Abstract
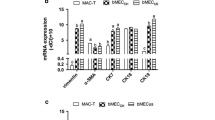
Mammary gland development is critically dependent on the interactions between its stromal and epithelial compartments. In this study, we established a co-culture of bovine mammary epithelial Mac-T cells and murine preadipocyte 3T3-L1 cells. Mac-T cells were co-cultured with 3T3-L1 cells for four days and production of milk proteins was induced for three days. After seven days of co-culturing, the number of alveolar-like colonies in the presence of 3T3-L1 cells was significantly higher than that in control group. Expression levels of αS1-casein and β-casein mRNAs were significantlyincreased by co-culturing with 3T3-L1 cells. In addition, casein protein production was significantly higher in the co-culture of Mac-T cells with 3T3-L1 cells than in control Mac-T cells. Substances that induced casein production in Mac-T cells also stimulated adipogenesis in 3T3-L1 cells. We suggest that a co-culture system of bovine mammary epithelial cells and preadipocyte cells is an efficient method for in vitro milk protein production.
Similar content being viewed by others
References
Hovey, R. C., J. F. Trott, and B. K. Vonderhaar (2002) Establishing a framework for the functional mammary gland: From endocrinology to morphology. J. Mammary Gland Biol. Neoplasia 7: 17–38.
Neville, M. C., D. Medina, J. Monks, and R. C. Hovey (1998) The mammary fat pad. J. Mammary Gland Biol. Neoplasia 3: 109–116.
Schmid, E., W. W. Franke, C. Grund, D. L. Schiller, H. Kolb, and N. Paweletz (1983) An epithelial cell line with elongated myoid morphology derived from bovine mammary gland. Expression of cytokeratins and desmosomal plaque proteins in unusual arrays. Exp. Cell Res. 146: 309–328.
Huynh, H. and M. Pollak (1995) HH2A, an immortalized bovine mammary epithelial cell line, expresses the gene encoding mammary derived growth inhibitor (MDGI). In Vitro Cell Dev. Biol. Anim. 31: 25–29.
Zavizion, B., M. van Duffelen, W. Schaeffer, and I. Politis (1996) Establishment and characterization of a bovine mammary myoepithelial cell line. In Vitro Cell Dev. Biol. Anim. 32: 149–158.
Huynh, H. T., G. Robitaille, and J. D. Turner (1991) Establishment of bovine mammary epithelial cells (MAC-T): An in vitro model for bovine lactation. Exp. Cell Res. 197: 191–199.
Johnson, T. L., B. A. Fujimoto, R. Jiménez-Flores, and D. G. Peterson (2010) Growth hormone alters lipid composition and increases the abundance of casein and lactalbumin mRNA in the MAC-T cell line. J. Dairy Res. 77: 199–204.
Kadegowda, A. K., M. Bionaz, L. S. Piperova, R. A. Erdman, and J. J. Loor (2009) Peroxisome proliferator-activated receptorgamma activation and long-chain fatty acids alter lipogenic gene networks in bovine mammary epithelial cells to various extents. J. Dairy Sci. 92: 4276–4289.
Appuhamy, J. A., N. A. Knoebel, W. A. Nayananjalie, J. Escobar, and M. D. HanIgAn (2012) Isoleucine and leucine independently regulate mTOR signaling and protein synthesis in MAC-T cells and bovine mammary tissue slices. J. Nutr. 142: 484–491.
Green, H. and O. Kehinde (1974) Sublines of mouse 3T3 cells that accumulate lipid. Cell 1: 113–116.
Ginger, M. R. and M. R. Grigor (1999) Comparative aspects of milk caseins. Comp. Biochem. Physiol. B. Biochem. Mol. Biol. 124: 133–145.
Lee, H. Y., Y. T. Heo, S. E. Lee, K. C. Hwang, H. G. Lee, S. H. Choi, and N. H. Kim (2013) Short communication: Retinoic acid plus prolactin to synergistically increase specific casein gene expression in MAC-T cells. J. Dairy Sci. 96: 3835–3839.
Monzani, P. S., S. Guemra, P. R. Adona, O. M. Ohashi, F. V. Meirelles, and M. B. Wheeler (2015) MAC-T cells as a tool to evaluate lentiviral vector construction targeting recombinant protein expression in milk. Anim. Biotechnol. 26: 136–142.
Peterson, D. G., E. A. Matitashvili, and D. E. Bauman (2004) The inhibitory effect of trans-10, cis-12 CLA on lipid synthesis in bovine mammary epithelial cells involves reduced proteolytic activation of the transcription factor SREBP-1. J. Nutr. 134: 2523–2527.
Heo, Y. T., W. T. Ha, R. Lee, W. Y. Lee, H. Y. Jeong, K. C. Hwang, and H. Song (2017) MAC-T cell as in vitro evaluation system for casein gene expression involved in glucose level. Asian-Australas. J. Anim. Sci. 30: 878–885.
Levine, J. F. and F. E. Stockdale (1984) 3T3-L1 adipocytes promote the growth of mammary epithelium. Exp. Cell Res. 151: 112–122.
Kumar, S. and G. Duester (2011) SnapShot: retinoic acid signaling. Cell 147: 1422–1422.
Kim, D. M. H. R. Choi, A. Park, S. M. Shin, K. H. Bae, S. Lee, I. C. Kim, and W. K. Kim (2013) Retinoic acid inhibits adipogenesis via activation of Wnt signaling pathway in 3T3-L1 preadipocytes. Biochem. Biophys. Res. Commun. 434: 455–459.
Reed, B. C., S. H. Kaufmann, J. C. Mackall, A. K. Student, and M. D. Lane (1977) Alterations in insulin binding accompanying differentiation of 3T3-L1 preadipocytes. Proc. Natl. Acad. Sci. U S A. 74: 4876–4880.
McAveney, K. M., J. M. Gimble, and L. Yu-Lee (1996) Prolactin receptor expression during adipocyte differentiation of bone marrow stroma. Endocrinol. 137: 5723–5726.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, WY., Park, HJ., Yeo, J.M. et al. Enhancement of milk protein expression in mammary epithelial cells via co-culturing with preadipocyte cells. Biotechnol Bioproc E 22, 556–560 (2017). https://doi.org/10.1007/s12257-017-0264-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-017-0264-3