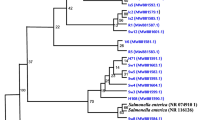
Abstract
All the V. cholerae non-O1, non-O139 isolates from ground and surface water samples collected during the rainy season (rainfall contributes significantly in the spread of cholera) contained ompW and a regulatory toxR gene, while many others possessed accessory cholera toxin (ace), hemolysin (hlyA) and outer membrane protein (ompU) genes. All the isolates lacked ctxAB, tcp, zot, rfbO1 and rfbO139 genes. The strains could be grouped into two main clusters colligating the isolates from ground water and surface water samples. The results suggest that surface water harbors various virulent V. cholerae strains that contaminate the ground water due to rain or poor hygienic practices, and result in the emergence of new toxigenic strains for cholera.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- APW:
-
alkaline peptone water
- BHI:
-
brain heart infusion
- CT:
-
cholera toxin
- NICED:
-
National Institute of Cholera and Enteric Diseases
- PCR:
-
polymerase chain reaction
- TCBS:
-
thiosulfate-citrate-bile salt-sucrose
- V.ch.:
-
Vibrio cholerae
References
Bidinost C., Saka H.A., Aliendro O., Sola C., Panzetta-Duttari G., Carranza P., Echenique J., Patrito E., Bocco J.L.: Virulence factors of non-O1 non-O139 Vibrio cholerae isolated in Cordoba, Argentina. Rev.Argent.Microbiol.36, 158–163 (2004).
Bik E.M., Bunschoten A.E., Gouw R.D., Mooi F.R.: Genesis of the novel epidemic Vibrio cholerae O139 strain: evidence for horizontal transfer of genes involved in polysaccharide synthesis. EMBO J.14, 209–216 (1995).
Chen Y., Johnson J.A., Pusch G.D., Morris J.G. Jr., Stine O.C.: The genome of non-O1 Vibrio cholerae NRT36S demonstrates the presence of pathogenic mechanisms that are distinct from those of O1 Vibrio cholerae. Infect.Immun.75, 2645–2647 (2007).
Colwell R.R., Huq A.: Environmental reservoir of Vibrio cholerae, the causative agent of cholera. Ann.N.Y.Acad.Sci.740, 44–54 (1994).
Crawford J.A., Kaper J.B., Dirita V.J.: Analysis of ToxR-dependent transcription activation of ompU, the gene encoding a major envelope protein in Vibrio cholerae. Mol.Microbiol.29, 235–246 (1998).
Dirita V.J., Parsot C., Jander G., Mekalanos J.J.: Regulatory cascade controls virulence in Vibrio cholerae. Proc.Nat.Acad.Sci. USA88, 5403–5407 (1991).
Faruque S.M., Sack D.A., Sack R.B., Colwell R.R., Takeda Y., Nair G.B.: Emergence and evolution of Vibrio cholerae O139. Proc.Nat.Acad.Sci.USA100, 1304–1309 (2003).
Goel A.K., Ponmariappan S., Kamboj D.V., Singh L.: Single multiplex polymerase chain reaction for environmental surveillance of toxigenic-pathogenic O1 and non-O1 Vibrio cholerae. Folia Microbiol.52, 81–85 (2007).
Kaper J.B., Morris J.G. Jr., LEVINE M.M.: Cholera. Clin.Microbiol.Rev.8, 48–86 (1995).
Lipp E.K., Rivera I.N., Gil A.I., Espeland E.M., Choopun N., Louis V.R., Russek-cohen E., Huq A., Colwell R.R.: Direct detection of Vibrio cholerae and ctxA in Peruvian coastal water and plankton by PCR. Appl.Environ.Microbiol.69, 3676–3680 (2003).
Mukhopadhyay A.K., Garg S., Mitra R., Basu A., Rajendran K., Dutta D., Bhattacharya S.K., Shimada T., Takeda T., Takeda Y., Nair G.B.: Temporal shifts in traits of Vibrio cholerae strains isolated from hospitalized patients in Calcutta: a 3-year (1993 to 1995) analysis. J.Clin.Microbiol.34, 2537–2543 (1996).
Nandi B., Nandy R.K., Mukhopadhyay S., Nair G.B., Shimada T., Ghose A.C.: Rapid method for species-specific identification of Vibrio cholerae using primers targeted to the gene of outer membrane protein OmpW. J.Clin.Microbiol.38, 4145–4151 (2000).
Provenzano D., Schuhmacher D.A., Barker J.L., Klose K.E.: The virulence regulatory protein ToxR mediates enhanced bile resistance in Vibrio cholerae and other pathogenic Vibrio species. Infect.Immun.68, 1491–1497 (2000).
Reidl J., Klose K.E.: Vibrio cholerae and cholera: out of the water and into the host. FEMS Microbiol.Rev.26, 125–139 (2002).
Rivera I.N., Chun J., Huq A., Sack R.B., Colwell R.R.: Genotypes associated with virulence in environmental isolates of Vibrio cholerae. Appl.Environ.Microbiol.67, 2421–2429 (2001).
Sharma C., Thungapathra M., Ghosh A., Mukhopadhyay A.K., Basu A., Mitra R., Basu I., Bhattacharya S.K., Shimada T., Ramamurthy T., Takeda T., Yamasaki S., Takeda Y., Nair G.B.: Molecular analysis of non-O1, non-O139 Vibrio cholerae associated with an unusual upsurge in the incidence of cholera-like disease in Calcutta, India. J.Clin.Microbiol.36, 756–763 (1998).
Shi L., Miyoshi S., Hiura M., Tomochika K., Shimada T., Shinoda S.: Detection of genes encoding cholera toxin (CT), zonula occludens toxin (ZOT), accessory cholera enterotoxin (ACE) and heat-stable enterotoxin (ST) in Vibrio mimicus clinical strains. Microbiol.Immunol.42, 823–828 (1998).
Singh D.V., Isac S.R., Colwell R.R.: Development of a hexaplex PCR assay for rapid detection of virulence and regulatory genes in Vibrio cholerae and Vibrio mimicus. J.Clin.Microbiol.40, 4321–4324 (2002).
Tamrakar A.K., Goel A.K., Kamboj D.V., Singh L.: Surveillance methodology for Vibrio cholerae in environmental samples. Internat. J.Environ.Health Res.16, 305–312 (2006).
Versalovic J., Schneider M., Brulin F.J.D., Lupski J.R.: Genomic fingerprinting of bacteria using repetitive sequence-based polymerase chain reaction. Methods Mol.Cell Biol.5, 25–40 (1994).
Yamamoto K., Al-omani M., Honda T., Takeda Y., Miwatani T.: Non-O1 Vibrio cholerae hemolysin: purification, partial characterization, and immunological relatedness to El Tor hemolysin. Infect.Immun.45, 192–196 (1984).
Yamamoto K., Ichinose Y., Nakasone N., Tanabe M., Nagahama M., Sakurai J., Iwanaga M.: Identity of hemolysins produced by Vibrio cholerae non-O1 and V. cholerae O1, biotype El Tor. Infect.Immun.51, 927–931 (1986).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Goel, A.K., Jain, M., Kumar, P. et al. Virulence profile and clonal relationship among the Vibrio cholerae isolates from ground and surface water in a cholera endemic area during rainy season. Folia Microbiol 55, 69–74 (2010). https://doi.org/10.1007/s12223-010-0011-z
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12223-010-0011-z