Abstract
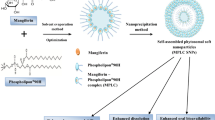
The naturally occurring lipids are non-toxic and biocompatible, hence suitable for pharmaceutical applications. The present study reports the application of mango lipid as a safe pharmaceutical excipient in the formulation of nanostructured lipid carriers (NLCs). The mango lipid was extracted by hot continuous percolation process using a Soxhlet apparatus and analyzed as per USP requirements by TLC and LC-MS. The safety profile of the lipid was studied by acute toxicity study in a rat model as per OECD guidelines. NLC formulation was developed with the physically modified mango lipid as the solid lipid and oleic acid as the liquid lipid by the modified emulsification technique. The compatibility of the lipid with other excipients was studied by differential scanning calorimetry (DSC) and FT-IR analysis. Cytotoxicity of the NLC formulation was studied with mouse brain endothelial cells (bEnd.3) by MTT assay. The TLC and LC-MS studies suggest that the mango lipid contains fatty acids composed of oleic acid (43.8% w/w), linoleic acid (3.6% w/w), stearic acid (43.2% w/w), palmitic acid (4.9% w/w), linolenic acid (2.3% w/w), and arachidic acid (2.2% w/w). The acute toxicity study and MTT assay suggest the safe use of the mango lipid at the cellular level in the living system. The spherical homogeneous nanometric NLCs were developed. DSC and FT-IR study confirm the compatibility of the lipid with the general components of the lipid nanoparticles. This work provides a new dimension to Mangifera indica L. lipid for its safe use in the development of novel lipid nanoformulations for drug delivery.









Similar content being viewed by others
References
Fowomola MA. Some nutrients and antinutrients contents of mango (Mangifera indica, L) seed. African J Food Sci. 2010;4:472–6.
Sriwimon W, Boonsupthip W. Utilization of partially ripe mangoes for freezing preservation by impregnation of mango juice and sugars. Food Sci Technol. 2011;44:375–83.
Ramteke RS, Eipeson WE. Effect of additives on the stability of mango aroma concentrate during storage. J Food Sci Technol. 1997;34:195–9.
Muchiri DR, Mahungu SM, Gituanja SN. Studies on mango (Mangifera indica, L.) kernel fat of some Kenyan varieties in Meru. J Am Oil Chem Soc. 2012;89:1567–75.
Omoregie E. Utilization and nutrient digestibility of mango seeds and palm kernel meal by juvenile Labeo senegalensis (Antheriniformes: Cyprinidae). Aquac Res. 2001;32:681–7.
Kittiphoom S. Utilization of mango seed. Int Food Res J. 2012;19:1325–35.
Puravankara D, Boghra V, Sharma RS. Effect of antioxidant principles isolated from mango (Mangifera indica, L.) seed kernels on oxidative stability of buffalo ghee (butter-fat). J Sci Food Agric. 2000;80:522–6.
Patel S. Potential of fruit and vegetable wastes as novel biosorbents: summarizing the recent studies. Rev Environ Sci Biotechnol. 2012;11:365–80.
Maisuthisakul P, Gordon MH. Antioxidant and tyrosinase inhibitory activity of mango seed kernel by product. Food Chem. 2009;117:332–41.
Bhattacharya K, Shukla VK. Mango butter in cosmetic formulations. Cosmet Toilet. 2002;117:65–70.
Mandawgade SD, Patravale VB. Formulation and evaluation of exotic fat based cosmeceuticals for skin repair. Indian J Pharm Sci. 2008;70:539–42.
Jahurul MHA, Zaidul ISM, Norulaini NAN. Cocoa butter replacers from blends of mango seed fat extracted by supercritical carbon dioxide and palm stearin. Food Res Int. 2014;65:401–6.
Solís-Fuentes JA, Durán-De-Bazúa MC. Mango seed uses: thermal behaviour of mango seed almond fat and its mixtures with cocoa butter. Bioresour Technol. 2004;92:71–8.
Jahurul MHA, Zaidul ISM, Norulaini NAN. Hard cocoa butter replacers from mango seed fat and palm stearin. Food Chem. 2014;154:323–9.
de Peña DG, Anguiano RGL, Arredondo JJM. Modification of the method 1 AOAC (CB-method) for the detection of aflatoxins. Bull Environ Contam Toxicol. 1992;49:485–9.
Hilditch TP. Industrial oil and fat products. USA: Wiley; 1946.
Mahale S, Goswami-Giri A. Composition and characterization of refined oil compared with its crude oil from waste obtained from Mangifera indica. Asia J Res Chem. 2011;4:1415–9.
Daksha A, Jaywant P, Bhagyashree C, Subodh P. Estimation of sterols content in edible oil and ghee samples. Int J Pharm Sci Rev Res. 2010;5:135–7.
Rutkowski M, Grzegorczyk K. Modifications of spectrophotometric methods for antioxidative vitamins determination convenient in analytic practice. Acta Sci Pol. 2007;6:17–28.
Asha V. Phytochemical investigations, extraction and thin layer chromatography of Acorus calamus Linn. Int J Res Stud Biosci. 2015;3:18–22.
Sasidharan S, Chen Y, Saravanan D, Sundram KM, Yoga Latha L. Extraction, isolation and characterization of bioactive compounds from plants’ extracts. African J Tradit Complement Altern Med. 2011;8:1–10.
Nour AH, Elhussein SA, Osman NA. Characterization and chemical composition of the fixed oil of fourteen basil (Ocimum basilicum, L.) accessions grown in Sudan. Int J Chem Technol. 2010;2:113–9.
Mamoci E, Cavoski I, Simeone V, Mondelli D, Al-Bitar L, Caboni P. Chemical composition and in vitro activity of plant extracts from ferula communis and Dittrichia viscosa against postharvest fungi. Molecules. 2011;16:2609–25.
Dubey A, Prabhu P, Kamath JV. Nano structured lipid carriers: a novel topical drug delivery system. Int J PharmTech Res. 2012;4:705–14.
Gaba B, Fazil M, Khan S, Ali A, Baboota S, Ali J. Nanostructured lipid carrier system for topical delivery of terbinafine hydrochloride. Bull Fac Pharmacy. 2015;53:147–59.
Iglesias G, Hlywka JJ, Berg JE, Khalil MH, Pope LE, Tamarkin D. The toxicity of behenyl alcohol I. Genotoxicity and subchronic toxicity in rats and dogs. Regul Toxicol Pharmacol. 2002;36:69–79.
Iglesias G, Hlywka JJ, Berg JE, Khalil MH, Pope LE, Tamarkin D. The toxicity of behenyl alcohol II. Reproduction studies in rats and rabbits. Regulatory Toxicol Pharmacol. 2002;36:80–5.
Farboud ES, Nasrollahi SA, Tabbakhi Z. Novel formulation and evaluation of a Q10-loaded solid lipid nanoparticle cream: in vitro and in vivo studies. Int J Nanomedicine. 2011;6:611–7.
Fan H, Liu G, Huang Y, Li Y, Xia Q. Development of a nanostructured lipid carrier formulation for increasing photo-stability and water solubility of phenylethyl resorcinol. Appl Surf Sci. 2014;288:193–200.
Tamjidi F, Shahedi M, Varshosaz J, Nasirpour A. Design and characterization of astaxanthin-loaded nanostructured lipid carriers. Innov Food Sci Emerg Technol. 2014;26:366–74.
Shete H, Patravale V. Long chain lipid based tamoxifen NLC. Part I: preformulation studies, formulation development and physicochemical characterization. Int J Pharm. 2013;454:573–83.
Tsai MJ, Wu PC, Bin Huang Y, Chang JS. Baicalein loaded in tocol nanostructured lipid carriers (tocol NLCs) for enhanced stability and brain targeting. Int J Pharm. 2012;423:461–70.
Tita B, Ledeti I, Bandur G, Tita D. Compatibility study between indomethacin and excipients in their physical mixtures. J Therm Anal Calorim. 2014;118:1293–304.
Meerloo JV, Kaspers GJL, Cloos J. Cell sensitivity assays: the MTT assay. Methods Mol Biol. 2011;731:237–45.
Angius F, Floris A. Liposomes and MTT cell viability assay: an incompatible affair. Toxicol Vitr. 2015;29:314–9.
Halim SZ, Abdullah NR, Afzan A, Rashid BAA, Jantan I, Ismail Z. Acute toxicity study of Carica papaya leaf extract in Sprague Dawley rats. J Med Plants Res. 2011;5:1867–72.
Verma RK, Garg S. Compatibility studies between isosorbide mononitrate and selected excipients used in the development of extended release formulations. J Pharm Biomed Anal. 2004;35:449–58.
Manikandan M, Kannan K, Manavalan R. Compatibility studies of camptothecin with various pharmaceutical excipients used in the development of nanoparticle formulation. Int J Pharm Sci. 2013;5:315–21.
Tompa AS, Bryant WF. Microcalorimetry and DSC study of the compatibility of energetic materials. Thermochim Acta. 2001;367–368:433–41.
Balestrieri F, Magri AD, Magri AL, Marini D, Sacchini A. Application of differential scanning calorimetry to the study of drug-excipient compatibility. Thermochim Acta. 1996;285:337–45.
Łaszcz M, Kosmacińska B, Korczak K. Study on compatibility of imatinib mesylate with pharmaceutical excipients. J Therm Anal Calorim. 2007;88:305–10.
Mallik S, Kshirsagar MD, Saini V. Studies on physical/chemical compatibility between synthetic and herbal drugs with various pharmaceutical excipients. Der Pharm Lett. 2011;3:173–8.
Zambiazi RC, Przybylski R, Zambiazi MW, Mendonça CB. Fatty acid composition of vegetable oils and fats. Bol do Cent Pesqui e Process Aliment. 2007;25:111–20.
Dixit S, Das M. Fatty acid composition including trans-fatty acids in edible oils and fats: probable intake in Indian population. J Food Sci. 2012;77:10.
Sagiri SS, Sharma V, Basak P, Pal K. Mango butter emulsion gels as cocoa butter equivalents: physical, thermal, and mechanical analyses. J Agric Food Chem. 2014;62:11357–68.
Abdalla AEM, Darwish SM, Ayad EHE, El-Hamahmy RM. Egyptian mango by-product 1. Compositional quality of mango seed kernel. Food Chem. 2007;103:1134–40.
Akhter S, McDonald MA, Marriott R. Mangifera sylvatica (wild mango): a new cocoa butter alternative. Sci Rep. 2016;6:32050.
Abdalla AEM, Darwish SM, Ayad EHE, El-Hamahmy RM. Egyptian mango by-product 2: antioxidant and antimicrobial activities of extract and oil from mango seed kernel. Food Chem. 2007;103:1141–52.
O’Neill CM, Gill S, Hobbs D, Morgan C, Bancroft I. Natural variation for seed oil composition in Arabidopsis thaliana. Phytochemistry. 2003;64:1077–90.
Hwang RJ, Ahmed AS, Moldowan JM. Oil composition variation and reservoir continuity: Unity Field, Sudan. Org Geochem. 1994;21:171–88.
Ali Z, Siddiqui HL, Waheed A, Mahmood S. Distribution of fatty acids in triacylglycerols from ungrafted Desi mango (Mangifera indica) kernel lipids. J Saudi Chem Soc. 2011;15:67–72.
Pena Muniz MA, Ferreira Dos Santos MN, da Costa CE, Morais L, Lamarão ML, Ribeiro-Costa RM, Silva-Júnior JO. Physicochemical characterization, fatty acid composition, and thermal analysis of Bertholletia excelsa HBK oil. Pharmacogn Mag. 2015;11:147–51.
Ibeto CN, Okoye COB, Ofoefule AU. Comparative study of the physicochemical characterization of some oils as potential feedstock for biodiesel production. ISRN Renew Energy. 2012;2012:1–5.
Sidohounde A, Guevara N. Physico-chemical characterization of vegetable oil and defatted meal from two varieties of Cyperus esculestus from Benin. Chem J. 2015;4:1–7.
Sciences C. Physicochemical characterization and frying quality of canola and sunflower oil samples. J Chem Soc Pak. 2012;34:513–7.
Emami J, Rezazadeh M, Varshosaz J, Tabbakhian M, Aslani A. Formulation of LDL targeted nanostructured lipid carriers loaded with paclitaxel: a detailed study of preparation, freeze drying condition, and in vitro cytotoxicity. J Nanomater. 2012;2012:1–10.
Andalib S, Varshosaz J, Hassanzadeh F, Sadeghi H. Optimization of LDL targeted nanostructured lipid carriers of 5-FU by a full factorial design. Adv Biomed Res. 2012;1:45.
Croci T, Landi M, Emonds-Alt X, Le Fur G, Maffrand JP, Manara L. Role of tachykinins in castor oil diarrhoea in rats. Br J Pharmacol. 1997;121:375–80.
Tunaru S, Althoff TF, Nusing RM, Diener M, Offermanns S. Castor oil induces laxation and uterus contraction via ricinoleic acid activating prostaglandin EP3 receptors. Proc Natl Acad Sci U S A. 2012;109:9179–84.
Zhang Y, Li J, Wu Z, Liu E. Acute and long-term toxicity of mango leaves extract in mice and rats, evidence-based complement. Altern Med. 2014;2014:691574.
Shah KA, Patel MB, Patel RJ, Parmar PK. Mongifera indica (Mango). Pharmacogn Rev. 2010;4:42–8.
Newton MJ, Kaur B (2016) Impact of Pluronic F-68 vs Tween 80 on fabrication and evaluation of acyclovir SLNs for skin delivery. Recent Pat Drug Deliv Formul.
Riss TL, Niles AL, Minor L. Cell viability assays assay guidance manual. Assay Guid Man. 2004:1–23.
Acknowledgements
The authors gratefully acknowledge the support of the Department of Biotechnology, Ministry of Science & Technology, Government of India, under Grant No. BT/504/NE/TBP/2013.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The Institutional Animal Ethical Committee of Dibrugarh University India approved the experimental protocol (Approval No. IAEC/DU/120 dated Feb. 18, 2016)
Conflict of Interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Chakraborty, T., Das, M.K. De Novo Approach to Utilize Mango (Mangifera indica L.) Seed Kernel Lipid in Pharmaceutical Lipid Nanoformulation. J Pharm Innov 12, 226–237 (2017). https://doi.org/10.1007/s12247-017-9284-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12247-017-9284-y