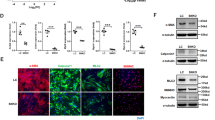
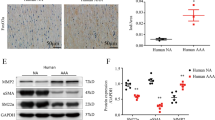
Abstract
Abdominal aortic aneurysm (AAA) is a fatal vascular disease. Vascular smooth muscle cells (VSMCs) play a crucial role in the pathogenesis of AAA. Increasing evidence has shown that Yes-associated protein (YAP) is involved in diverse vascular diseases. However, the role of YAP in AAA remains unclear. The current study aimed to determine the role of YAP in AAA formation and the underlying mechanism. We found that YAP expression in VSMCs was markedly decreased in human and experimental AAA samples. Furthermore, VSMC-specific YAP overexpression prevented several pathogenic factor–induced AAA. Mechanistically, YAP overexpression in VSMCs promoted latent transforming growth factor-β binding protein 4 (LTBP4) expression, an important factor in elastic fiber assembly. Finally, silencing of LTBP4 in VSMCs abolished the protective role of YAP in AAA formation in vivo. Our results suggest that YAP promotes LTBP4-mediated elastic fibril assembly in VSMCs, which mitigates elastin degradation and AAA formation.
Graphical abstract







Similar content being viewed by others
Abbreviations
- AAA:
-
Abdominal aortic aneurysm
- ANOVA:
-
Analysis of variance
- ECM:
-
Extracellular matrix
- EVG:
-
Elastica van Gieson
- H&E:
-
Hematoxylin and eosin
- HVSMCs:
-
Human vascular smooth muscle cell
- LTBP4:
-
Latent transforming growth factor-β binding protein 4
- PBS:
-
Phosphate-buffered saline
- SMC:
-
Smooth muscle cell
- TGF-β:
-
Transforming growth factor-β
- VSMC:
-
Vascular smooth muscle cell
- YAP:
-
Yes-associated protein
References
Kent, K. C. (2014). Clinical practice Abdominal aortic aneurysms. New England Journal of Medicine, 371(22), 2101–2108. https://doi.org/10.1056/NEJMcp1401430https://www.ncbi.nlm.nih.gov/pubmed/25427112.
Erbel, R., Aboyans, V., Boileau, C., Bossone, E., Bartolomeo, R. D., Eggebrecht, H., et al. (2014). ESC Guidelines on the diagnosis and treatment of aortic diseases: Document covering acute and chronic aortic diseases of the thoracic and abdominal aorta of the adult. The Task Force for the Diagnosis and Treatment of Aortic Diseases of the European Society of Cardiology (ESC). European Heart Journal, 35(41), 2873–2926. https://doi.org/10.1093/eurheartj/ehu281https://www.ncbi.nlm.nih.gov/pubmed/25173340.
Hu, J., Xu, J., Shen, S., Zhang, W., Chen, H., Sun, X., et al. (2022). Trimethylamine N-oxide promotes abdominal aortic aneurysm formation by aggravating aortic smooth muscle cell senescence in mice. Journal of Cardiovascular Translational Research. https://doi.org/10.1007/s12265-022-10211-6https://www.ncbi.nlm.nih.gov/pubmed/35143032.
Xu, J., & Shi, G. P. (2014). Vascular wall extracellular matrix proteins and vascular diseases. Biochimica et Biophysica Acta, 1842(11), 2106–2119. https://doi.org/10.1016/j.bbadis.2014.07.008https://www.ncbi.nlm.nih.gov/pubmed/25045854.
Schmelzer, C. E. H., & Duca, L. (2021). Elastic fibers: Formation, function, and fate during aging and disease. FEBS Journal. https://doi.org/10.1111/febs.15899https://www.ncbi.nlm.nih.gov/pubmed/33896108.
Midwood, K. S., & Schwarzbauer, J. E. (2002). Elastic fibers: Building bridges between cells and their matrix. Current Biology, 12(8), R279–R281. https://doi.org/10.1016/s0960-9822(02)00800-xhttps://www.ncbi.nlm.nih.gov/pubmed/11967167.
Lemaire, R., Bayle, J., Mecham, R. P., & Lafyatis, R. (2007). Microfibril-associated MAGP-2 stimulates elastic fiber assembly. Journal of Biology Chemistry, 282(1), 800–808. https://doi.org/10.1074/jbc.M609692200https://www.ncbi.nlm.nih.gov/pubmed/17099216.
Yanagisawa, H, Wagenseil, J (2020). Elastic fibers and biomechanics of the aorta: Insights from mouse studies. Matrix Biology, 85-86, 160-172. https://www.ncbi.nlm.nih.gov/pubmed/30880160https://doi.org/10.1016/j.matbio.2019.03.001
Noda, K., Dabovic, B., Takagi, K., Inoue, T., Horiguchi, M., Hirai, M., et al. (2013). Latent TGF-beta binding protein 4 promotes elastic fiber assembly by interacting with fibulin-5. Proceedings of the National Academy of Sciences of the United States of America, 110(8), 2852–2857. https://doi.org/10.1073/pnas.1215779110https://www.ncbi.nlm.nih.gov/pubmed/23382201.
Tai, H. C., Tsai, P. J., Chen, J. Y., Lai, C. H., Wang, K. C., Teng, S. H., et al. (2016). Peroxisome proliferator-activated receptor gamma level contributes to structural integrity and component production of elastic fibers in the aorta. Hypertension, 67(6), 1298–1308. https://doi.org/10.1161/HYPERTENSIONAHA.116.07367https://www.ncbi.nlm.nih.gov/pubmed/27045031.
Nosoudi, N., Chowdhury, A., Siclari, S., Parasaram, V., Karamched, S., & Vyavahare, N. (2016). Systemic delivery of nanoparticles loaded with pentagalloyl glucose protects elastic lamina and prevents abdominal aortic aneurysm in rats. Journal of Cardiovascular Translational Research, 9(5–6), 445–455. https://doi.org/10.1007/s12265-016-9709-xhttps://www.ncbi.nlm.nih.gov/pubmed/27542007.
Sterner-Kock, A., Thorey, I. S., Koli, K., Wempe, F., Otte, J., Bangsow, T., et al. (2002). Disruption of the gene encoding the latent transforming growth factor-beta binding protein 4 (LTBP-4) causes abnormal lung development, cardiomyopathy, and colorectal cancer. Genes & Development, 16(17), 2264–2273. https://doi.org/10.1101/gad.229102https://www.ncbi.nlm.nih.gov/pubmed/12208849.
Callewaert, B., Su, C. T., Van Damme, T., Vlummens, P., Malfait, F., Vanakker, O., et al. (2013). Comprehensive clinical and molecular analysis of 12 families with type 1 recessive cutis laxa. Human Mutation, 34(1), 111–21. https://doi.org/10.1002/humu.22165https://www.ncbi.nlm.nih.gov/pubmed/22829427.
Urban, Z., Hucthagowder, V., Schurmann, N., Todorovic, V., Zilberberg, L., Choi, J., et al. (2009). Mutations in LTBP4 cause a syndrome of impaired pulmonary, gastrointestinal, genitourinary, musculoskeletal, and dermal development. American Journal of Human Genetics, 85(5), 593–60. https://doi.org/10.1016/j.ajhg.2009.09.013https://www.ncbi.nlm.nih.gov/pubmed/19836010.
Thompson, A. R., Cooper, J. A., Jones, G. T., Drenos, F., van Bockxmeer, F. M., Biros, E., et al. (2010). Assessment of the association between genetic polymorphisms in transforming growth factor beta, and its binding protein (LTBP), and the presence, and expansion, of Abdominal Aortic Aneurysm. Atherosclerosis, 209(2), 367–73. https://doi.org/10.1016/j.atherosclerosis.2009.09.073https://www.ncbi.nlm.nih.gov/pubmed/19897194.
Yu, Y., Su, X., Qin, Q., Hou, Y., Zhang, X., Zhang, H., et al. (2020). Yes-associated protein and transcriptional coactivator with PDZ-binding motif as new targets in cardiovascular diseases. Pharmacological Research, 159, 105009. https://doi.org/10.1016/j.phrs.2020.105009https://www.ncbi.nlm.nih.gov/pubmed/32553712.
Li, H., Jiang, W., Ren, W., Guo, D., Guo, J., Wang, X., et al. (2016). Downregulation of the Yes-associated protein is associated with extracellular matrix disorders in ascending aortic aneurysms. Stem Cells International, 2016, 6786184. https://doi.org/10.1155/2016/6786184https://www.ncbi.nlm.nih.gov/pubmed/26904131.
Jiang, W. J., Ren, W. H., Liu, X. J., Liu, Y., Wu, F. J., Sun, L. Z., et al. (2016). Disruption of mechanical stress in extracellular matrix is related to Stanford type A aortic dissection through down-regulation of Yes-associated protein. Aging (Albany NY), 8(9), 1923–1939. https://doi.org/10.18632/aging.101033https://www.ncbi.nlm.nih.gov/pubmed/27608489.
Marti, P., Stein, C., Blumer, T., Abraham, Y., Dill, M. T., Pikiolek, M., et al. (2015). YAP promotes proliferation, chemoresistance, and angiogenesis in human cholangiocarcinoma through TEAD transcription factors. Hepatology, 62(5), 1497–510. https://doi.org/10.1002/hep.27992https://www.ncbi.nlm.nih.gov/pubmed/26173433.
Kim, H. W., & Weintraub, N. L. (2016). Aortic aneurysm: In defense of the vascular smooth muscle cell. Arteriosclerosis Thrombosis Vascular Biology, 36(11), 2138–2140. https://doi.org/10.1161/ATVBAHA.116.308356https://www.ncbi.nlm.nih.gov/pubmed/27784700.
Dabovic, B., Chen, Y., Choi, J., Vassallo, M., Dietz, H. C., Ramirez, F., et al. (2009). Dual functions for LTBP in lung development: LTBP-4 independently modulates elastogenesis and TGF-beta activity. Journal of Cellular Physiology, 219(1), 14–22. https://doi.org/10.1002/jcp.21643https://www.ncbi.nlm.nih.gov/pubmed/19016471.
Chen, H. Z., Wang, F., Gao, P., Pei, J. F., Liu, Y., Xu, T. T., et al. (2016). Age-associated sirtuin 1 reduction in vascular smooth muscle links vascular senescence and inflammation to abdominal aortic aneurysm. Circulation Research, 119(10), 1076–1088. https://doi.org/10.1161/CIRCRESAHA.116.308895https://www.ncbi.nlm.nih.gov/pubmed/27650558.
Panciera, T., Azzolin, L., Cordenonsi, M., & Piccolo, S. (2017). Mechanobiology of YAP and TAZ in physiology and disease. Nature Reviews Molecular Cell Biology, 18(12), 758–770. https://doi.org/10.1038/nrm.2017.87https://www.ncbi.nlm.nih.gov/pubmed/28951564.
Seo, J., & Kim, J. (2018). Regulation of Hippo signaling by actin remodeling. BMB Reports, 51(3), 151–156. https://doi.org/10.5483/bmbrep.2018.51.3.012https://www.ncbi.nlm.nih.gov/pubmed/29353600.
Yamashiro, Y., Thang, B. Q., Ramirez, K., Shin, S. J., Kohata, T., Ohata, S., et al. (2020). Matrix mechanotransduction mediated by thrombospondin-1/integrin/YAP in the vascular remodeling. Proceedings of the National Academy of Sciences of the United States of America, 117(18), 9896–9905. https://doi.org/10.1073/pnas.1919702117https://www.ncbi.nlm.nih.gov/pubmed/32321834.
Albig, A. R., Becenti, D. J., Roy, T. G., & Schiemann, W. P. (2008). Microfibril-associate glycoprotein-2 (MAGP-2) promotes angiogenic cell sprouting by blocking notch signaling in endothelial cells. Microvascular Research, 76(1), 7–14. https://doi.org/10.1016/j.mvr.2008.01.001https://www.ncbi.nlm.nih.gov/pubmed/18417156.
Sun, T., Peng, H., Mao, W., Ma, L., Liu, H., Mai, J., et al. (2021). Autophagy-mediated negative feedback attenuates the oncogenic activity of YAP in pancreatic cancer. International Journal of Biological Sciences, 17(13), 3634–3645. https://doi.org/10.7150/ijbs.61795https://www.ncbi.nlm.nih.gov/pubmed/34512171.
Kang, W., Huang, T., Zhou, Y., Zhang, J., Lung, R. W. M., Tong, J. H. M., et al. (2018). miR-375 is involved in Hippo pathway by targeting YAP1/TEAD4-CTGF axis in gastric carcinogenesis. Cell Death and Disease, 9(2), 92. https://doi.org/10.1038/s41419-017-0134-0https://www.ncbi.nlm.nih.gov/pubmed/29367737.
Zhou, X., Li, Y., Wang, W., Wang, S., Hou, J., Zhang, A., et al. (2020). Regulation of Hippo/YAP signaling and Esophageal Squamous Carcinoma progression by an E3 ubiquitin ligase PARK2. Theranostics, 10(21), 9443–9457. https://doi.org/10.7150/thno.46078https://www.ncbi.nlm.nih.gov/pubmed/32863938.
Luo, Y., Zhou, J., Tang, J., Zhou, F., He, Z., Liu, T., et al. (2021). MINDY1 promotes bladder cancer progression by stabilizing YAP. Cancer Cell International, 21(1), 395. https://doi.org/10.1186/s12935-021-02095-4https://www.ncbi.nlm.nih.gov/pubmed/34315490.
Zhang, H., Pasolli, H. A., & Fuchs, E. (2011). Yes-associated protein (YAP) transcriptional coactivator functions in balancing growth and differentiation in skin. Proceedings of the National Academy of Sciences of the United States of America, 108(6), 2270–5. https://doi.org/10.1073/pnas.1019603108https://www.ncbi.nlm.nih.gov/pubmed/21262812.
Stein, C., Bardet, A. F., Roma, G., Bergling, S., Clay, I., Ruchti, A., et al. (2015). YAP1 exerts its transcriptional control via TEAD-mediated activation of enhancers. PLoS Genetics, 11(8), e1005465. https://doi.org/10.1371/journal.pgen.1005465https://www.ncbi.nlm.nih.gov/pubmed/26295846.
Bultmann, I., Conradi, A., Kretschmer, C., & Sterner-Kock, A. (2013). Latent transforming growth factor beta-binding protein 4 is downregulated in esophageal cancer via promoter methylation. PLoS One, 8(5), e65614. https://doi.org/10.1371/journal.pone.0065614https://www.ncbi.nlm.nih.gov/pubmed/23741501.
Wang, Y., Hu, G., Liu, F., Wang, X., Wu, M., Schwarz, J. J., et al. (2014). Deletion of yes-associated protein (YAP) specifically in cardiac and vascular smooth muscle cells reveals a crucial role for YAP in mouse cardiovascular development. Circulation Research, 114(6), 957–65. https://doi.org/10.1161/CIRCRESAHA.114.303411https://www.ncbi.nlm.nih.gov/pubmed/24478334.
Rowe, V. L., Stevens, S. L., Reddick, T. T., Freeman, M. B., Donnell, R., Carroll, R. C., et al. (2000). Vascular smooth muscle cell apoptosis in aneurysmal, occlusive, and normal human aortas. Journal of Vascular Surgery, 31(3), 567–76. https://www.ncbi.nlm.nih.gov/pubmed/10709071.
Wang, X., Liang, X., Zhou, L., Liu, S., Fang, Z., Hu, C., et al. (2022). Yes-associated protein reacts differently in vascular smooth muscle cells under different intensities of mechanical stretch. Aging (Albany NY), 14(1), 286–296. https://doi.org/10.18632/aging.203768https://www.ncbi.nlm.nih.gov/pubmed/34983026.
Rifkin DB, Rifkin WJ, Zilberberg L. (2018). LTBPs in biology and medicine: LTBP diseases. Matrix Biology, 71-72, 90-99. https://www.ncbi.nlm.nih.gov/pubmed/29217273https://doi.org/10.1016/j.matbio.2017.11.014
Tomasovic, A., Kurrle, N., Wempe, F., De-Zolt, S., Scheibe, S., Koli, K., et al. (2017). Ltbp4 regulates Pdgfrbeta expression via TGFbeta-dependent modulation of Nrf2 transcription factor function. Matrix Biology, 59, 109–120. https://doi.org/10.1016/j.matbio.2016.09.006https://www.ncbi.nlm.nih.gov/pubmed/27645114.
Heydemann, A., Ceco, E., Lim, J. E., Hadhazy, M., Ryder, P., Moran, J. L., et al. (2009). Latent TGF-beta-binding protein 4 modifies muscular dystrophy in mice. Journal of Clinical Investigation, 119(12), 3703–12. https://doi.org/10.1172/JCI39845https://www.ncbi.nlm.nih.gov/pubmed/19884661.
Lareyre, F., Clement, M., Raffort, J., Pohlod, S., Patel, M., Esposito, B., et al. (2017). TGFbeta (transforming growth factor-beta) blockade induces a human-like disease in a nondissecting mouse model of abdominal aortic aneurysm. Arteriosclerosis Thrombosis Vascular Biology, 37(11), 2171–2181. https://doi.org/10.1161/ATVBAHA.117.309999https://www.ncbi.nlm.nih.gov/pubmed/28912363.
Angelov, S. N., Hu, J. H., Wei, H., Airhart, N., Shi, M., & Dichek, D. A. (2017). TGF-beta (transforming growth factor-beta) signaling protects the thoracic and abdominal aorta from angiotensin II-induced pathology by distinct mechanisms. Arteriosclerosis Thrombosis Vascular Biology, 37(11), 2102–2113. https://doi.org/10.1161/ATVBAHA.117.309401https://www.ncbi.nlm.nih.gov/pubmed/28729364.
Wang, Y., Ait-Oufella, H., Herbin, O., Bonnin, P., Ramkhelawon, B., Taleb, S., et al. (2010). TGF-beta activity protects against inflammatory aortic aneurysm progression and complications in angiotensin II-infused mice. Journal of Clinical Investigation, 120(2), 422–32. https://doi.org/10.1172/JCI38136https://www.ncbi.nlm.nih.gov/pubmed/20101093.
Hu, J. H., Wei, H., Jaffe, M., Airhart, N., Du, L., Angelov, S. N., et al. (2015). Postnatal deletion of the type II transforming growth factor-beta receptor in smooth muscle cells causes severe aortopathy in mice. Arteriosclerosis Thrombosis and Vascular Biology, 35(12), 2647–56. https://doi.org/10.1161/ATVBAHA.115.306573https://www.ncbi.nlm.nih.gov/pubmed/26494233.
Acknowledgements
We thank Dr. Yi Fu for his excellent technical assistance.
Funding
This work was supported by the National Natural Science Foundation of China Grants (81970392, 82070305, 82100268), Beijing Natural Science Foundation of China (J190010), and Tianjin Natural Science Foundation of China (19JCQNJC10100).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Gang Liu, Liu Yao, and Hong-feng Jiang. Material contribution: Meng Yan. Data collection and analysis were performed by Ya-nan Liu, Xue Lv, and Xin Chen. The first draft of the manuscript was written by Ya-nan Liu, Liu Yao, and Hong-feng Jiang. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Statement
The animal study was reviewed and approved by the Committee of Tianjin Medical University on Animal Experimentation (TMUaMEC2021014). All protocols were approved by the First Affiliated Hospital of Soochow University Ethics Committee.
Statement of the Clinical Relevance
AAA is highly lethal disease in humans. This study suggests that early boosting of YAP-LTBP4 pathway in patients with AAA might be an effective strategy to protect against lethality by reducing the risk of aneurysm rupture.
Conflict of Interest
The authors declare no competing interests.
Additional information
Associate Editor Junjie Xiao oversaw the review of this article .
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Liu, Yn., Lv, X., Chen, X. et al. Specific Overexpression of YAP in Vascular Smooth Muscle Attenuated Abdominal Aortic Aneurysm Formation by Activating Elastic Fiber Assembly via LTBP4. J. of Cardiovasc. Trans. Res. 16, 65–76 (2023). https://doi.org/10.1007/s12265-022-10278-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12265-022-10278-1