Abstract
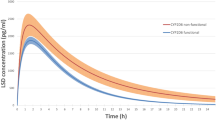
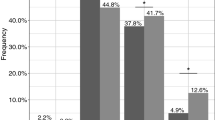
The aim of this study was to investigate the effects of paroxetine, a potent inhibitor of CYP2D6, on the pharmacokinetics of atomoxetine and its two metabolites, 4-hydroxyatomoxetine and N-desmethylatomoxetine, in different CYP2D6 genotypes. Twenty-six healthy subjects were recruited and divided into CYP2D6*wt/*wt (*wt=*1 or *2, n = 10), CYP2D6*wt/*10 (n = 9), and CYP2D6*10/*10 groups (n = 7). In atomoxetine phase, all subjects received a single oral dose of atomoxetine (20 mg). In paroxetine phase, after administration of a single oral dose of paroxetine (20 mg) for six consecutive days, all subjects received a single oral dose of atomoxetine with paroxetine. Plasma concentrations of atomoxetine and its metabolites were determined up to 24 h after dosing. During atomoxetine phase, there were significant differences in Cmax and AUC0−24 of atomoxetine and N-desmethylatomoxetine among three genotype groups, whereas significant differences were not found in relation to CYP2D6*10 allele after administration of paroxetine. AUC ratios of 4-hydroxyatomoxetine and N-desmethylatomoxetine to atomoxetine were significantly different among three genotype groups during atomoxetine phase (all, P < 0.001), but after paroxetine treatment significant differences were not found. After paroxetine treatment, AUC0−24 of atomoxetine was increased by 2.3-, 1.7-, and 1.3-fold, in CYP2D6*wt/*wt, CYP2D6*wt/*10, and CYP2D6*10/*10 groups in comparison to atomoxetine phase, respectively. AUC ratio of 4-hydroxyatomoxetine to atomoxetine in each group was significantly decreased, whereas AUC ratio of N-desmethylatomoxetine to atomoxetine significantly increased after administration of paroxetine. In conclusion, paroxetine coadministration significantly affected pharmacokinetic parameters of atomoxetine and its two metabolites, 4-hydroxyatomoxetine and N-desmethylatomoxetine. When atomoxetine was administered alone, Cmax, AUC0-24 and CL/F of atomoxetine were significantly different among the three CYP2D6 genotype groups. However, after paroxetine coadministration, no significant differences in these pharmacokinetic parameters were observed among the CYP2D6 genotype groups.


Similar content being viewed by others
References
Atomoxetine ADHD, Comorbid MDDS, Group, Bangs ME, Emslie GJ, Spencer TJ, Ramsey JL, Carlson C, Bartky EJ, Busner J, Duesenberg DA, Harshawat P, Kaplan SL, Quintana H, Allen AJ, Sumner CR (2007) Efficacy and safety of atomoxetine in adolescents with attention-deficit/hyperactivity disorder and major depression. J Child Adolesc Psychopharmacol 17:407–420. doi:https://doi.org/10.1089/cap.2007.0066
Belle DJ, Ernest CS, Sauer JM, Smith BP, Thomasson HR, Witcher JW (2002) Effect of potent CYP2D6 inhibition by paroxetine on atomoxetine pharmacokinetics. J Clin Pharmacol 42:1219–1227. doi:https://doi.org/10.1177/009127002762491307
Byeon JY, Kim YH, Na HS, Jang JH, Kim SH, Lee YJ, Bae JW, Kim IS, Jang CG, Chung MW, Lee SY (2015) Effects of the CYP2D6*10 allele on the pharmacokinetics of atomoxetine and its metabolites. Arch Pharm Res 38:2083–2091. doi:https://doi.org/10.1007/s12272-015-0646-z
Byeon JY, Kim YH, Kim SH, Lee CM, Jung EH, Chae WK, Jang CG, Lee SY, Lee YJ (2018a) The influences of CYP2C9*1/*3 genotype on the pharmacokinetics of zolpidem. Arch Pharm Res 41(9):931–936. doi:https://doi.org/10.1007/s12272-018-1070-y
Byeon JY, Kim YH, Kim SH, Lee CM, Jung EH, Chae WK, Jang CG, Lee SY, Lee YJ (2018b) Effects of genetic polymorphisms of CYP2C19 on the pharmacokinetics of zolpidem. Arch Pharm Res 41(8):861–866. doi:https://doi.org/10.1007/s12272-018-1065-8
Byeon JY, Kim YH, Lee CM, Kim SH, Chae WK, Jung EH, Choi CI, Jang CG, Lee SY, Bae JW, Lee YJ (2018c) CYP2D6 allele frequencies in Korean population, comparison with East Asian, Caucasian and African populations, and the comparison of metabolic activity of CYP2D6 genotypes. Arch Pharm Res 41(9):921–930. doi:https://doi.org/10.1007/s12272-018-1075-6
Byeon JY, Lee YJ, Kim YH, Kim SH, Lee CM, Bae JW, Jang CG, Lee SY, Choi CI (2018d) Effects of diltiazem, a moderate inhibitor of CYP3A4, on the pharmacokinetics of tamsulosin in different CYP2D6 genotypes. Arch Pharm Res 41(5):564–570. doi:https://doi.org/10.1007/s12272-018-1030-6
Byeon JY, Lee CM, Lee YJ, Kim YH, Kim SH, Jung EH, Chae WK, Lee YJ, Jang CG, Lee SY (2019) Influence of CYP2D6 genetic polymorphism on pharmacokinetics of active moiety of tolterodine. Arch Pharm Res 42(2):182–190. https://doi.org/10.1007/s12272-018-1099-y
Choi CI, Bae JW, Lee HI, Jang CG, Sohn UD, Lee SY (2012b) Determination of atomoxetine metabolites in human plasma by liquid chromatography/tandem mass spectrometry and its application to a pharmacokinetic study. J Chromatogr B Analyt Technol Biomed Life Sci 885–886:103–108. doi:https://doi.org/10.1016/j.jchromb.2011.12.023
Choi CI, Bae JW, Jang CG, Lee SY (2012a) Tamsulosin exposure is significantly increased by the CYP2D6* 10/* 10 genotype. J Clin Pharmacol 52:1934–1938. doi:https://doi.org/10.1177/0091270011432168
Choi CI, Jang CG, Bae JW, Lee SY (2013) Validation of an analytical LC–MS/MS method in human plasma for the pharmacokinetic study of atomoxetine. J Anal Chem 68:986–991. https://doi.org/10.1134/S1061934813110051
Choi CI, Bae JW, Lee YJ, Lee HI, Jang CG, Lee SY (2014) Effects of CYP2C19 genetic polymorphisms on atomoxetine pharmacokinetics. J Clin Psychopharmacol 34:139–142. doi:https://doi.org/10.1097/JCP.0b013e3182a608a2
Crewe HK, Lennard MS, Tucker GT, Woods FR, Haddock RE (1992) The effect of selective serotonin re-uptake inhibitors on cytochrome P4502D6 (CYP2D6) activity in human liver microsomes. Br J Clin Pharmacol 34:262–265. doi:https://doi.org/10.1111/j.1365-2125.1992.tb04134.x
Cui YM, Teng CH, Pan AX, Yuen E, Yeo KP, Zhou Y, Zhao X, Long AJ, Bangs ME, Wise SD (2007) Atomoxetine pharmacokinetics in healthy Chinese subjects and effect of the CYP2D6*10 allele. Br J Clin Pharmacol 64:445–449. doi:https://doi.org/10.1111/j.1365-2125.2007.02912.x
Desta Z, Zhao X, Shin J-G, Flockhart D (2002) Clinical significance of the cytochrome P450 2C19 genetic polymorphism. Clin Pharmacokinet 41:913–958. https://doi.org/10.2165/00003088-200241120-00002
Gehlert DR, Gackenheimer SL, Robertson DW (1993) Localization of rat brain binding sites for [3H] tomoxetine, an enantiomerically pure ligand for norepinephrine reuptake sites. Neurosci Lett 157:203–206. doi:https://doi.org/10.1016/0304-3940(93)90737-6
Heydorn WE (1999) Paroxetine: a review of its pharmacology, pharmacokinetics and utility in the treatment of a variety of psychiatric disorders. Expert Opin Investig Drugs 8:417–441. doi:https://doi.org/10.1517/13543784.8.4.417
Jeppesen U, Gram L, Vistisen K, Loft S, Poulsen H, Brøsen K (1996) Dose-dependent inhibition of CYP1A2, CYP2C19 and CYP2D6 by citalopram, fluoxetine, fluvoxamine and paroxetine. Eur J Clin Pharmacol 51:73–78. doi:https://doi.org/10.1007/s002280050163
Kim SH, Kim DH, Byeon JY, Kim YH, Kim DH, Lim HJ, Lee CM, Whang SS, Choi CI, Bae JW, Lee YJ, Jang CG, Lee SY (2017) Effects of CYP2C9 genetic polymorphisms on the pharmacokinetics of celecoxib and its carboxylic acid metabolite. Arch Pharm Res 40:382–390. doi:https://doi.org/10.1007/s12272-016-0861-2
Kim MJ, Byeon JY, Kim YH, Kim SH, Lee CM, Jung EH, Chae WK, Lee YJ, Jang CG, Lee SY, Choi CI (2018a) Effect of the CYP2D6*10 allele on the pharmacokinetics of clomiphene and its active metabolites. Arch Pharm Res 41:347–353. doi:https://doi.org/10.1007/s12272-018-1005-7
Kim SH, Byeon JY, Kim YH, Lee CM, Lee YJ, Jang CG, Lee SY (2018b) Physiologically based pharmacokinetic modelling of atomoxetine with regard to CYP2D6 genotypes. Sci Rep 8:12405. doi:https://doi.org/10.1038/s41598-018-30841-8
Lee HI, Bae JW, Choi CI, Lee YJ, Byeon JY, Jang CG (2014) Lee SY (2014) Strongly increased exposure of meloxicam in CYP2C9*3/*3 individuals. Pharmacogenet Genomics. 24(2):113–7. https://doi.org/10.1097/FPC.0000000000000025
Lee HJ, Kim YH, Kim SH, Lee CM, Yang AY, Jang CG, Lee SY, Bae JW, Choi CI (2016) Effects of CYP2C9 genetic polymorphisms on the pharmacokinetics of zafirlukast. Arch Pharm Res 39:1013–1019. doi:https://doi.org/10.1007/s12272-016-0785-x
Lee HI, Byeon JY, Kim YH, Lee CM, Choi CI, Jang CG, Bae JW, Lee YJ, Lee SY (2018) Effects of CYP2C19 and CYP3A5 genetic polymorphisms on the pharmacokinetics of cilostazol and its active metabolites. Eur J Clin Pharmacol 74(11):1417–1426. doi:https://doi.org/10.1007/s00228-018-2522-5
Matsui A, Azuma J, Witcher JW, Long AJ, Sauer JM, Smith BP, DeSante KA, Read HA, Takahashi M, Nakano M (2012) Pharmacokinetics, safety, and tolerability of atomoxetine and effect of CYP2D6*10/*10 genotype in healthy Japanese men. J Clin Pharmacol 52:388–403. doi:https://doi.org/10.1177/0091270011398657
Munir K, Biederman J, Knee D (1987) Psychiatric comorbidity in patients with attention deficit disorder: a controlled study. J Am Acad Child Adolesc Psychiatry 26:844–848. doi:https://doi.org/10.1097/00004583-198726060-00008
Pary R, Lewis S, Matuschka PR, Rudzinskiy P, Safi M, Lippmann S (2002) Attention deficit disorder in adults. Ann Clin Psychiatry 14:105–111. doi:https://doi.org/10.1023/a:1016807021779
Ring BJ, Gillespie JS, Eckstein JA, Wrighton SA (2002) Identification of the human cytochromes P450 responsible for atomoxetine metabolism. Drug Metab Dispos 30:319–323. doi:https://doi.org/10.1124/dmd.30.3.319
Sauer JM, Ponsler GD, Mattiuz EL, Long AJ, Witcher JW, Thomasson HR, Desante KA (2003) Disposition and metabolic fate of atomoxetine hydrochloride: the role of CYP2D6 in human disposition and metabolism. Drug Metab Dispos 31:98–107. doi:https://doi.org/10.1124/dmd.31.1.98
Sim SC, Risinger C, Dahl ML, Aklillu E, Christensen M, Bertilsson L, Ingelman-Sundberg M (2006) A common novel CYP2C19 gene variant causes ultrarapid drug metabolism relevant for the drug response to proton pump inhibitors and antidepressants. Clin Pharmacol Ther 79:103–113. doi:https://doi.org/10.1016/j.clpt.2005.10.002
Sindrup SH, Brosen K, Gram LF, Hallas J, Skjelbo E, Allen A, Allen GD, Cooper SM, Mellows G, Tasker TCG, Zussman BD (1992) The relationship between paroxetine and the sparteine oxidation polymorphism. Clin Pharmacol Ther 51:278–287. doi:https://doi.org/10.1038/clpt.1992.23
Spencer T, Biederman J, Wilens T, Harding M, O’Donnell D, Griffin S (1996) Pharmacotherapy of attention-deficit hyperactivity disorder across the life cycle. J Am Acad Child Adolesc Psychiatry 35:409–432. doi:https://doi.org/10.1080/10826084.2016.1273955
Swanson JM, Sergeant JA, Taylor E, Sonuga-Barke EJ, Jensen PS, Cantwell DP (1998) Attention-deficit hyperactivity disorder and hyperkinetic disorder. Lancet 351:429–433. https://doi.org/10.12968/hmed.2010.71.11.79663
Teh LK, Bertilsson L (2012) Pharmacogenomics of CYP2D6: molecular genetics, interethnic differences and clinical importance. Drug Metab Pharmacokinet 27:55–67. https://doi.org/10.2133/dmpk.dmpk-11-rv-121
Wong DT, Threlkeld PG, Best KL, Bymaster FP (1982) A new inhibitor of norepinephrine uptake devoid of affinity for receptors in rat brain. J Pharmacol Exp Ther 222:61–65
Acknowledgements
This research was supported the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT, & Future Planning (NRF-2019R1A2C1004582).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Eui Hyun Jung and Yun Jeong Lee contributed equally to this study.
Rights and permissions
About this article
Cite this article
Jung, E.H., Lee, Y.J., Kim, DH. et al. Effects of paroxetine on the pharmacokinetics of atomoxetine and its metabolites in different CYP2D6 genotypes. Arch. Pharm. Res. 43, 1356–1363 (2020). https://doi.org/10.1007/s12272-020-01300-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-020-01300-8