Abstract
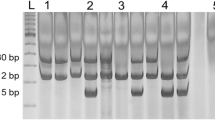
Breast cancer is the most common cause of cancer death in women with the incidence rising in young women. GST gene polymorphisms are significant because of their role in the detoxification of both environmental carcinogens and also cytotoxic drugs used in therapy for breast cancer. The present study has been designed to identify the role of polymorphisms in GSTT1 and GSTM1 genes in the risk of development of breast cancer, in the prognostication of breast cancer, and in the prediction of response towards chemotherapy. Ninety-nine patients with breast cancer and 100 healthy controls with no history of cancer were taken from blood donors after informed consent. Epidemiological and clinical data was collected from participants and 5 ml of peripheral venous blood was collected for genotype analysis. Null genotype of GSTT1 was detected in 51.04 % of the controls in comparison to 20.2 % of patients with carcinoma breast, which was found to be statistically significant (OR 4.18; 95 % CI 2.01–8.75; P = 0.0001). GSTM1 gene deletion was also significantly more common among controls (60 %) than in patients with breast cancer (33 %) (OR 4.57; 95 % CI 2.20–9.51; P = 0.0001). Tumors more than 5 cm in size had greater tendency for GSTM1 gene expression (P value = 0.019), but other clinicopathological parameters did not show any correlation. GSTT1 and GSTM1 genes status did not show any association with response to chemotherapy. The results indicated the null genotype of both GSTT1 and GSTM1 to be protective for the development of carcinoma breast. None of the known etiological factors have any correlation with GSTT1 and GSTM1 gene deletion. Patients with small tumor size expressed GSTM1 gene deletion. Other tumor characteristics and clinicopathological parameters did not have any correlation with gene deletion.
Similar content being viewed by others
References
Mitrunen K, Jourenkova N, Kataja V, Eskelinen M, Kosma V-M, Benhamou S et al (2001) Glutathione S-transferase M1, M3, P1, and T1 genetic polymorphisms and susceptibility to breast cancer. Cancer Epidemiol Biomark Prev 10(3):229–236
Lichtenstein P, Holm NV, Verkasalo PK, Iliadou A, Kaprio J, Koskenvuo M et al (2000) Environmental and heritable factors in the causation of cancer— analyses of cohorts of twins from Sweden, Denmark, and Finland. N Engl J Med 343(2):78–85
Easton D, Ford D, Peto J (1993) Inherited susceptibility to breast cancer. Cancer Surv 18:95–113
Oesterreich S, Fuqua SA (1999) Tumor suppressor genes in breast cancer. Endocr Relat Cancer 6(3):405–419
Ross RK, Jones PA, Yu MC (1996) Bladder cancer epidemiology and pathogenesis. Semin Oncol 23(5):536–545
Li D, Wang M, Dhingra K, Hittelman WN (1996) Aromatic DNA adducts in adjacent tissues of breast cancer patients: clues to breast cancer etiology. Cancer Res 56(2):287–293
Johnson-Thompson MC, Guthrie J (2000) Ongoing research to identify environmental risk factors in breast carcinoma. Cancer 88:1224–1229
Chen XX, Zhao RP, Qiu LX, Yuan H, Mao C, Hu XC, Guo XM (2011) Glutathione S-transferase T1 polymorphism is associated with breast cancer susceptibility. Cytokine 56(2):477–480
Townsend DM, Tew KD (2003) The role of glutathione-S-transferase in anti-cancer drug resistance. Oncogene. 22(47):7369–75
Khedhaier A, Remadi S, Corbex M, Ahmed SB, Bouaouina N, Mestiri S, Azaiez R, Helal AN, Chouchane L (2003) Glutathione S-transferases (GSTT1 and GSTM1) gene deletions in Tunisians: susceptibility and prognostic implications in breast carcinoma. Br J Cancer 89(8):1502–1507
Xu S, Wang Y, Roe B, Pearson WR (1998) Characterization of the human class Mu glutathione S-transferase gene cluster and the GSTM1 deletion. J Biol Chem 273(6):3517–3527
La Torre F, Orlando A, Silipigni A, Giacobello T, Pergolizzi S, Aragona M (1997) Increase of oxygen free radicals and their derivatives in chemo- and radiation treated neoplasm patients. Minerva Med 88(4):121–126
Oliveira AL, Rodrigues FF, Santos RE, Aoki T, Rocha MN, Longui CA, Melo MB (2010) GSTT1, GSTM1, and GSTP1 polymorphisms and chemotherapy response in locally advanced breast cancer. Genet Mol Res 9(2):1045–1053
Helzlsouer KJ, Selmin O, Huang HY, Strickland PT, Hoffman S, Alberg AJ, Watson M, Comstock GW, Bell D (1998) Association between glutathione S-transferase M1, P1, and T1 genetic polymorphisms and development of breast cancer. J Natl Cancer Inst 90(7):512–518
Bailey LR, Roodi N, Verrier CS, Yee CJ, Dupont WD, Parl FF (1998) Breast cancer and CYPIA1, GSTM1, and GSTT1 polymorphisms: evidence of a lack of association in Caucasians and African Americans. Cancer Res 58(1):65–70
Ketterer B (1988) Protective role of glutathione and glutathione transferases in mutagenesis and carcinogenesis. Mutat Res 202(2):343–361
Hellman S (1993) Principles of radiation therapy. In: DeVita VT, Hellman S, Rosenberg SA (eds) Cancer: principles and practice of oncology, 4th edn. Lippincott Co., Philadelphia, pp 248–275
Faber M, Coudray C, Hida H, Mousseau M, Favier A (1995) Lipid peroxidation products, and vitamin and trace element status in patients with cancer before and after chemotherapy, including adriamycin. A preliminary study. Biol Trace Elem Res 47(1–3):117–123
Sulkowska M, Sulkowski S, Skrzydlewska E, Farbiszewski R (1998) Cyclophosphamide-induced generation of reactive oxygen species. Comparison with morphological changes in type II alveolar epithelial cells and lung capillaries. Exp Toxicol Pathol 50(3):209–220
Ambrosone CB, Freudenheim JL, Graham S, Marshall JR, Vena JE, Brasure JR, Laughlin R, Nemoto T, Michalek AM, Harrington A (1995) Cytochrome P4501A1 and glutathione S-transferase (M1) genetic polymorphisms and postmenopausal breast cancer risk. Cancer Res 55(16):3483–3485
Millikan R, Pittman G, Tse C-K, Savitz DA, Newman B, Bell D (2000) Glutathione S-transferases M1, T1, and P1 and breast cancer. Cancer Epidemiol Biomark Prev 9(6):567–573
García-Closas M, Kelsey KT, Hankinson SE, Spiegelman D, Springer K, Willett WC, Speizer FE, Hunter DJ (1999) Glutathione S-transferase mu and theta polymorphisms and breast cancer susceptibility. J Natl Cancer Inst 91(22):1960–1964
Parl FF (2005) Glutathione S-transferase genotypes and cancer risk. Cancer Lett 221(2):123–129
Charrier J, Maugard CM, Le Mevel B, Bignon YJ (1999) Allelotype influence at glutathione S-transferase M1 locus on breast cancer susceptibility. Br J Cancer 79(2):346–353
Siegelmann-Danieli N, Buetow KH (2002) Significance of genetic variation at the glutathione S-transferase M1 and NAD(P)H:quinone oxidoreductase 1 detoxification genes in breast cancer development. Oncology 62(1):39–45
Lallas TA, McClain SK, Shahin MS, Buller RE (2000) The glutathione S-transferase M1 genotype in ovarian cancer. Cancer Epidemiol Biomarkers Prev 9(6):587–590
Howells RE, Redman CW, Dhar KK, Sarhanis P, Musgrove C, Jones PW, Alldersea J, Fryer AA, Hoban PR, Strange RC (1998) Association of glutathione S-transferase GSTM1 and GSTT1 null genotypes with clinical outcome in epithelial ovarian cancer. Clin Cancer Res 4(10):2439–2445
Berrigan MJ, Struck RF, Gurtoo HL (1987) Lipid peroxidation induced by cyclophosphamide. Cancer Biochem Biophys 9(3):265–270
Acknowledgments
The study was conducted with the help of Institute Research Grant, from the All India Institute of Medical Sciences, New Delhi.
Conflict of Interest
Authors Dr Virinder Kumar Bansal, Dr Karthik Rajan, Dr Arundhati Sharma, Dr Gaurav Charbal, Dr Vikas Jindal, Dr Mahesh C Misra, and Dr Kiran Kucheria have no competing conflict of interests or financial disclosures to make.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bansal, V.K., Rajan, K., Sharma, A. et al. Prospective Case–Control Study to Evaluate the Role of Glutathione S Transferases (GSTT1 and GSTM1) Gene Deletion in Breast Carcinoma and Its Prognostic Significance. Indian J Surg 77 (Suppl 3), 1067–1072 (2015). https://doi.org/10.1007/s12262-014-1152-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-014-1152-0