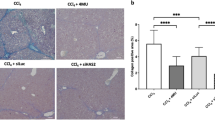
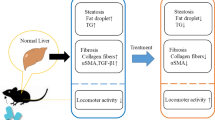
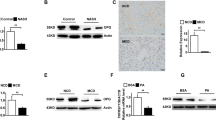
Abstract
Hyaluronan (HA) as a glycosaminoglycan can bind to cell-surface receptors, such as TLR4, to regulate inflammation, tissue injury, repair, and fibrosis. 4-methylumbelliferone (4-MU), an inhibitor of HA synthesis, is a drug used for the treatment of biliary spasms. Currently, therapeutic interventions are not available for non-alcoholic steatohepatitis (NASH). In this study, we investigated the effects of 4-MU on NASH using a choline-deficient amino acid (CDAA) diet model. CDAA diet-fed mice showed NASH characteristics, including hepatocyte injury, hepatic steatosis, inflammation, and fibrogenesis. 4-MU treatment significantly reduced hepatic lipid contents in CDAA diet-fed mice. 4-MU reversed CDAA diet-mediated inhibition of Ppara and induction of Srebf1 and Slc27a2. Analysis of serum ALT and AST levels revealed that 4-MU treatment protected against hepatocellular damage induced by CDAA diet feeding. TLR4 regulates low molecular weight-HA-induced chemokine expression in hepatocytes. In CDAA diet-fed, 4-MU-treated mice, the upregulated chemokine/cytokine expression, such as Cxcl1, Cxcl2, and Tnf was attenuated with the decrease of macrophage infiltration into the liver. Moreover, HA inhibition repressed CDAA diet-induced mRNA expression of fibrogenic genes, Notch1, and Hes1 in the liver. In conclusion, 4-MU treatment inhibited liver steatosis and steatohepatitis in a mouse model of NASH, implicating that 4-MU may have therapeutic potential for NASH.






Similar content being viewed by others
References
Aly FZ, Kleiner DE (2011) Update on fatty liver disease and steatohepatitis. Adv Anat Pathol 18:294–300. https://doi.org/10.1097/PAP.0b013e318220f59b
Andreichenko IN, Tsitrina AA, Fokin AV, Gabdulkhakova AI, Maltsev DI, Perelman GS, Bulgakova EV, Kulikov AM, Mikaelyan AS, Kotelevtsev YV (2019) 4-Methylumbelliferone prevents liver fibrosis by affecting hyaluronan deposition, FSTL1 expression and cell localization. Int J Mol Sci. https://doi.org/10.3390/ijms20246301
Bertola A, Bonnafous S, Anty R, Patouraux S, Saint-Paul MC, Iannelli A, Gugenheim J, Barr J, Mato JM, Le Marchand-Brustel Y, Tran A, Gual P (2010) Hepatic expression patterns of inflammatory and immune response genes associated with obesity and NASH in morbidly obese patients. PLoS ONE 5:e13577. https://doi.org/10.1371/journal.pone.0013577
Bourguignon V, Flamion B (2016) Respective roles of hyaluronidases 1 and 2 in endogenous hyaluronan turnover. FASEB J 30:2108–2114. https://doi.org/10.1096/fj.201500178R
Chedid MF (2017) Nonalcoholic steatohepatitis: the second leading indication for liver transplantation in the USA. Dig Dis Sci 62:2621–2622. https://doi.org/10.1007/s10620-017-4724-6
Csak T, Velayudham A, Hritz I, Petrasek J, Levin I, Lippai D, Catalano D, Mandrekar P, Dolganiuc A, Kurt-Jones E, Szabo G (2011) Deficiency in myeloid differentiation factor-2 and toll-like receptor 4 expression attenuates nonalcoholic steatohepatitis and fibrosis in mice. Am J Physiol Gastrointest Liver Physiol 300:G433-441. https://doi.org/10.1152/ajpgi.00163.2009
Diehl AM, Day C (2017) Cause, pathogenesis, and treatment of nonalcoholic steatohepatitis. N Engl J Med 377:2063–2072. https://doi.org/10.1056/NEJMra1503519
Dietrich CG, Rau M, Jahn D, Geier A (2017) Changes in drug transport and metabolism and their clinical implications in non-alcoholic fatty liver disease. Expert Opin Drug Metab Toxicol 13:625–640. https://doi.org/10.1080/17425255.2017.1314461
Flanagan BF, Dalchau R, Allen AK, Daar AS, Fabre JW (1989) Chemical composition and tissue distribution of the human CDw44 glycoprotein. Immunology 67:167–175
Gao XX, Shi DH, Chen YX, Cui JT, Wang YR, Jiang CP, Wu JH (2012) The therapeutic effects of tectorigenin on chemically induced liver fibrosis in rats and an associated metabonomic investigation. Arch Pharm Res 35:1479–1493. https://doi.org/10.1007/s12272-012-0819-y
Girish KS, Kemparaju K (2007) The magic glue hyaluronan and its eraser hyaluronidase: a biological overview. Life Sci 80:1921–1943. https://doi.org/10.1016/j.lfs.2007.02.037
Gourd E (2018) PEGPH20 for metastatic pancreatic ductal adenocarcinoma. Lancet Oncol 19:e81. https://doi.org/10.1016/S1470-2045(17)30953-1
Haldar D, Kern B, Hodson J, Armstrong MJ, Adam R, Berlakovich G, Fritz J, Feurstein B, Popp W, Karam V, Muiesan P, O’grady J, Jamieson N, Wigmore SJ, Pirenne J, Malek-Hosseini SA, Hidalgo E, Tokat Y, Paul A, Pratschke J, Bartels M, Trunecka P, Settmacher U, Pinzani M, Duvoux C, Newsome PN, Schneeberger S, European L, Intestine Transplant A (2019) Outcomes of liver transplantation for non-alcoholic steatohepatitis: a European Liver Transplant Registry study. J Hepatol 71:313–322. https://doi.org/10.1016/j.jhep.2019.04.011
Haukeland JW, Damas JK, Konopski Z, Loberg EM, Haaland T, Goverud I, Torjesen PA, Birkeland K, Bjoro K, Aukrust P (2006) Systemic inflammation in nonalcoholic fatty liver disease is characterized by elevated levels of CCL2. J Hepatol 44:1167–1174. https://doi.org/10.1016/j.jhep.2006.02.011
Hoshino K, Takeuchi O, Kawai T, Sanjo H, Ogawa T, Takeda Y, Takeda K, Akira S (1999) Cutting edge: toll-like receptor 4 (TLR4)-deficient mice are hyporesponsive to lipopolysaccharide: evidence for TLR4 as the Lps gene product. J Immunol 162:3749–3752
Hwang S, He Y, Xiang X, Seo W, Kim SJ, Ma J, Ren T, Park SH, Zhou Z, Feng D, Kunos G, Gao B (2019) Interleukin-22 ameliorates neutrophil-driven nonalcoholic steatohepatitis through multiple targets. Hepatology. https://doi.org/10.1002/hep.31031
Hwang S, He Y, Xiang X, Seo W, Kim SJ, Ma J, Ren T, Park SH, Zhou Z, Feng D, Kunos G, Gao B (2020a) Interleukin-22 ameliorates neutrophil-driven nonalcoholic steatohepatitis through multiple targets. Hepatology 72:412–429. https://doi.org/10.1002/hep.31031
Hwang S, Wang X, Rodrigues RM, Ma J, He Y, Seo W, Park SH, Kim SJ, Feng D, Gao B (2020b) Protective and detrimental roles of p38alpha MAPK in different stages of nonalcoholic fatty liver disease. Hepatology. https://doi.org/10.1002/hep.31390
Itano N, Sawai T, Yoshida M, Lenas P, Yamada Y, Imagawa M, Shinomura T, Hamaguchi M, Yoshida Y, Ohnuki Y, Miyauchi S, Spicer AP, Mcdonald JA, Kimata K (1999) Three isoforms of mammalian hyaluronan synthases have distinct enzymatic properties. J Biol Chem 274:25085–25092. https://doi.org/10.1074/jbc.274.35.25085
Jiang D, Liang J, Noble PW (2011) Hyaluronan as an immune regulator in human diseases. Physiol Rev 91:221–264. https://doi.org/10.1152/physrev.00052.2009
Jiang D, Liang J, Fan J, Yu S, Chen S, Luo Y, Prestwich GD, Mascarenhas MM, Garg HG, Quinn DA, Homer RJ, Goldstein DR, Bucala R, Lee PJ, Medzhitov R, Noble PW (2005) Regulation of lung injury and repair by Toll-like receptors and hyaluronan. Nat Med 11:1173–1179. https://doi.org/10.1038/nm1315
Kim SS, Kim HK, Kim H, Lee WT, Lee ES, Oh KT, Choi HG, Youn YS (2020) Hyperthermal paclitaxel-bound albumin nanoparticles co-loaded with indocyanine green and hyaluronidase for treating pancreatic cancers. Arch Pharm Res. https://doi.org/10.1007/s12272-020-01264-9
Kodama Y, Taura K, Miura K, Schnabl B, Osawa Y, Brenner DA (2009) Antiapoptotic effect of c-Jun N-terminal Kinase-1 through Mcl-1 stabilization in TNF-induced hepatocyte apoptosis. Gastroenterology 136:1423–1434. https://doi.org/10.1053/j.gastro.2008.12.064
Krenkel O, Tacke F (2017) Liver macrophages in tissue homeostasis and disease. Nat Rev Immunol 17:306–321. https://doi.org/10.1038/nri.2017.11
Lake AD, Novak P, Fisher CD, Jackson JP, Hardwick RN, Billheimer DD, Klimecki WT, Cherrington NJ (2011) Analysis of global and absorption, distribution, metabolism, and elimination gene expression in the progressive stages of human nonalcoholic fatty liver disease. Drug Metab Dispos 39:1954–1960. https://doi.org/10.1124/dmd.111.040592
Lee J, Park JS, Roh YS (2019) Molecular insights into the role of mitochondria in non-alcoholic fatty liver disease. Arch Pharm Res 42:935–946. https://doi.org/10.1007/s12272-019-01178-1
Liang J, Jiang D, Noble PW (2016) Hyaluronan as a therapeutic target in human diseases. Adv Drug Deliv Rev 97:186–203. https://doi.org/10.1016/j.addr.2015.10.017
Machado MV, Diehl AM (2016) Pathogenesis of nonalcoholic steatohepatitis. Gastroenterology 150:1769–1777. https://doi.org/10.1053/j.gastro.2016.02.066
Mahlbacher V, Sewing A, Elsasser HP, Kern HF (1992) Hyaluronan is a secretory product of human pancreatic adenocarcinoma cells. Eur J Cell Biol 58:28–34
Matsumoto M, Hada N, Sakamaki Y, Uno A, Shiga T, Tanaka C, Ito T, Katsume A, Sudoh M (2013) An improved mouse model that rapidly develops fibrosis in non-alcoholic steatohepatitis. Int J Exp Pathol 94:93–103. https://doi.org/10.1111/iep.12008
Mckallip RJ, Ban H, Uchakina ON (2015) Treatment with the hyaluronic acid synthesis inhibitor 4-methylumbelliferone suppresses LPS-induced lung inflammation. Inflammation 38:1250–1259. https://doi.org/10.1007/s10753-014-0092-y
Michelotti GA, Machado MV, Diehl AM (2013) NAFLD, NASH and liver cancer. Nat Rev Gastroenterol Hepatol 10:656–665. https://doi.org/10.1038/nrgastro.2013.183
Nagy N, Kuipers HF, Frymoyer AR, Ishak HD, Bollyky JB, Wight TN, Bollyky PL (2015) 4-methylumbelliferone treatment and hyaluronan inhibition as a therapeutic strategy in inflammation, autoimmunity, and cancer. Front Immunol 6:123. https://doi.org/10.3389/fimmu.2015.00123
Nagy N, Gurevich I, Kuipers HF, Ruppert SM, Marshall PL, Xie BJ, Sun W, Malkovskiy AV, Rajadas J, Grandoch M, Fischer JW, Frymoyer AR, Kaber G, Bollyky PL (2019) 4-Methylumbelliferyl glucuronide contributes to hyaluronan synthesis inhibition. J Biol Chem 294:7864–7877. https://doi.org/10.1074/jbc.RA118.006166
Patouraux S, Rousseau D, Bonnafous S, Lebeaupin C, Luci C, Canivet CM, Schneck AS, Bertola A, Saint-Paul MC, Iannelli A, Gugenheim J, Anty R, Tran A, Bailly-Maitre B, Gual P (2017) CD44 is a key player in non-alcoholic steatohepatitis. J Hepatol 67:328–338. https://doi.org/10.1016/j.jhep.2017.03.003
Piccioni F, Malvicini M, Garcia MG, Rodriguez A, Atorrasagasti C, Kippes N, Piedra Buena IT, Rizzo MM, Bayo J, Aquino J, Viola M, Passi A, Alaniz L, Mazzolini G (2012) Antitumor effects of hyaluronic acid inhibitor 4-methylumbelliferone in an orthotopic hepatocellular carcinoma model in mice. Glycobiology 22:400–410. https://doi.org/10.1093/glycob/cwr158
Piccioni F, Fiore E, Bayo J, Atorrasagasti C, Peixoto E, Rizzo M, Malvicini M, Tirado-Gonzalez I, Garcia MG, Alaniz L, Mazzolini G (2015) 4-methylumbelliferone inhibits hepatocellular carcinoma growth by decreasing IL-6 production and angiogenesis. Glycobiology 25:825–835. https://doi.org/10.1093/glycob/cwv023
Ramanathan RK, Mcdonough SL, Philip PA, Hingorani SR, Lacy J, Kortmansky JS, Thumar J, Chiorean EG, Shields AF, Behl D, Mehan PT, Gaur R, Seery T, Guthrie KA, Hochster HS (2019) Phase IB/II randomized study of folfirinox plus pegylated recombinant human hyaluronidase versus FOLFIRINOX alone in patients with metastatic pancreatic adenocarcinoma: SWOG S1313. J Clin Oncol 37:1062–1069. https://doi.org/10.1200/JCO.18.01295
Roh YS, Zhang B, Loomba R, Seki E (2015) TLR2 and TLR9 contribute to alcohol-mediated liver injury through induction of CXCL1 and neutrophil infiltration. Am J Physiol Gastrointest Liver Physiol 309:G30-41. https://doi.org/10.1152/ajpgi.00031.2015
Saiman Y, Friedman SL (2012) The role of chemokines in acute liver injury. Front Physiol 3:213. https://doi.org/10.3389/fphys.2012.00213
Schinzel RT, Higuchi-Sanabria R, Shalem O, Moehle EA, Webster BM, Joe L, Bar-Ziv R, Frankino PA, Durieux J, Pender C, Kelet N, Kumar SS, Savalia N, Chi H, Simic M, Nguyen NT, Dillin A (2019) The hyaluronidase, TMEM2, promotes ER homeostasis and longevity independent of the UPR(ER). Cell 179(1306–1318):e1318. https://doi.org/10.1016/j.cell.2019.10.018
Schuster S, Cabrera D, Arrese M, Feldstein AE (2018) Triggering and resolution of inflammation in NASH. Nat Rev Gastroenterol Hepatol 15:349–364. https://doi.org/10.1038/s41575-018-0009-6
Seki E, De Minicis S, Osterreicher CH, Kluwe J, Osawa Y, Brenner DA, Schwabe RF (2007) TLR4 enhances TGF-beta signaling and hepatic fibrosis. Nat Med 13:1324–1332. https://doi.org/10.1038/nm1663
Sim MO, Ham JR, Lee HI, Seo KI, Lee MK (2014) Long-term supplementation of umbelliferone and 4-methylumbelliferone alleviates high-fat diet induced hypertriglyceridemia and hyperglycemia in mice. Chem Biol Interact 216:9–16. https://doi.org/10.1016/j.cbi.2014.03.003
Simone P, Alberto M (2015) Caution should be used in long-term treatment with oral compounds of hyaluronic acid in patients with a history of cancer. Clin Drug Investig 35:689–692. https://doi.org/10.1007/s40261-015-0339-x
Stern R (2004) Hyaluronan catabolism: a new metabolic pathway. Eur J Cell Biol 83:317–325. https://doi.org/10.1078/0171-9335-00392
Tamaki S, Ueno T, Torimura T, Sata M, Tanikawa K (1996) Evaluation of hyaluronic acid binding ability of hepatic sinusoidal endothelial cells in rats with liver cirrhosis. Gastroenterology 111:1049–1057
Tsuchida T, Friedman SL (2017) Mechanisms of hepatic stellate cell activation. Nat Rev Gastroenterol Hepatol 14:397–411. https://doi.org/10.1038/nrgastro.2017.38
Yang L, Miura K, Zhang B, Matsushita H, Yang YM, Liang S, Song J, Roh YS, Seki E (2017) TRIF differentially regulates hepatic steatosis and inflammation/fibrosis in mice. Cell Mol Gastroenterol Hepatol 3:469–483. https://doi.org/10.1016/j.jcmgh.2016.12.004
Yang YM, Fukui M, Wang Z, Miao F, Karriker MJ, Seki E (2018) Interventional potential of recombinant feline hepatocyte growth factor in a mouse model of non-alcoholic steatohepatitis. Front Endocrinol (Lausanne) 9:378. https://doi.org/10.3389/fendo.2018.00378
Yang YM, Noureddin M, Liu C, Ohashi K, Kim SY, Ramnath D, Powell EE, Sweet MJ, Roh YS, Hsin IF, Deng N, Liu Z, Liang J, Mena E, Shouhed D, Schwabe RF, Jiang D, Lu SC, Noble PW, Seki E (2019) Hyaluronan synthase 2-mediated hyaluronan production mediates Notch1 activation and liver fibrosis. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aat9284
Yates TJ, Lopez LE, Lokeshwar SD, Ortiz N, Kallifatidis G, Jordan A, Hoye K, Altman N, Lokeshwar VB (2015) Dietary supplement 4-methylumbelliferone: an effective chemopreventive and therapeutic agent for prostate cancer. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djv085
Ye D, Li FY, Lam KS, Li H, Jia W, Wang Y, Man K, Lo CM, Li X, Xu A (2012) Toll-like receptor-4 mediates obesity-induced non-alcoholic steatohepatitis through activation of X-box binding protein-1 in mice. Gut 61:1058–1067. https://doi.org/10.1136/gutjnl-2011-300269
Zhu C, Kim K, Wang X, Bartolome A, Salomao M, Dongiovanni P, Meroni M, Graham MJ, Yates KP, Diehl AM, Schwabe RF, Tabas I, Valenti L, Lavine JE, Pajvani UB (2018) Hepatocyte Notch activation induces liver fibrosis in nonalcoholic steatohepatitis. Sci Transl Med. https://doi.org/10.1126/scitranslmed.aat0344
Acknowledgements
This study was supported by NIH Grant R01DK085252, R21AA025841, and P01CA233452 (E.S). This study was supported by 2019 Research Grant from Kangwon National University (Y.M.Y.) and the National Research Foundation of Korea (NRF) grant funded by the Korea government (MIST) (# 2020R1C1C1004185) (Y.M.Y).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, Y.M., Wang, Z., Matsuda, M. et al. Inhibition of hyaluronan synthesis by 4-methylumbelliferone ameliorates non-alcoholic steatohepatitis in choline-deficient l-amino acid-defined diet-induced murine model. Arch. Pharm. Res. 44, 230–240 (2021). https://doi.org/10.1007/s12272-021-01309-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-021-01309-7