Abstract
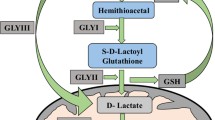
Glyoxal (GO) and methylglyoxal (MG) are reactive carbonyl compounds that are accumulated in vivo through various pathways. They are presumably detoxified through multiple pathways including glutathione (GSH)-dependent/independent glyoxalase systems and NAD(P)H dependent reductases. Previously, we reported an involvement of aldo-ketoreductases (AKRs) in MG detoxification. Here, we investigated the role of various AKRs (YqhE, YafB, YghZ, YeaE, and YajO) in GO metabolism. Enzyme activities of the AKRs to GO were measured, and GO sensitivities of the corresponding mutants were compared. In addition, we examined inductions of the AKR genes by GO. The results indicate that AKRs efficiently detoxify GO, among which YafB, YghZ, and YeaE are major players.
Similar content being viewed by others
References
Abordo, E.A., Minhas, H.S., and Thornalley, P.J. 1999. Accumulation of alpha-oxoaldehydes during oxidative stress: a role in cytotoxicity. Biochem. Pharmacol. 58, 641–648.
Altaras, N.E. and Cameron, D.C. 1999. Metabolic engineering of a 1,2-propanediol pathway in Escherichia coli. Appl. Environ. Microbiol. 65, 1180–1185.
Baldoma, L. and Aguilar, J. 1987. Involvement of lactaldehyde dehydrogenase in several metabolic pathways of Escherichia coli K12. J. Biol. Chem. 262, 13991–13996.
Benov, L. and Fridovich, I. 2002. Induction of the soxRS regulon of Escherichia coli by glycolaldehyde. Arch. Biochem. Biophys. 407, 45–48.
Boronat, A. and Aguilar, J. 1979. Rhamnose-induced propanediol oxidoreductase in Escherichia coli: Purification, properties, and comparison with the fucose-induced enzyme. J. Bacteriol. 140, 320–326.
Brownlee, M. 1995. Advanced protein glycosylation in diabetes and aging. Annu. Rev. Med. 46, 223–234.
Cocks, G.T., Aguilar, T., and Lin, E.C. 1974. Evolution of L-1, 2-propanediol catabolism in Escherichia coli by recruitment of enzymes for L-fucose and L-lactate metabolism. J. Bacteriol. 118, 83–88.
Kasai, H., Iwamoto-Tanaka, N., and Fukada, S. 1998. DNA modifications by the mutagen glyoxal: adduction to G and C, deamination of C and GC and GA cross-linking. Carcinogenesis 19, 1459–1465.
Ko, J., Kim, I., Yoo, S., Min, B., Kim, K., and Park, C. 2005. Conversion of methylglyoxal to acetol by Escherichia coli aldo-keto reductases. J. Bacteriol. 187, 5782–5789.
Kwon, M., Lee, J., Lee, C., and Park, C. 2012. Genomic rearrangements leading to overexpression of aldo-keto reductase YafB of Escherichia coli confer resistance to glyoxal. J. Bacteriol. 194, 1979–1988.
Lee, C., Kim, I., Lee, J., Lee, K.L., Min, B., and Park, C. 2010. Transcriptional activation of the aldehyde reductase YqhD by YqhC and its implication in glyoxal metabolism of Escherichia coli K-12. J. Bacteriol. 192, 4205–4214.
Luth, H.J., Ogunlade, V., Kuhla, B., Kientsch-Engel, R., Stahl, P., Webster, J., Arendt, T., and Munch, G. 2005. Age- and stage-dependent accumulation of advanced glycation end products in intracellular deposits in normal and Alzheimer’s disease brains. Cereb. Cortex 15, 211–220.
Munch, G., Luth, H.J., Wong, A., Arendt, T., Hirsch, E., Ravid, R., and Riederer, P. 2000. Crosslinking of alpha-synuclein by advanced glycation endproducts—an early pathophysiological step in Lewy body formation? J. Chem. Neuroanat. 20, 253–257.
Obradors, N., Cabiscol, E., Aguilar, J., and Ros, J. 1998. Site-directed mutagenesis studies of the metal-binding center of the iron-dependent propanediol oxidoreductase from Escherichia coli. Eur. J. Biochem. 258, 207–213.
Okado-Matsumoto, A. and Fridovich, I. 2000. The role of alpha, beta-dicarbonyl compounds in the toxicity of short chain sugars. J. Biol. Chem. 275, 34853–34857.
Rondeau, P. and Bourdon, E. 2011. The glycation of albumin: Structural and functional impacts. Biochimie 93, 645–658.
Salmon, K., Hung, S.P., Mekjian, K., Baldi, P., Hatfield, G.W., and Gunsalus, R.P. 2003. Global gene expression profiling in Escherichia coli K12. The effects of oxygen availability and FNR. J. Biol. Chem. 278, 29837–29855.
Subedi, K.P., Choi, D., Kim, I., Min, B., and Park, C. 2011. Hsp31 of Escherichia coli K-12 is glyoxalase III. Mol. Microbiol. 81, 926–936.
Thornalley, P.J. 1990. The glyoxalase system: new developments towards functional characterization of a metabolic pathway fundamental to biological life. Biochem. J. 269, 1–11.
Thornalley, P.J. 1998. Glutathione-dependent detoxification of alpha-oxoaldehydes by the glyoxalase system: Involvement in disease mechanisms and antiproliferative activity of glyoxalase I inhibitors. Chem. Biol. Interact. 111-112, 137–151.
Thornalley, P.J., Langborg, A., and Minhas, H.S. 1999. Formation of glyoxal, methylglyoxal and 3-deoxyglucosone in the glycation of proteins by glucose. Biochem. J. 344 Pt 1, 109–116.
Tucker, N.P., Le Brun, N.E., Dixon, R., and Hutchings, M.I. 2010. There’s NO stopping NsrR, a global regulator of the bacterial NO stress response. Trends Microbiol. 18, 149–156.
Turner, P.C., Miller, E.N., Jarboe, L.R., Baggett, C.L., Shanmugam, K.T., and Ingram, L.O. 2011. YqhC regulates transcription of the adjacent Escherichia coli genes yqhD and dkgA that are involved in furfural tolerance. J. Ind. Microbiol. Biotechnol. 38, 431–439.
Vander Jagt, D.L. and Hunsaker, L.A. 2003. Methylglyoxal metabolism and diabetic complications: Roles of aldose reductase, glyoxalase-I, betaine aldehyde dehydrogenase and 2-oxoaldehyde dehydrogenase. Chem. Biol. Interact. 143-144, 341–351.
Vander Jagt, D.L., Robinson, B., Taylor, K.K., and Hunsaker, L.A. 1992. Reduction of trioses by NADPH-dependent aldo-keto reductases. Aldose reductase, methylglyoxal, and diabetic complications. J. Biol. Chem. 267, 4364–4369.
Zhu, Y. and Lin, E.C. 1989. L-1,2-propanediol exits more rapidly than L-lactaldehyde from Escherichia coli. J. Bacteriol. 171, 862–867.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, C., Kim, I. & Park, C. Glyoxal detoxification in Escherichia coli K-12 by NADPH dependent aldo-keto reductases. J Microbiol. 51, 527–530 (2013). https://doi.org/10.1007/s12275-013-3087-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12275-013-3087-8