Abstract
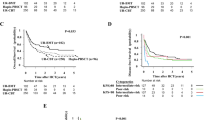
The clinical implications of recipient bone marrow nucleated cell count (NCC) prior to allogeneic hematopoietic stem cell transplantation (allo-HSCT) remain unknown. We conducted a multicenter retrospective study to evaluate the clinical significance of bone marrow NCC prior to allo-HSCT in patients with acute lymphoblastic leukemia. Patients who were in remission and underwent the initial allo-HSCT were included and stratified into high- and low-NCC groups using an NCC of 10 × 104/µL as the cut-off. The 3-year overall survival (OS), non-relapse mortality (NRM), and relapse rates for the high- and low-NCC groups were 51.2 vs. 84.5% (p < 0.001), 27.5 vs. 6.5% (p < 0.001), and 31.1 vs. 24.4% (p = 0.322), respectively. The high-NCC group had significantly poorer OS and higher NRM when compared with the low-NCC group. In summary, high recipient bone marrow NCC is associated with higher NRM and lower OS following allo-HSCT.


Similar content being viewed by others
Data availability
The data supporting the results of this study are available from the corresponding author upon reasonable request.
References
Lee SH, Erber WN, Porwit A, Tomonaga M, Peterson LC, International Council for Standardization in Hematology. ICSH guidelines for the standardization of bone marrow specimens and reports. Int J Lab Hematol. 2008;30:349–64. https://doi.org/10.1111/j.1751-553X.2008.01100.x.
Dominietto A, Lamparelli T, Raiola AM, Van Lint MT, Gualandi F, Berisso G, et al. Transplant-related mortality and long-term graft function are significantly influenced by cell dose in patients undergoing allogeneic marrow transplantation. Blood. 2002;100:3930–4. https://doi.org/10.1182/blood-2002-01-0339.
Tachibana T, Kanda J, Ishizaki T, Najima Y, Tanaka M, Doki N, et al. Outcomes and prognostic factors for patients with relapsed or refractory acute lymphoblastic leukemia who underwent allogeneic hematopoietic cell transplantation: a KSGCT multicenter analysis. Biol Blood Marrow Transplant. 2020;26:998–1004. https://doi.org/10.1016/j.bbmt.2020.01.007.
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. World Health Organization classification of tumours of haematopoietic and lymphoid tissues. ref. 4th ed. IARC: Lyon; 2017.
Hoelzer D, Gökbuget N. New approaches to acute lymphoblastic leukemia in adults: where do we go? Semin Oncol. 2000;27:540–59.
Giralt S, Ballen K, Rizzo D, Bacigalupo A, Horowitz M, Pasquini M, et al. Reduced-intensity conditioning regimen workshop: defining the dose spectrum. Report of a workshop convened by the center for international blood and marrow transplant research. Biol Blood Marrow Transplant. 2009;15:367–9. https://doi.org/10.1016/j.bbmt.2008.12.497.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J, et al. 1994 consensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15:825–8.
Shulman HM, Sullivan KM, Weiden PL, McDonald GB, Striker GE, Sale GE, et al. Chronic graft-versus-host syndrome in man. A long-term clinicopathologic study of 20 Seattle patients. Am J Med. 1980;69:204–17. https://doi.org/10.1016/0002-9343(80)90380-0.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013;48:452–8. https://doi.org/10.1038/bmt.2012.244.
Streck BP, Naufal G, Carrum G, Hill L, Heslop HE, Ramos CA, et al. Demographic and clinical donor characteristics as predictors of total nucleated cell concentrations in harvested marrow products. Transplant Cell Ther. 2021;27(785):e1-6. https://doi.org/10.1016/j.jtct.2021.05.021.
Ince UE, Ileri T, Dogu F, Ates C, Cakmakli H, Dalva K, et al. The impact of donor age and sex on the nucleated cell count and CD34 count in healthy bone marrow donors. Pediatr Transplant. 2015;19(4):385–90. https://doi.org/10.1111/petr.12453.
Nakasone H, Fukuda T, Kanda J, Mori T, Yano S, Kobayashi T, et al. Impact of conditioning intensity and TBI on acute GVHD after hematopoietic cell transplantation. Bone Marrow Transplant. 2015;50:559–65. https://doi.org/10.1038/bmt.2014.293.
Gupta A, Punatar S, Gawande J, Mathew L, Bagal B, Kannan S, et al. Risk factors, pattern and clinical outcome of acute graft versus host disease in acute leukemia patients undergoing allogeneic stem cell transplant. Indian J Hematol Blood Transfus. 2015;31:404–12. https://doi.org/10.1007/s12288-014-0499-5.
Lazaryan A, Weisdorf DJ, DeFor T, Brunstein CG, MacMillan ML, Bejanyan N, et al. Risk factors for acute and chronic graft-versus-host disease after allogeneic hematopoietic cell transplantation with umbilical cord blood and matched sibling donors. Biol Blood Marrow Transplant. 2016;22:134–40. https://doi.org/10.1016/j.bbmt.2015.09.008.
Flowers ME, Inamoto Y, Carpenter PA, Lee SJ, Kiem HP, Petersdorf EW, et al. Comparative analysis of risk factors for acute graft-versus-host disease and for chronic graft-versus-host disease according to National Institutes of Health consensus criteria. Blood. 2011;117:3214–9. https://doi.org/10.1182/blood-2010-08-302109.
Atsuta Y, Suzuki R, Nagamura-Inoue T, Taniguchi S, Takahashi S, Kai S, et al. Disease-specific analyses of unrelated cord blood transplantation compared with unrelated bone marrow transplantation in adult patients with acute leukemia. Blood. 2009;113:1631–8. https://doi.org/10.1182/blood-2008-03-147041.
Fowler DH, Gress RE. Th2 and Tc2 cells in the regulation of GVHD, GVL, and graft rejection: considerations for the allogeneic transplantation therapy of leukemia and lymphoma. Leuk Lymphoma. 2000;38:221–34. https://doi.org/10.3109/10428190009087014.
Kawashima N, Terakura S, Nishiwaki S, Koyama D, Ozawa Y, Ito M, et al. Increase of bone marrow macrophages and CD8+ T lymphocytes predict graft failure after allogeneic bone marrow or cord blood transplantation. Bone Marrow Transplant. 2017;52:1164–70. https://doi.org/10.1038/bmt.2017.58.
Acknowledgements
We would like to thank the medical staff at the Yokohama City University Medical Center, Yokohama City University School of Medicine, and Kanagawa Cancer Center for their excellent patient care.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
We gratefully acknowledge the contributions of all co-authors for data collection and analysis support. JN collected and analyzed data and wrote the manuscript. TT designed the study concept and comprehensively supported the study. TM collected data. MT and KY supported the statistical analysis. KM, YI, AN, YN, AM, TS, AI, and NH were the physicians for the patients. MH, SF, and HK were responsible for the participating centers. HN supervised the study. All authors critically revised the report, commented on drafts of the manuscript, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
All authors, except those mentioned below, declare no conflict of interest. TT reports honoraria from Pfizer, Otsuka, MSD, Chugai, Parma, and Astellas outside the submitted work. SF has received honoraria from Bristol-Myers-Squibb, Astellas, Nippon Shinyaku, Otsuka, Pfizer, Novartis, MSD, Sanofi, Janssen, SymBio, Kyowa Hakko Kirin, AstraZeneca, CSL Behring, Meiji Seika Pharma, AbbVie, Takeda, and Chugai Pharma, and received research funding from Shionogi, Kyowa Hakko Kirin, Chugai Pharma, Otsuka, Asahi-Kasei, and Daiichi Sankyo outside the submitted work. HN reports honoraria from Novartis and Daiichi-Sankyo, scholarships from Daiichi-Sankyo, Celgene, Chugai, Nihon-Shinyaku, Astellas, Asahi Kasei pharma, Takeda, Pfizer, and Eisai outside the submitted work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
12185_2023_3688_MOESM1_ESM.pptx
Supplementary Figure 1. A consort diagram of the patient selection. ALL, acute lymphoblastic leukemia; allo-HSCT, allogeneic hematopoietic stem cell transplantation; BM, bone marrow; NCC, nucleated cell count. Supplementary Figure 2. Receiver operating characteristic curve for the OS by NCC.OS, overall survival; NCC, nucleated cell count. Supplemental Figure 3. Correlations between NCC and sex (A), karyotype (B), and MRD (C). MRD, minimal residual disease; NCC, nucleated cell count. Supplemental Figure 4. The correlation between patient age and NCC; NCC, nucleated cell count. Supplemental Figure 5. Survival probability and cumulative incidence of relapse and NRM in Ph-negative and Ph-positive subgroups. Probability of OS in Ph-negative (A) and Ph-positive (B) subgroups, cumulative incidence of NRM in Ph-negative (C) and Ph-positive (D) subgroups, cumulative incidence of relapse in Ph-negative (E) and Ph-positive (F) subgroups. NRM, non-relapse mortality; OS, overall survival; Ph, Philadelphia
About this article
Cite this article
Nukui, J., Tachibana, T., Miyazaki, T. et al. Clinical significance of total nucleated cell count in bone marrow of patients with acute lymphoblastic leukemia who underwent allogeneic hematopoietic stem cell transplantation. Int J Hematol 119, 62–70 (2024). https://doi.org/10.1007/s12185-023-03688-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-023-03688-7