Abstract
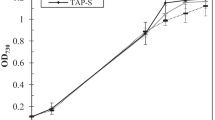
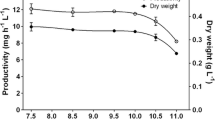
To study the effect of culture medium on hydrogen production by the marine green algae, Platymonas subcordiformis under sulfur deprivation, cell growth, hydrogen production, and starch and protein catabolism was investigated in the work. Algae cells cultured only in optimized medium required 6∼8 days to reach the late logarithmic at the approximate density of (2.00 ± 0.18) × 106 cells/mL, which in traditional medium needed 18∼22 days to reach (1.85 ± 0.20) × 106 cells/mL. Increased levels of Chlorophyll (10.74 ± 0.20 μg/mL), starch (149.50 ± 6.15 μg/mL), and protein (213.00 ± 7.36 μg/mL) were accumulated in optimized medium, which were 1.06, 1.47, and 1.87-fold of the algae cells cultured in traditional medium, respectively. The sealed culture of algae cells in sulfur-deprived optimized medium shifted to anaerobic conditions after 96 h of light illumination and produced 0.45 ± 0.12 mL H2, but in traditional medium maintained aerobic condition and no hydrogen was produced. In addition, changes in starch and protein content during continuous light illumination indicated that more endogenous substrate was consumed in the sulfur-deprived optimized medium than that in the sulfur-deprived traditional medium.
Similar content being viewed by others
References
Gaffron, H. (1939) Reduction of CO2 with H2 in green algae. Nature 143: 204–205.
Gaffron, H. and J. Rubin (1942) Fermentative and photochemical production of hydrogen in algae. J. Gen. Physiol. 26: 219–240.
Wykoff, D. D., J. P. Davies, A. Melis, and A. R. Grossman (1998) The regulation of photosynthetic electron transport during nutrient deprivation in Chlamydomonas reinhardtii1. Plant Physiol. 117: 129–139.
Melis, A., L. P. Zhang, M. Forestier, M. L. Ghirardi, and M. Seibert (2000) Sustained photobiological hydrogen gas production upon reversible inactivation of oxygen evolution in the green alga Chlamydomonas reinhardtii1. Plant Physiol. 122: 127–136.
Ghirardi, M. L., L. P. Zhang, J. W. Lee, T. Flynn, M. Seibert, E. Greenbaum, and A. Melis (2000) Microalgae: a green source of renewable H2. Trends Biotechnol. 18: 506–511.
Zhang, L. P., T. Happe, and A. Melis (2002) Biochemical and morphological characterization of sulfur-deprived and H2-producing Chlamydomonas reinhardtii (green alga). Planta 214: 552–561.
Kaltwasser, H., T. S. Stuart, and H. Gaffron (1969) Light-dependent hydrogen evolution by Scenedesmus. Planta 89: 309–322.
Melis, A. and T. Happe (2001) Hydrogen production. Green algae as a source of energy. Plant Physiol. 127: 740–748.
Ran, C. Q., X. J. Yu, M. F. Jin, and W. Zhang (2006) Role of carbonyl cyanide m-chlorophenylhydrazone in enhancing photobiological hydrogen production by marine green alga Platymonas subcordiformis. Biotechnol. Prog. 22: 438–443.
Ran, C. Q., W. Zhang, X. J. Yu, M. F. Jin, and M. C. Deng (2006) Regulation of hydrogen production by uncoupler CCCP in green algae Chlamydomonas reinhardtii. Chem. J. Chinese Univer. 27: 62–66.
Ghirardi, M. L., S. Kosourov, A. Tsygankov, and M. Seibert (2000) Two-phase photobiological algal H2-production system. Proceedings of the 2000 DOE Hydrogen Program Review. May 9–11. San Ramon, CA, USA.
Antal, T. K., T. E. Krendeleva, T. V. Laurinavichene, V. V. Makarova, A. A. Tsygankov, M. Seibert, and A. B. Rubin (2001) The relationship between the photosystem 2 activity and hydrogen production in sulfur deprived Chlamydomonas reinhardtii cells. Dokl. Biochem. Biophys. 381: 371–374.
Kosourov, S., M. Seibert, and M. L. Ghirardi (2003) Effects of extracellular pH on the metabolic pathways in sulfur-deprived, H2-producing Chlamydomonas reinhardtii cultures. Plant Cell Physiol. 44: 146–155.
Skjånes, K., G. Knutsen, T. Källqvist, and P. Lindblad (2008) H2 production from marine and freshwater species of green algae during sulfur deprivation and considerations for bioreactor design. Int. J. Hydrogen Energy 32: 511–521.
Winkler, M., A. Hemschemeier, C. Gotor, A. Melis, and T. Happe (2002) [Fe]-Hydrogenases in green algae: photo-fermentation and hydrogen evolution under sulfur deprivation. Int. J. Hydrogen Energy 27: 1431–1439.
Guo, Z., Z. A. Chen, W. Zhang, X. J. Yu, and M. F. Jin (2008) Improved hydrogen photoproduction regulated by carbonylcyanide m-chlorophenylhrazone from marine green alga Platymonas subcordiformis grown in CO2-supplemented air bubble column bioreactor. Biotechnol. Lett. 30: 877–883.
Guan, Y. F., W. Zhang, M. C. Deng, M. F. Jin, and X. J. Yu (2004) Significant enhancement of photobiological H2 evolution by carbonylcyanide m-chlorophenylhydrazone in the marine green alga Platymonas subcordiformis. Biotechnol. Lett. 26: 1031–1035.
Lowry, O. H., N. J. Rosebrough, A. L. Farr, and R. J. Randall (1951) Protein measurement with the folin phenol reagent. J. Biol. Chem. 193: 265–275.
Gfeller, R. P. and M. Gibbs (1984) Fermentative metabolism of Chlamydomonas reinhardtii: I. Analysis of fermentative products from starch in dark and light. Plant Physiol. 75: 212–218.
Arnon, D. I. (1949) Copper enzymes in isolated chloroplasts: polyphenol oxidase in Beta vulgaris. Plant Physiol. 24: 1–15.
Ran, C. Q. (2006) Comparative characterization of photobiological hydrogen production between marine and freshwater green algae. Ph.D. Thesis. Dalian Institute of Chemical Physics, Chinese Academy of Sciences, China.
Chen, F. and Y. Jiang (1999) Microalgae Biotechnology. pp. 254–259. China light industry Publishers, Beijing, China.
Borowitzka, M. A. and L. J. Borowitzka (1988) Dunaliella. In: M. A. Borowitzka and L. J. Borowitzka (eds.). Microalgal Biotechnology. Cambridge University Press, Cambridge, London, UK.
Zhan jian Fisheries College (1980) Marine Bait Bioculture. China Agriculture Publisher, Beijing, China.
Happe, T., A. Hemschemeier, M. Winkler, and A. Kaminski (2002) Hydrogenases in green algae: do they save the algae’s life and solve our energy problems? Trends Plant Sci. 7: 246–250.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ran, C., Zhang, F., Sun, H. et al. Effect of culture medium on hydrogen production by sulfur-deprived marine green algae Platymonas subcordiformis . Biotechnol Bioproc E 14, 835–841 (2009). https://doi.org/10.1007/s12257-008-0287-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-008-0287-x