Abstract
Background
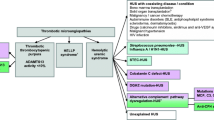
CD36-deficient individuals may produce anti-CD36 antibodies through antigenic exposure to CD36, in situations including blood transfusions. Therefore, allogeneic hematopoietic stem cell transplantation (HSCT) from CD36-positive donors to CD36-negative patients remains a challenge.
Case report
A 64-year-old man with acute myeloid leukemia became refractory to platelet transfusions during chemotherapy. Anti-CD36 antibodies without anti-HLA antibodies were detected in serum, and the absence of CD36 expression on platelets and monocytes confirmed type I CD36 deficiency. The patient achieved complete remission, and received maintenance therapy with CD36-negative platelet transfusions. However, he relapsed soon afterward, and thus underwent peripheral blood stem cell transplantation (PBSCT) from a CD36-positive unrelated donor. The anti-CD36 antibody titer had decreased before the transplant, and the PBSCT-course was uneventful. The patient has been well without any complications associated with CD36 status mismatch.
Discussion
The few reports of allogeneic HSCT in patients with CD36 deficiency have suggested that anti-CD36 antibodies could be involved in several post-transplant complications, such as delayed platelet recovery, transfusion refractoriness, and transfusion-related acute lung injury. Our present case confirmed that stem cell transplantation from CD36-positive donors to negative patients is feasible, when it includes careful prior assessment of anti-CD36 antibody titers and interventions to attenuate them.

Similar content being viewed by others
Data availability
The authors declare that data supporting the findings of this study are fully described within the manuscript.
Abbreviations
- AML:
-
Acute myeloid leukemia
- CCI:
-
Corrected count increment
- DSA:
-
Donor-specific anti-HLA antibodies
- FNAIT:
-
Fetal-neonatal alloimmune thrombocytopenia
- GVHD:
-
Graft-versus-host disease
- HSCT:
-
Hematopoietic stem cell transplantation
- PBSCT:
-
Peripheral blood stem cell transplantation
- PT:
-
Platelet transfusion
- PTR:
-
Platelet transfusion refractoriness
- TRALI:
-
Transfusion-related acute lung injury
References
Tomiyama Y, Take H, Ikeda H, Mitani T, Furubayashi T, Mizutani H, et al. Identification of the platelet-specific alloantigen, Naka, on platelet membrane glycoprotein IV. Blood. 1990;75:684–7.
Greenwalt DE, Lipsky RH, Ockenhouse CF, Ikeda H, Tandon NN, Jamieson GA. Membrane glycoprotein CD36: a review of its roles in adherence, signal transduction, and transfusion medicine. Blood. 1992;80:1105–15.
Park YM. CD36, a scavenger receptor implicated in atherosclerosis. Exp Mol Med. 2014;46:e99.
Bierling P, Godeau B, Fromont P, Bettaieb A, Debili N, el-Kassar N, et al. Posttransfusion purpura-like syndrome associated with CD36 (Naka) isoimmunization. Transfusion. 1995;35:777–82.
Lee K, Godeau B, Fromont P, Plonquet A, Debili N, Bachir D, et al. CD36 deficiency is frequent and can cause platelet immunization in Africans. Transfusion. 1999;39:873–9.
Yamamoto N, Akamatsu N, Sakuraba H, Yamazaki H, Tanoue K. Platelet glycoprotein IV (CD36) deficiency is associated with the absence (type I) or the presence (type II) of glycoprotein IV on monocytes. Blood. 1994;83:392–7.
Ikeda H, Mitani T, Ohnuma M, Haga H, Ohtzuka S, Kato T, et al. A new platelet-specific antigen, Naka, involved in the refractoriness of HLA-matched platelet transfusion. Vox Sang. 1989;57:213–7.
Shibata Y, Kim N, Morita S, Ishijima A, Okazaki S. Monoclonal antibody OKM5 and platelet alloantibody anti-Naka have the same specificity. Proc Jpn Acad Ser B. 1990;66:41–2.
Yamamoto N, Ikeda H, Tandon NN, Herman J, Tomiyama Y, Mitani T, et al. A platelet membrane glycoprotein (GP) deficiency in healthy blood donors: Naka-platelets lack detectable GPIV (CD36). Blood. 1990;76:1698–703.
Nakajima F, Nishimura M, Hashimoto S, Okazaki H, Tadokoro K. Role of anti-Naka antibody, monocytes and platelets in the development of transfusion-related acute lung injury. Vox Sang. 2008;95:318–23.
Matsui M, Kajiwara K, Tsukada M, Iwama K, Yamada K, Kodo H, et al. A case of Philadelphia chromosome-positive acute lymphocytic leukaemia with type I CD36 deficiency. Vox Sang. 2022;117:128–32.
Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127:2391–405.
Ito T, Daitoku S, Nishi Y, Yoshida M, Yamanoha A, Numata A, et al. Anti-CD36 allo-antibodies in a patient undergoing hematopoietic stem cell transplantation. In: The 32nd annual meeting of the Japan Society for Hematopoietic Cell Transplantation. Hamamatsu, 2010;PS-1–50:245 (in Japanese).
Zhou Y, Li LL, Zhong ZL, Liu XJ, Liu JL, Shen WD, et al. Anti-CD36 mediated platelet transfusion refractoriness and related cases after stem cell transplantation. Zhongguo Shi Yan Xue Ye Xue Za Zhi [J Exp Hematol]. 2018;26:541–6 (in Chinese).
Yamamoto H, Uchida N, Yuasa M, Kageyama K, Ota H, Kaji D, et al. A novel reduced-toxicity myeloablative conditioning regimen using full-dose busulfan, fludarabine, and melphalan for single cord blood transplantation provides durable engraftment and remission in nonremission myeloid malignancies. Biol Blood Marrow Transplant. 2016;22:1844–50.
Yuasa-Kawase M, Masuda D, Yamashita T, Kawase R, Nakaoka H, Inagaki M, et al. Patients with CD36 deficiency are associated with enhanced atherosclerotic cardiovascular diseases. J Atheroscler Thromb. 2012;19:263–75.
Sato T, Tsuno NH, Kozai Y, Yokoyama H, Yano S. Should unrelated haematopoietic progenitor cell donors be tested for CD36 in Japan? Lancet Haematol. 2022;9:e802.
Chen L, Gao Z, Zhu J, Rodgers GP. Identification of CD13+CD36+ cells as a common progenitor for erythroid and myeloid lineages in human bone marrow. Exp Hematol. 2007;35:1047–55.
Nestorowa S, Hamey FK, Pijuan Sala B, Diamanti E, Shepherd M, Laurenti E, et al. A single-cell resolution map of mouse hematopoietic stem and progenitor cell differentiation. Blood. 2016;128:e20–31.
Chen D-W, Kang T, Xu X-Z, Xia W-J, Ye X, Wu Y-B, et al. Mechanism and intervention of murine transfusion-related acute lung injury caused by anti-CD36 antibodies. JCI Insight. 2023;8:e165142.
Khatri SS, Curtis BR, Yamada C. A case of platelet transfusion refractoriness due to anti-CD36 with a successful treatment outcome. Immunohematology. 2019;35:139–44.
Yae H, Shiragami H, Matsuhashi M, Takahashi D, Tsuno H, Kawazoe M, et al. Successful engraftment for Ph+ALL with anti-CD36 antibody in allogeneic BMT. In: The 44th annual meeting of the Japanese Society for Transplantation and Cellular Therapy. Yokohama, 2022;PS-29–3:327 (in Japanese)
Ciurea SO, Al Malki MM, Kongtim P, Zou J, Aung FM, Rondon G, et al. Treatment of allosensitized patients receiving allogeneic transplantation. Blood Adv. 2021;5:4031–43.
Yoshihara S, Maruya E, Taniguchi K, Kaida K, Kato R, Inoue T, et al. Risk and prevention of graft failure in patients with preexisting donor-specific HLA antibodies undergoing unmanipulated haploidentical SCT. Bone Marrow Transplant. 2012;47:508–15.
Leffell MS, Jones RJ, Gladstone DE. Donor HLA-specific Abs: to BMT or not to BMT? Bone Marrow Transplant. 2015;50:751–8.
Acknowledgements
We would like to express our sincere gratitude to Dr. Takuo Ito from the National Hospital Organization Kure Medical Center and Chugoku Cancer Center for providing crucial clinical information on the patient, and to Dr. Katsushi Tajima from the Yamagata Prefectural Central Hospital for offering invaluable feedback on this manuscript.
Funding
No funding was secured for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Okuyama, S., Sumi, M., Ishikawa, R. et al. Successful allogeneic hematopoietic stem cell transplantation in a patient with type I CD36 deficiency: a case study and literature review. Int J Hematol 118, 656–660 (2023). https://doi.org/10.1007/s12185-023-03637-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-023-03637-4