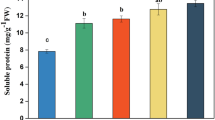
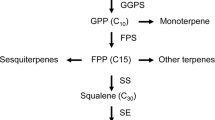
Abstract
Methyl jasmonate (MeJA) has been widely used to improve the biosynthesis of secondary metabolites such as triterpenoid saponins in medicinal plants. However, the underlying molecular mechanisms remain poorly understood. Differing from roots that accumulate protopanaxatriol-type saponins, Panax notoginseng leaves with a lower biomass mainly contain protopanaxadiol (PPD)-type saponins. Therefore, it is interesting to explore whether MeJA can activate the biosynthesis of PPD-type saponins in P. notoginseng leaves. In this study, we found MeJA could effectively induce the accumulation of PPD-type saponins, including ginsenoside Rb1, Rc, Rb2, Rb3 and notoginsenoside Fa, Fe in P. notoginseng leaves based on a newly established high-performance liquid chromatography method. Transcriptome analysis showed that differentially expressed genes (DEGs) induced by MeJA were mainly enriched in “terpenoid backbone biosynthesis”, “biosynthesis of unsaturated fatty acids”, “sesquiterpenoid and triterpenoid biosynthesis”, “fatty acid metabolism”, and “phenylpropanoid biosynthesis”. Furthermore, the expression profile and quantitative real-time PCR analysis of DEGs showed that MeJA could positively induce the molecular response of endogenous jasmonic acid (JA) signaling pathway, and increased PPD-type saponins mediated by MeJA in P. notoginseng leaves may be related to the high expression of FPS, SS, SE, DS and UGTs, and the low expression of CYP716A53v2 and β-AS. The results provide a molecular understanding for MeJA-elicited biosynthesis of triterpenoid saponins and facilitate the further characterization of the genes responsible for biosynthesis of PPD-type saponins in P. notoginseng leaves.





Similar content being viewed by others
Availability of data and material
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Afrin S, Huang J, Luo Z (2015) JA-mediated transcriptional regulation of secondary metabolism in medicinal plants. Sci Bull 60(12):1062–1072. https://doi.org/10.1007/s11434-015-0813-0
Bentebibel S, Moyano E, Palazon J, Cusido RM, Bonfill M, Eibl R et al (2005) Effects of immobilization by entrapment in alginate and scale-up on paclitaxel and baccatin III production in cell suspension cultures of Taxus baccata. Biotechnol Bioeng 89(6):647–655. https://doi.org/10.1002/bit.20321
Biswas T, Mathur A, Gupta V, Singh M, Mathur AK (2019) Salicylic acid and ultrasonic stress modulated gene expression and ginsenoside production in differentially affected Panax quinquefolius (L.) and Panax sikkimensis (Ban.) cell suspensions. Plant Cell Tissue Organ Cult 136(3):575–588. https://doi.org/10.1007/s11240-018-01538-7
Chen R, Li Q, Tan H, Chen J, Xiao Y, Ma R et al (2015) Gene-to-metabolite network for biosynthesis of lignans in MeJA-elicited Isatis indigotica hairy root cultures. Front Plant Sci 6:952. https://doi.org/10.3389/fpls.2015.00952
Corey EJ, Matsuda SP, Bartel B (1993) Isolation of an Arabidopsis thaliana gene encoding cycloartenol synthase by functional expression in a yeast mutant lacking lanosterol synthase by the use of a chromatographic screen. PNAS 90(24):11628–11632. https://doi.org/10.1073/pnas.90.24.11628
Cui Y, Mao R, Chen J, Guo Z (2019) Regulation mechanism of MYC family transcription factors in jasmonic acid signalling pathway on taxol biosynthesis. Int J Mol Sci 20(8):1843. https://doi.org/10.3390/ijms20081843
Dombrecht B, Xue GP, Sprague SJ, Kirkegaard JA, Ross JJ, Reid JB et al (2007) MYC2 differentially modulates diverse jasmonate-dependent functions in Arabidopsis. Plant Cell 19(7):2225–2245. https://doi.org/10.1105/tpc.106.048017
Grabherr MG, Haas BJ, Yassour M, Levin JZ, Thompson DA, Amit I et al (2011) Full-length transcriptome assembly from RNA-Seq data without a reference genome. Nat Biotechnol 29(7):644–652. https://doi.org/10.1038/nbt.1883
Han J, Hwang H, Choi S, Kim H, Choi Y (2012) Cytochrome P450 CYP716A53v2 catalyzes the formation of protopanaxatriol from protopanaxadiol during ginsenoside biosynthesis in Panax Ginseng. Plant Cell Physiol 53(9):1535–1545. https://doi.org/10.1093/pcp/pcs106
Hu F, Zhong J (2008) Jasmonic acid mediates gene transcription of ginsenoside biosynthesis in cell cultures of Panax notoginseng treated with chemically synthesized 2-hydroxyethyl jasmonate. Process Biochem 43(1):113–118. https://doi.org/10.1016/j.procbio.2007.10.010
Jiao C, Gu Z (2018) iTRAQ-based proteomic analysis reveals changes in response to sodium nitroprusside treatment in soybean sprouts. Food Chem 292:372–376. https://doi.org/10.1016/j.foodchem.2018.02.054
Jung SC, Kim W, Park SC, Jeong J, Park MK, Lim S et al (2014) Two ginseng UDP glycosyltransferases synthesize ginsenoside Rg3 and Rd. Plant Cell Physiol 55(12):2177–2188. https://doi.org/10.1093/pcp/pcu147
Ketchum RE, Gibson DM, Croteau RB, Shuler ML (1999) The kinetics of taxoid accumulation in cell suspension cultures of Taxus following elicitation with methyl jasmonate. Biotechnol Bioeng 62(1):97–105. https://doi.org/10.1002/(SICI)1097-0290(19990105)62:1%3c97::AID-BIT11%3e3.0.CO;2-C
Khezerluo M, Hosseini B, Amiri J (2018) Sodium nitroprusside stimulated production of tropane alkaloids and antioxidant enzymes activity in hairy root culture of Hyoscyamus reticulatus L. Acta Biol Hung 69(4):437–448. https://doi.org/10.1556/018.69.2018.4.6
Kikowska M, Kowalczyk M, Stochmal A, Thiem B (2019) Enhanced accumulation of triterpenoid saponins in in vitro plantlets and dedifferentiated cultures of Eryngium planum L.: a medicinal plant. Hortic Environ Biotechnol 60(1):147–154. https://doi.org/10.1007/s13580-018-0103-2
Kim D (2012) Chemical diversity of Panax ginseng, Panax quinquifolium, and Panax notoginseng. J Ginseng Res 36(1):1–15. https://doi.org/10.5142/jgr.2012.36.1.1
Kim OT, Bang KH, Kim YC et al (2009) Upregulation of ginsenoside and gene expression related to triterpene biosynthesis in ginseng hairy root cultures elicited by methyl jasmonate. Plant Cell Tissue Organ Cultn 98(1):25–33. https://doi.org/10.1007/s11240-009-9535-9
Kim Y, Zhang D, Yang D (2015) Biosynthesis and biotechnological production of ginsenosides. Biotechnol Adv 33(6):717–735. https://doi.org/10.1016/j.biotechadv.2015.03.001
Kochan E, Balcerczak E, Szymczyk P, Sienkiewicz M, Zielińska-Bliźniewska H, Szymańska G (2019) Abscisic acid regulates the 3-hydroxy-3-methylglutaryl CoA reductase gene promoter and ginsenoside production in Panax quinquefolium hairy root cultures. Int J Mol Sci 20(6):1310. https://doi.org/10.3390/ijms20061310
Kushiro T, Ohno Y, Shibuya Y, Ebizuka Y (1997) In vitro conversion of 2,3-oxidosqualene into dammarenediol by Panax ginseng microsomes. Biol Pharm Bull 20(3):292–294. https://doi.org/10.1248/bpb.20.292
Kushiro T, Shibuya M, Ebizuka Y (1998) Beta-amyrin synthase-cloning of oxidosqualene cyclase that catalyzes the formation of the most popular triterpene among higher plants. Eur J Biochem 256(1):238–244. https://doi.org/10.1046/j.1432-1327.1998.2560238.x
Lee MH, Jeong JH, Seo JW, Shin CG, Kim YS, In JG et al (2004) Enhanced triterpene and phytosterol biosynthesis in Panax ginseng overexpressing squalene synthase gene. Plant Cell Physiol 45(8):976–984. https://doi.org/10.1093/pcp/pch126
Major IT, Yoshida Y, Campos ML, Kapali G, Xin XF, Sugimoto K et al (2017) Regulation of growth-defense balance by the JASMONATE ZIM-DOMAIN (JAZ)-MYC transcriptional module. New Phytol 215(4):1533–1547. https://doi.org/10.1111/nph.14638
Mortazavi A, Williams BA et al (2008) Mapping and quantifying mammalian transcriptomes by RNA-Seq. Nat Methods 5(7):621–628
Patra B, Pattanaik S, Schluttenhofer C, Yuan L (2018) A network of jasmonate-responsive bHLH factors modulate monoterpenoid indole alkaloid biosynthesis in Catharanthus roseus. New Phytol 217(4):1566–1581. https://doi.org/10.1111/nph.14910
Sabater-Jara A, Onrubia M, Moyano E, Bonfill M, Palazón J, Pedreño MA et al (2014) Synergistic effect of cyclodextrins and methyl jasmonate on taxane production in Taxus × media cell cultures. Plant Biotechnol J 12(8):1075–1084. https://doi.org/10.1111/pbi.12214
Shi H, Liu G, Wei Y, Chan Z (2018) The zinc-finger transcription factor ZAT6 is essential for hydrogen peroxide induction of anthocyanin synthesis in Arabidopsis. Plant Mol Biol 97(1–2):165–176. https://doi.org/10.1007/s11103-018-0730-0
Thanh NT, Murthy HN, Yu KW, Hahn EJ, Paek KY (2005) Methyl jasmonate elicitation enhanced synthesis of ginsenoside by cell suspension cultures of Panax ginseng in 5-l balloon type bubble bioreactors. Appl Microbiol Biotechnol 67(2):197–201. https://doi.org/10.1007/s00253-004-1759-3
Tung B, Hai N (2016) Phytochemical and pharmacology effect of Panax notoginseng. J Pharm Sci US 6(8):174–178. https://doi.org/10.7324/JAPS.2016.60828
Um Y, Lee Y, Kim S, Jeong Y, Kim G, Choi D et al (2017) Expression analysis of ginsenoside biosynthesis-related genes in methyl jasmonate-treated adventitious roots of Panax ginseng via DNA microarray analysis. Hortic Environ Biotechnol 58(4):376–383. https://doi.org/10.1007/s13580-017-0041-4
Wan JB, Yang FQ, Li SP, Wang YT, Cui XM (2006) Chemical characteristics for different parts of Panax notoginseng using pressurized liquid extraction and HPLC-ELSD. J Pharm Biomed 41(5):1596–1601. https://doi.org/10.1016/j.jpba.2006.01.058
Wang W, Zhang Z, Zhong J (2005) Enhancement of ginsenoside biosynthesis in high-density cultivation of Panax notoginseng cells by various strategies of methyl jasmonate elicitation. Appl Microbiol Biotechnol 67(6):752–758. https://doi.org/10.1007/s00253-004-1831-z
Wang T, Yu X, Qu S, Xu H, Han B, Sui D (2010) Effects of ginsenoside Rb3 on myocardial and heart function impairment induced by isoproterenol in rats. Eur J Pharmacol 636(1–3):121–125. https://doi.org/10.1016/j.ejphar.2010.03.035
Wang J, Gao W, Zhang J, Zuo B, Zhang L, Huang L (2012) Advances in study of ginsenoside biosynthesis pathway in Panax ginseng C. A. Meyer. Acta Physiol Plant 34(2):397–403. https://doi.org/10.1007/s11738-011-0844-3
Wang Y, Dong J, Liu P, Lau CW, Gao Z, Zhou D et al (2014) Ginsenoside Rb3 attenuates oxidative stress and preserve endothelial function in renal arteries from hypertensive rats. Br J Pharmacol 171(13):3171–3181. https://doi.org/10.1111/bph.12660
Wang T, Guo R, Zhou G et al (2016) Traditional uses, botany, phytochemistry, pharmacology and toxicology of Panax notoginseng (Burk.) F.H. Chen: a review. J Ethnopharmacol 188:234–258. https://doi.org/10.1016/j.jep.2016.05.005
Wang N, Wang L, Qi L, Lu X (2018) Construct a gene-to-metabolite network to screen the key genes of triterpene saponin biosynthetic pathway in Panax notoginseng. Biotechnol Appl Biochem 65(2):119–127. https://doi.org/10.1002/bab.1580
Wang P, Wei W, Ye W, Li X, Zhao W, Yang C et al (2019) Synthesizing ginsenoside Rh2 in Saccharomyces cerevisiae cell factory at high-efficiency. Cell Discov 5(1):5. https://doi.org/10.1038/s41421-018-0075-5
Wang D, Wang J, Shi Y, Li R, Fan F, Huang Y, Li W, Chen N, Huang L, Dai Z et al (2020a) Elucidation of the complete biosynthetic pathway of the main triterpene glycosylation products of Panax notoginseng using a synthetic biology platform. Metab Eng 61:131–140. https://doi.org/10.1016/j.ymben.2020.05.007
Wang J, Song L, Gong X, Xu J, Li M (2020b) Functions of jasmonic acid in plant regulation and response to abiotic stress. Int J Mol Sci 21(4):1446. https://doi.org/10.3390/ijms21041446
Weber H (2002) Fatty acid-derived signals in plants. Trends Plant Sci 7(5):217–224. https://doi.org/10.1016/S1360-1385(02)02250-1
Wei G, Dong L, Yang J, Zhang L, Xu J, Yang F et al (2018) Integrated metabolomic and transcriptomic analyses revealed the distribution of saponins in Panax notoginseng. Acta Pharm Sin B 8(3):458–465. https://doi.org/10.1016/j.apsb.2017.12.010
Xiang L, Zhu S, Zhao T, Zhang M, Liu W, Chen M et al (2015) Enhancement of artemisinin content and relative expression of genes of artemisinin biosynthesis in Artemisia annua by exogenous MeJA treatment. Plant Growth Regul 75(2):435–441. https://doi.org/10.1007/s10725-014-0004-z
Yang X, Xiong X, Wang H, Wang J (2014) Protective effects of Panax notoginseng saponins on cardiovascular diseases: a comprehensive overview of experimental studies. Evid Based Complement Altern 2014:1–13. https://doi.org/10.1155/2014/204840
Yang J, Hu Z, Zhang T, Gu A, Gong T, Zhu P (2018) Progress on the studies of the key enzymes of ginsenoside biosynthesis. Molecules 23(3):589. https://doi.org/10.3390/molecules23030589
Yu KW, Gao WY, Hahn EJ, Paek KY (2002) Jasmonic acid improves ginsenoside accumulation in adventitious root culture of Panax ginseng C.A. Meyer. Biochem Eng J 11(2–3):211–215. https://doi.org/10.1016/S1369-703X(02)00029-3
Yu X, Zhang W, Zhang Y, Zhang X, Lang D, Zhang X (2019) The roles of methyl jasmonate to stress in plants. Funct Plant Biol 46(3):197–212. https://doi.org/10.1071/FP18106
Zhang H, Hedhili S, Montiel G, Zhang Y, Chatel G, Pré M et al (2011) The basic helix-loop-helix transcription factor CrMYC2 controls the jasmonate-responsive expression of the ORCA genes that regulate alkaloid biosynthesis in Catharanthus roseus. Plant J 67(1):61–71. https://doi.org/10.1111/j.1365-313X.2011.04575.x
Zhao S, Wang L, Liu L, Liang Y, Sun Y, Wu J (2014) Both the mevalonate and the non-mevalonate pathways are involved in ginsenoside biosynthesis. Plant Cell Rep 33(3):393–400. https://doi.org/10.1007/s00299-013-1538-7
Zhao Y, Guo W, Sun X, Li K, Liu K, Wang J et al (2020) A culture system for the stable and high-efficiency proliferation of adventitious roots of Panax notoginseng and ginsenoside accumulation. Ind Crop Prod 157:112882. https://doi.org/10.1016/j.indcrop.2020.112882
Zhou P, Yang J, Zhu J, He S, Zhang W, Yu R et al (2015) Effects of β-cyclodextrin and methyl jasmonate on the production of vindoline, catharanthine, and ajmalicine in Catharanthus roseus cambial meristematic cell cultures. Appl Microbiol Biot 99(17):7035–7045. https://doi.org/10.1007/s00253-015-6651-9
Funding
This work was supported by the National Key Research and Development Plant (no. 2017YFC1702500), the Major Science and Technique Programs in Yunnan Province (no. 2016ZF001), the Yunnan Ten Thousand Talents Plan Young & Elite Talents Project (YNWR-QNBJ-2018-047) and the project of young and middle-aged talent of Yunnan province (no. 2019HB019).
Author information
Authors and Affiliations
Contributions
YL and YL designed the experiments; BJ, GC, HS, RX, XL, JT and QT carried out the experiments and analyzed the data; WF and YJ wrote the manuscript, SY and GZ reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declared that they have no conflicts of interest to this work. We declare that we do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Ethical approval
Manuscript is approved by all authors for publication. I would like to declare on behalf of my co-authors that this work described was original comment that has not been published previously, and not under consideration for publication elsewhere, in whole or in part. All the authors listed have approved the manuscript that is enclosed.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Lin, Y., Jia, B. et al. Transcriptome Analysis Reveals Molecular Mechanisms Underlying Methyl Jasmonate-mediated Biosynthesis of Protopanaxadiol-type Saponins in Panax notoginseng Leaves. J. Plant Biol. 65, 29–41 (2022). https://doi.org/10.1007/s12374-021-09331-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12374-021-09331-4