorganization by advanced glycation end-products in dilated cardiomyopathy. J Biol Chem 286(40):35007–35019
Domke J, Parak W, George M, Gaub H, Radmacher M (1999) Mapping the mechanical pulse of single cardiomyocytes with the atomic force microscope. Eur Biophys J 28(3):179–186
Du Roure O, Saez A, Buguin A, Austin R, Chavrier P, Siberzan P, Ladoux B (2005) Force mapping in epithelial cell migration. Proc Natl Acad Sci USA 102(7):2390–2395
Edman K (1980) Depression of mechanical performance by active shortening during twitch and tetanus of vertebrate muscle fibres. Acta Physiol Scand 109(1):15–26
Fabiato A, Fabiato F (1976) Techniques of skinned cardiac cells and of isolated cardiac fibers with disrupted sarcolemmas with reference to the effects of catecholamines and of caffeine. Recent Adv Stud Cardiac Struct Metab 9:1–94
Fabry B, Maksym G, Shore S, Moore P, Panettieri R Jr, Butler J, Fredberg J (2001) Signal transduction in smooth muscle selected contribution: time course and heterogeneity of contractile responses in cultured human airway smooth muscle cells. J Appl Physiol 91(2):986–994
Farkasfalvi K, Stagg M, Coppen S, Siedlecka U, Lee J, Soppa G, Marczin N, Szokodi I, Yacoub M, Terracciano C (2007) Direct effects of apelin on cardiomyocyte contractility and electrophysiology. Biochem Biophys Res Commun 357(4):889–895
Flitney F, Hirst D (1978) Cross-bridge detachment and sarcomere "give" during stretch of active frog’s muscle. J Physiol 276:449–465
Gannier F, Bernengo J, Jacquemond V, Garnier D (1993) Measurements of sarcomere dynamics simultaneously with auxotonic force in isolated cardiac cells. IEEE Trans Biomed Eng 40(12):1226–1232
Garcia-Webb M, Taberner A, Hogan N, Hunter I (2007) A modular instrument for exploring the mechanics of cardiac myocytes. Am J Physiol Heart Circ Physiol 293(1):H866–H874
Gillis T, Liang B, Chung F, Tibbits G (2005) Increasing mammalian cardiomyocyte contractility with residues identified in trout troponin C. Physiol Genomics 22:1–7
Gosse C, Croquette V (2002) Magnetic tweezers: micromanipulation and force measurement at the molecular level. Biophys J 82(6):3314–3329
Hansma P, Drake B (1990) Scanning ion conductance microscopy. U.S. Patent 4,924,091, issued May 8, 1990
Harris A, Wild P, Stopak D (1980) Silicone rubber substrata: a new wrinkle in the study of cell locomotion. Science 208(4440):177–179
Haworth R, Griffin P, Saleh B, Goknur A, Berkoff H (1987) Contractile function of isolated young and adult rat heart cells. Am J Physiol 253(6 Pt 2):H1484–H1491
Hochmuth R (2000) Micropipette aspiration of living cells. J Biomech 33(1):15–22
Hu S, Eberhard L, Chen J, Love J, Butler J, Fredberg J, Whitesides G, Wang N (2004) Mechanical anisotropy of adherent cells probed by a three-dimensional magnetic twisting device. Am J Physiol Cell Ph 287(5):C1184–C1191
Huang J, Wu J, Qin L, Zhu T, Xiong C, Zhang Y, Fang J (2008) Mechanical behavior study of single cell contraction by digital image correlation technique. In: He X, Xie H, Kang Y (eds) Proc. SPIE, Nanjing, China, vol 7375, p 73754X
Iribe G, Helmes M, Kohl P (2007) Force-length relations in isolated intact cardiomyocytes subjected to dynamic changes in mechanical load. Am J Physiol Heart Circ Physiol 292(3):H1487–H1497
Iribe G, Ward C, Camelliti P, Bollensdorff C, Mason F, Burton R, Garny A, Morphew M, Hoenger A, Lederer W, Kohl P (2009) Axial stretch of rat single ventricular cardiomyocytes causes an acute and transient increase in Ca2+ spark rate. Circ Res 104(6):787–795
Ivester C, Kent R, Tagawa H, Tsutsui H, Imamura T, Cooper G, McDermott P (1993) Electrically stimulated contraction accelerates protein synthesis rates in adult feline cardiocytes. Am J Physiol 265(2 Pt 2):H666–H674
Iwazumi T (1987) High-speed ultrasensitive instrumentation for myofibril mechanics measurements. Am J Physiol Cell Physiol 252:C253–C262
Kajzar A, Cesa C, Kirchgeßner N, Hoffmann B, Merkel R (2008) Toward physiological conditions for cell analyses: forces of heart muscle cells suspended between elastic micropillars. Biophys J 94(5):1854–1866
Kamm R, Lammerding J, Mofrad M (2010) Cellular nanomechanics. Chapter 35 in: Bhushan B (ed) Handbook of nanotechnology. Springer, Berlin
Kawai M, Kuntz I (1973) Optical diffraction studies of muscle fibers. Biophys J 13(9):857–876
Kedrov A, Janovjak H, Sapra K, Muller D (2007) Deciphering molecular interactions of native membrane proteins by single-molecule force spectroscopy. Annu Rev Biophys Biomol 36:233–260
Kim D, Wong P, Park J, Levchenko A, Sun Y (2009) Microengineered platforms for cell mechanobiology. Annu Rev Biomed Eng 11:203–233
Korchev Y, Raval M, Lab M, Gorelik J, Edwards C, Rayment T, Klenerman D (2000) Hybrid scanning ion conductance and scanning near-field optical microscopy for the study of living cells. Biophys J 78(5):2675–2679
Korhonen T, Hanninnen S, Tavi P (2009) Model of excitation-contraction coupling of rat neonatal ventricular myocytes. Biophys J 96(3):1189–1209
Krueger J, Forletti D, Wittenberg B (1980) Uniform sarcomere shortening behavior in isolated cardiac muscle cells. J Gen Physiol 76(5):587–607
Lal R, John S (1994) Biological applications of atomic force microscopy. Am J Physiol Cell Physiol 266(1):C1–C21
Lal R, Drake B, Blumberg D, Saner D, Hansma P, Feinstein S (1995) Imaging real-time neurite outgrowth and cytoskeletal reorganization with an atomic force microscope. Am J Physiol Cell Physiol 269(1):C275–C285
Le Guennec J, Peineau N, Argibay J, Mongo K, Garnier D (1990) A new method of attachment of isolated mammalian ventricular myocytes for tension recording: length dependence of passive and active tension. J Mol Cell Cardiol 22(10):1083–1093
Lee J, Leonard M, Oliver T, Ishihara A, Jacobson K (1994) Traction forces generated by locomoting keratocytes. J Cell Biol 127(6):1957–1964
Lieber R, Yeh Y, Baskin R (1984) Sarcomere length determination using laser diffraction. Biophys J 45(5):1007–1016
Lin G, Pister K, Roos K (1995) Novel MEMS force transducer to quantify contractile characteristics from isolated cardiac muscle cells. J Electrochem Soc 142(3):L31–L33
Lin G, Pister K, Roos K (2000) Surface micromachined polysilicon heart cell force transducer. J Microeletromech Syst 9(1):9–17
Lin G, Palmer R, Pister K, Roos K (2001) Miniature heart cell force transducer system implemented in MEMS technology. IEEE Trans Biomed Eng 48(9):996–1006
Liu Y, Sonek G, Berns M, Tromberg B (1996) Physiological monitoring of optically trapped cells: effects of the confinement by 1064-nm laser tweezers using microfluorometry. Biophys J 71(4):2158–2167
Magid A, Reedy M (1980) X-ray diffraction observations of chemically skinned frog skeletal muscle processed by an improved method. Biophys J 30(1):27–40
Maltsev V, Sabbah H, Tanimura M, Lesch M, Goldstein S, Undrovinas A (1998) Relationship between action potential, contraction-relaxation pattern, and intracellular Ca2+ transient in cardiomyocytes of dogs with chronic heart failure. Cell Mol Life Sci 54(5):597–605
Mathur A, Collinsworth A, Reichert W, Kraus W, Truskey G (2001) Endothelial, cardiac muscle and skeletal muscle exhibit different viscous and elastic properties as determined by atomic force microscopy. J Biomech 34(12):1545–1553
Miragoli M, Moshkov A, Novak P, Shevchuk A, Nikolaev V, El-Hamamsy I, Potter C, Wright P, Abdul Kadir S, Lyon A, Mitchell J, Chester A, Klenerman D, Lab M, Korchev Y, Harding S, Gorelik J (2011) Scanning ion conductance microscopy: a convergent high-resolution technology for multi-parametric analysis of living cardiovascular cells. J R Soc Interface 8(60):913–925
Mitcheson JS, Hancox J, Levi A (1998) Cultured adult cardiac myocytes: future applications, culture methods, morphological and electrophysiological properties. Cardiovasc Res 39(2):280–300
Moss R, Halpern W (1977) Elastic and viscous properties of resting frog muscle. Biophys J 17(3):213–228
Mukherjee R, Spinale F, Crawford F (1992) Measurement of dynamic cellular and sarcomere contractile properties from the same cardiocyte. Proc IEEE Med Biol 2:392–393
Mukherjee R, Crawford F, Hewett K, Spinale F (1993) Cell and sarcomere contractile performance from the same cardiocyte using video microscopy. J Appl Physiol 74(4):2023–2033
Neuman K, Chadd E, Liou G, Bergman K, Block S (1999) Characterization of photo-damage to Escherichia coli in optical traps. Biophys J 77(5):2856–2863
Niggli E (1988) A laser diffraction system with improved sensitivity for long-time measurements of sarcomere dynamics in isolated cardiac myocytes. Pflugers Arch 411(4):462–468
Nishimura S, Yasuda S, Katoh M, Yamada K, Yamashita H, Saeki Y, Sunagawa K, Nagai R, Hisada T, Sugiura S (2004) Single cell mechanics of rat cardiomyocytes under isometric, unloaded, and physiologically loaded conditions. Am J Physiol Heart Circ Physiol 287(1):H196–H202
Nobelprize.org (2012) The nobel prize in physics 1986. http://www.nobelprize.org/nobel prizes/physics/laureates/1986/, accessed 10 Feb 2012
Oliver T, Dembo M, Jacobson K (1995) Traction forces in locomoting cells. Cell Motil Cytoskeleton 31(3):225–240
Palmer R, Brady A, Proos K (1996a) Mechanical measurements from isolated cardiac myocytes using a pipette attachment system. Am J Physiol Cell Physiol 270(39):C697–C704
Palmer R, Brady A, Roos K (1996b) Mechanical measurements from isolated cardiac myocytes using a pipette attachment system. Am J Physiol Cell Physiol 270(2):C697–C704
Paolini P, Sabbadini R, Roos K, Baskin R (1976) Sarcomere length dispersion in single skeletal muscle fibers and fiber bundles. Biophys J 16(8):919–929
Park J, Ryu J, Choi S, Seo E, Cha J, Ryu S, Kim J, Kim B, Lee S (2005) Realtime measurement of the contractile forces of self-organized cardiomyocytes on hybrid biopolymer microcantilevers. Anal Chem 77(20):6571–6580
Pelham R, Wang Y (1997) Cell locomotion and focal adhesions are regulated by substrate flexibility. Proc Natl Acad Sci USA 94(25):13,661–13,665
Prosser B, Ward C, Lederer W (2011) X-ROS signaling: rapid mechano-chemo transduction in heart. Science 333(6048):1440–1445
Puig de Morales M, Grabulosa M, Alcaraz J, Mullol J, Maksym G, Fredberg J, Navajas D (2001) Measurements of cell microheology by magnetic twisting cytometry with frequency domain demodulation. J Appl Physiol 91(3):1152–1159
Radmacher M (2002) Measuring the elastic properties of living cells by the atomic force microscope. In: Jena BP, Hörber JKH (eds) Atomic force microscopy in cell biology. Methods in cell biology, vol 68, Academic, San Diego, pp 67-90
Ren J, Wold L (2001) Measurement of cardiac mechanical function in isolated ventricular myocytes from rats and mice by computerized video-based imaging. Biol Proced Online 3(1)
Reuben H, Godinez M, Paolini P, Bejar E (1998) Analysis of contractile dynamics of adult cardiac myocytes using a computer controlled edge detector: effects of 5-hydroxytryptamine. Cardiovasc Pathobiol 2:149–158
Rüdel R, Zite-Ferenczy F (1979) Do laser diffraction studies on striated muscle indicate stepwise sarcomere shortening? Nature 278:573–575
Rugar D, Hansma P (1990) Atomic force microscope. Phys Today 43(10):23
Saks V, Veksler V, Kuznetsov A, Kay L, Sikk P, Tiivel T, Tranqui L, Olivares J, Winkler K, Wiedemann F, Kunz W (1998) Permeabilized cell and skinned fiber techniques in studies of mitochondrial function in vitro. Mol Cell Biochem 184(1–2):81–100
Schoenberg M, Wells J, Podolsky R (1974) Muscle compliance and the longitudinal transmission of mechanical impulses. J Gen Physiol 64(6):623–642
Schwarz U, Balaban N, Riveline D, Bershadsky A, Geiger B, Safran S (2002) Calculation of forces at focal adhesions from elastic substrata data: the effect of localized force and the need for regularization. Biophys J 83(3):1380–1394
Seki S, Ngashima M, Yamada Y, Tsutsuura M, Kobayashi T, Namiki A, Tohse N (2003) Fetal and postnatal development of Ca2+ transient and Ca2+ sparks in rat cardiomyocytes. Cardiovasc Res 58(3):535–548
Shevchuk A, Gorelik J, Harding S, Lab M, Klenerman D, Korchev Y (2001) Simultaneous measurement of Ca2+ and cellular dynamics: combined scanning ion conductance and optical microscopy to study contracting cardiac myocytes. Biophys J 81(3):1759–1764
Shroff S, Saner D, Lal R (1995) Dynamic micromechanical properties of cultured rat atrial myocytes measured by atomic force microscopy. Am J Physiol Cell Physiol 269(1):C286–C292
Slawnych M, Morishita L, Bressler B (1996) Spectral analysis of muscle fiber images as a means of assessing sarcomere heterogeneity. Biophys J 70(1):38–47
Sprenkle A, Murrary S, Glembotski C (1995) Involvement of multiple cis elements in basal-and α-adrenergic agonist-inducible atrial natriuretic factor transcription. Circ Res 77:1060–1069
Steadman B, Moore K, Spitzer K, Bridge J (1988) A video system for measuring motion in contracting heart cells. IEEE Trans Biomed Eng 35(4):264–272
Sugiura S, Nishimura S, Yasuda S, Hosoya Y, Katoh K (2006) Carbon fiber technique for the investigation of single-cell mechanics in intact cardiac myocytes. Nat Protoc 1(3):1453–1457
Sweitzer N, Moss R (1993) Determinants of loaded shortening velocity in single cardiac myocytes permeabilized with α-hemolysin. Circ Res 73(6):1150–1162
Tan J, Tien J, Pirone D, Gray D, Bhadriraju K, Chen C (2003) Cells lying on a bed of microneedles: an approach to isolate mechanical force. Proc Natl Acad Sci USA 100(4):1484–1489
ter Keurs H (2012) The interaction of Ca2+ with sarcomeric proteins: role in function and dysfunction of the heart. Am J Physiol Heart Circ Physiol 302(1):H38–H50
ter Keurs H, Boyden P (2007) Calcium and arrhythmogenesis. Physiol Rev 87(2):457–506
Ting L, Sniadecki N (2011) Biological microelectromechanical systems (BioMEMS) devices. In: Ducheyne P, Healy KE, Hutmacher DW, Grainger DW, Kirkpatrick CJ (eds) Comprehensive biomaterials, vol. 3. Elsevier, Waltham, pp. 257-276
Tseng Y, Kole T, Wirtz D (2002) Micromechanical mapping of live cells by multiple-particle-tracking microrheology. Biophys J 83(3):162–176
Veksler V, Lechene P, Matrougui K, Ventura-Clapier R (1997) Rigor tension in single skinned rat cardiac cell: role of myofibrillar creatine kinase. Cardiovasc Res 36(3):354–362
Walcott B, Dewey M (1980) Length-tension relation in limulus striated muscle. J Cell Biol 87(1):204–208
Weisenhorn A, Khorsandi M, Kasas S, Gotzos V, Butt HJ (1993) Deformation and height anomaly of soft surfaces studied with an AFM. Nanotechnology 4:106–113
Wolfe J, Ishiwata S, Braet F, Whan R, Su Y, dos Remedios C (2011) Spontaneous oscillatory contraction (spoc): auto-oscillations observed in striated muscle at partial activation. Biophys Rev 3:53–62
Wuite G, Davenport R, Rappaport A, Bustamante C (2000) An integrated laser trap/flow control video microscope for the study of single biomolecules. Biophys J 79(2):1155–1167
Wussling M, Schenk W, Nilius B (1987) A study of dynamic properties in isolated myocardial cells by the laser diffraction method. J Mol Cell Cardiol 19(9):897–907
Yamane M, Matsuda T, Ito T, Fujio Y, Takahashi K, Azuma J (2007) Rac1 activity is required for cardiac myocyte alignment in response to mechanical stress. Biochem Biophys Res Commun 353(4):1023–1027
Yasuda S, Sugiura S, Kobayakawa N, Fujita H, Yamashita H, Katoh K, Saeki Y, Kaneko H, Suda Y, Nagai R, Sugi H (2001) A novel method to study contraction characteristics of a single cardiac myocyte using carbon fibers. Am J Physiol Heart Circ Physiol 281(3):H1442–H1446
Yuan Y, Verma R (2006) Measuring microelastic properties of stratum corneum. Colloids Surf B 48(1):6–12
Zhao Y, Lim C, Sawyer D, Liao R, Zhang X (2005) Cellular force measurements using single-spaced polymeric microstructures: isolating cells from base substrate. J Micromech Microeng 15(9):1649–1656
Zhao Y, Lim C, Sawyer D, Liao R, Zhang X (2006) Microchip for subcellular mechanics study in living cells. Sensors Actuators B Chem 114(2):1108–1115
Zhao Y, Lim C, Sawyer D, Liao R, Zhang X (2007) Simultaneous orientation and cellular force measurements in adult cardiac myocytes using three-dimensional polymeric microstructures. Cell Motil Cytoskeleton 64(9):718–725
Zheng X, Zhang X (2011) Microsystems for cellular force measurement: a review. J Micromech Microeng 21:054003
Zhu J, Sabharwal T, Kalyanasundaram A, Guo L, Wang G (2009) Topo-graphic mapping and compression elasticity analysis of skinned cardiac muscle fibers in vitro with atomic force microscopy and nanoindentation. J Biomech 42(13):2143–2150
Acknowledgements
This work has been supported in part by NIH Roadmap Initiative award R90 DK07015 and NIH NIDDK, the California Metabolic Research Foundation, and the Computational Science Research Center at San Diego State University.
Conflict of interest
None
Author information
Authors and Affiliations
Corresponding author
Additional information
Celebrating emeritus professor Paul Paolini’s 70th birthday. On behalf of the students whose dreams he helped fulfill.
Special issue: Computational Biophysics
Rights and permissions
About this article
Cite this article
Bazan, C., Barba, D.T., Hawkins, T. et al. Contractility assessment in enzymatically isolated cardiomyocytes. Biophys Rev 4, 231–243 (2012). https://doi.org/10.1007/s12551-012-0082-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12551-012-0082-y