Abstract
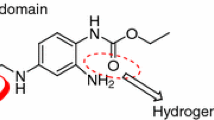
Selective GABAA receptor ligands are widely used clinically to reduce the occurrence of convulsions. Hence there is an intense interest in developing new benzimidazole derivatives demonstrating high selectivity and high affinity for GABAA receptors. With the purpose of designing new chemical entities with an enhanced binding affinity for GABAA/BZd receptor complex, we carried out a QSAR study on benzotriazine derivatives. We studied 28 potent GABAA receptor ligands; derivatives of benzotriazines, using a combination of various tested physicochemical, steric, electronic and thermodynamic descriptors to determine the quantitative correlation between binding affinity and structural features. The developed and validated final model showed a good correlative and predictive ability expressed by a squared correlation co-efficient (r2) of 0.954. The equation indicated that the binding affinity is strongly dependent upon the thermodynamic properties (CDE, DDE and PC). Correlation between these properties and anticonvulsant activity was used to synthesize compounds possessing potent anticonvulsant activity. Most of the compounds showed an ability to inhibit the maximum electroshock (MES) and pentylenetetrazole (PTZ)-induced convulsions. Compound 1A, i.e. 2-(4-Chloro-phenyl)-5-nitro-1H-benzimidazole exhibited maximum activity in both the convulsion models.
Similar content being viewed by others
References
Abbas, S., Rastkari, N., and Sharifzadeh, M., Anticonvulsant activities of new 1,4-dihydropyridine derivatives containing 4-nitroimidazolyl substituents. DARU, 12, 81–86 (2004).
Beguin, C., Arnaud, L. T., Stables, J. P., Voyksner, R. D., and Kohn, H., N-Substituted amino acid N-benzylamides: synthesis, anticonvulsant, and metabolic activities. Bioorg. Med. Chem., 12, 3079–3096 (2004).
Chaltterjee, S., Hadi, A. S., and Price, B., Regression Analysis by Examples. Wiley VCH, New York, USA, (2000).
Chen, J., Sun, X. Y., Chai, K. Y., Lee, J. S., Song, M. S., and Quana, Z. S., Synthesis and anticonvulsant evaluation of 4-(4-alkoxylphenyl)-3-ethyl-4H-1, 2, 4-triazoles as openchain analogues of 7-alkoxyl-4,5-dihydro [1, 2, 4] triazolo [4, 3-a] quinolines. Bioorg. Med. Chem., 15, 6775–6781 (2007).
Clark, C. R., Lin, C. M., and Sansom, R. T., Anticonvulsant activity of 2- and 3-aminobenzanilides. J. Med. Chem., 29, 1534–1537 (1986).
Dawood, K. M., Gawad, H. A., Rageb, E. A., Ellithey, M., and Mohamed, H. A., Synthesis, anticonvulsant, and antiinflammatory evaluation of some new benzotriazole and benzofuran-based heterocycles. Bioorg. Med. Chem., 14, 3672–3680 (2006).
Diudea, M. V., QSPR/QSAR Studies for Molecular Descriptors, Nova Science. Huntingdon, New York, USA, (2000).
Falcó, J. L., Piqué, M., González, M., Buira, I., Méndez, E., Terencio, J., Pérez, C., PrÍncep, M., Palomer, A., and Guglietta, A., Synthesis, pharmacology and molecular modeling of N-substituted 2-phenyl-indoles and benzimidazoles as potent GABAA agonists. Eur. J. Med. Chem., 41, 985–990 (2006).
Guerrini, G., Ciciani, G., Cambi, G., Bruni, F., Selleri, S., Melani, F., Montali, M., Martini, C., Ghelardini, C., Norcini, M., and Costanzoa, A., Novel 3-aroylpyrazolo [5,1-c][1,2,4] benzotriazine 5-oxides 8-substituted, ligands at GABAA/benzodiazepine receptor complex: Synthesis, pharmacological and molecular modeling studies. Bioorg. Med. Chem., 16, 4471–4489 (2008).
Guo, L. J., Cheng, X. W., Jing, H. J., Zhao, L. M., and Quan, Z. S., Design and synthesis of 5-alkoxy-[1,2,4]triazolo[4,3-a]quinoline derivatives with anticonvulsant activity. Eur. J. Med. Chem., 44, 954–958 (2009).
Hansch, C., Maloney, P. P., Fujita, T., and Muir, R. M., Correlation of biological activity of phenoxyacetic acids with Hammett substituent constants and partition coefficients. Nature, 194, 178–180 (1962).
Hansch, C., Hoekman, D., and Gao H., Comparative QSAR: Towards a deeper understanding of chemicobiological interactions. Chem. Rev., 96, 1045–1076 (1996).
Hemmateenejad, B., Correlation ranking procedure for factor selection in PC-ANN modeling and application to ADMETox evaluation. Chemom. Intell. Lab. Syst., 75, 231–245 (2005).
Kamal, A., Kumar, P. P., Sreekanth, K., Seshadri, B. N., and Ramulu, P., Synthesis of new benzimidazole linked pyrrolo [2, 1-c] [1, 4] benzodiazepine conjugates with efficient DNA-binding affinity and potent cytotoxicity. Bioorg. Med. Chem. Lett., 18, 2594–2598 (2008).
Kung, C. H., John, N. D., and Kwon, C. H., Synthesis and structure activity study of N-1 substituted (S)-(+)-5-n- Propyl-2-iminohydantoins as potential anticonvulsant agents. Drug Dev. Res., 47, 17–26 (1999).
Kuzmanovic, S. O. P., Cvetkoviæ, D. D., and Barna, D. J., QSAR analysis of 2-Amino or 2-Methyl-1-substituted benzimidazoles against Pseudomonas aeruginosa. Int. J. Mol. Sci., 10, 1670–1682 (2009).
Malawska, B., Kulig, K., Agnieszka, S., and Stables, J. P., Investigation into new anticonvulsant derivatives of substituted N-benzylamides of hydroxy and acetoxybutyric acid. Part 5: Search for new anticonvulsant compounds. Bioorg. Med. Chem., 12, 625–632 (2004).
Moreau, S., Coudert, P., Rubat, C., Danielle, V. G., Gardette, D., Gramainc, J. C., and Couquelet, J., Synthesis and anticonvulsant properties of Triazolo- and Imidazopyridazinyl carboxamides and carboxylic acids. Bioorg. Med. Chem., 6, 983–991 (1998).
Pandeya, S. N., Yogeeswari, P., and Stables, J. P., Synthesis and anticonvulsant activity of 4-bromophenyl substituted aryl semicarbazones. Eur. J. Med. Chem., 35, 879–886 (2000).
Patel, H. J., Sarra, J., Caruso, F., Rossi, M., Doshia, U., and Stephan, R. A., Synthesis and anticonvulsant activity of new N-1′, N-3′-disubstituted-2′H, 3H, 5′H-spiro-(2-benzofuran-1,4′-imidazolidine)-2′, 3, 5′-triones. Bioorg. Med. Chem. Lett., 16, 4644–4647 (2006).
Shafiee, A., Rastkari, N., and Sharifzadeh, M., Anticonvulsant activities of new 1,4-Dihydropyridine derivatives containing 4-Nitroimidazolyl substituents. DARU, 12, 81–86 (2004).
Shimshoni, J. A., Bialera, M., and Yagen, B., Synthesis and anticonvulsant activity of aromatic tetramethylcyclopro-panecarboxamide derivatives. Bioorg. Med. Chem., 16, 6297–6305 (2008).
Snedecor, G. W. and Cochran, W. G., in Statistical Methods. Oxford and IBH, New Delhi, India, (1967).
Su, Q. C., Ren, T. R., Cai, X. M., and Zhi, G. S., Modeling of αk/γ2 (k = 1, 2, 3 and 5) interface of GABAA receptor and docking studies with zolpidem: Implications for selectivity. J. Mol. Graph. Model., 26, 537–545 (2007).
Tomczuk, B. E., Taylor, C. R., Moses, L. M., Sutherland, D. B., Young, S. L, Johnson, D. N., Kinnier, W. B., and Kilpatrick, B. F., 2-Phenyl-3H-imidazo [4, 5-b] pyridine-3- acetamides as nonbenzodiazepine anticonvulsants and anxiolytics. J. Med. Chem., 34, 2993–3006 (1991).
White, D. C., Greenwood, T. D., Downey, A. L., Bloomquistc, J. R., and Wolfea, J. F., Synthesis and anticonvulsant evaluation of some new 2-substituted-3-arylpyrido [2,3-d] pyrimidinones. Bioorg. Med. Chem., 12, 5711–5717 (2004).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jain, P., Sharma, P.K., Rajak, H. et al. Design, synthesis and biological evaluation of some novel benzimidazole derivatives for their potential anticonvulsant activity. Arch. Pharm. Res. 33, 971–980 (2010). https://doi.org/10.1007/s12272-010-0701-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-010-0701-8