Abstract
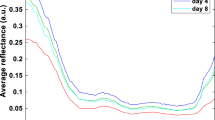
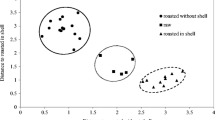
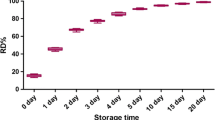
Raman scattering and chemometrics were used to predict the lipid degradation and storage time of chilled beef flank by using a portable Raman spectrometer. Results showed that the performance of the combination of the three indicators in terms of predicting the storage time was more accurate than that of their independent usage (R2 = 0.92 compared with 0.88, 0.35, and 0.42). Raman spectroscopy could predict the storage time (R2cv = 0.76), but it could not precisely predict acid value [Av], peroxide value [Pov], and thiobarbituric acid-reactive substances [TBARS] (R2cv = 0.42–0.50, 0.18–0.40, 0.41–0.43) by using three processing methods. The prediction of Av, Pov, TBARS, and storage time based on Raman intensities at the characteristic Raman shifts were more precise (R2 = 0.87–0.95) than based on Raman intensities at full bands. Therefore, Raman spectroscopy and chemometrics could be applied to perform nondestructive quantitation and determine the lipid degradation and storage time of chilled beef flank.



Similar content being viewed by others
Data Availability
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
References
Afseth NK, Wold JP, Segtnan VH (2006) The potential of Raman spectroscopy for characterisation of the fatty acid unsaturation of salmon. Anal Chim Acta 572(1):85–92. https://doi.org/10.1016/j.aca.2006.05.013
Amaral AB, Silva MVD, Lannes SCDS (2018) Lipid oxidation in meat: mechanisms and protective factors - a review. Ciênc Tecnol Aliment 38(29):1–15. https://doi.org/10.1590/fst.32518
Berhe DT, Engelsen SB, Hviid MS, Lametsch R (2014) Raman spectroscopic study of effect of the cooking temperature and time on meat proteins. Food Res Int 66:123–131. https://doi.org/10.1016/j.foodres.2014.09.010
Berhe DT, Eskildsen CE, Lametsch R, Hviid MS, Berg FVD, Engelsen SB (2016) Prediction of total fatty acid parameters and individual fatty acids in pork backfat using raman spectroscopy and chemometrics: understanding the cage of covariance between highly correlated fat parameters. Meat Sci 111(1):18–26. https://doi.org/10.1016/j.meatsci.2015.08.009
Beattie JR, Bell SEJ, Moss BW (2004) A critical evaluation of Raman spectroscopy for the analysis of lipids: fatty acid methyl esters. Lipids 39(5):407–419. https://doi.org/10.1007/s11745-004-1245-z
Beattie JR, Bell SEJ, Borgaard C, Fearon AM, Moss BW (2004) Multivariate prediction of clarified butter composition using Raman spectroscopy. Lipids 39(9):897–906. https://doi.org/10.1007/s11745-004-1312-5
Beattie JR, Bell SEJ, Borgaard C, Fearon A, Moss BW (2006) Prediction of adipose tissue composition using raman spectroscopy: average properties and individual fatty acids. Lipids 41(3):287–294. https://doi.org/10.1007/s11745-006-5099-1
Beattie JR, Bell SEJ, Borggaard C, Fearon AM, Moss BW (2007) Classification of adipose tissue species using Raman spectroscopy. Lipids 42(7):679–685. https://doi.org/10.1007/s11745-007-3059-z
Bystricky P, Turek P (1998) Fat oxidation in cooked meat products during their storage in a chilled shop display. Folia Veterinaria 42(2):101–104
Daniel I, Edwards HGM (2012) Raman spectroscopy in biogeology and astrobiology. European Mineralogical Union Notes Mineral 12(1):391–413
Das RS, Agrawal YK (2011) Raman spectroscopy: recent advancements, techniques and applications. Vib Spectrosc 57(2):163–176. https://doi.org/10.1016/j.vibspec.2011.08.003
Dragoev S, Balev D, Ivanov G, Nikolova-Damyanova B, Grozdeva T, Filizov E et al (2014) Effect of superficial treatment with new natural antioxidant on salmon (salmo salar) lipid oxidation. Acta Aliment 43(1):1–8. https://doi.org/10.1556/AAlim.43.2014.1.1
Du H, Li H (2008) Antioxidant effect of cassia essential oil on deep-fried beef during the frying process. Meat Sci 78(4):461–468. https://doi.org/10.1016/j.meatsci.2007.07.015
Engelsen SB (1997) Explorative spectrometric evaluations of frying oil deterioration. J Am Oil Chem Soc 74(12):1495–1508. https://doi.org/10.1007/s11746-997-0068-2
Estevez M (2015) Oxidative damage to poultry: from farm to fork. Poult Sci 94(6):1368–1378. https://doi.org/10.3382/ps/pev094
Fowler, S. M. , Ponnampalam, E. N. , Schmidt, H. , Wynn P Hopkins, DL 2015 Prediction of intramuscular fat content and major fatty acid groups of lamb m. longissimus lumborum using raman spectroscopy. Meat ence 110 11 70–75 https://doi.org/10.1016/j.meatsci.2015.06.016
Dollish FR, Fately WG, Bentley FF (1974) Characteristic Raman frequencies of organic compounds. Wiley 1974:21–75
Fugger J, Zilch KT, Cannon JA, Dutton HJ (1951) Analysis of fat acid oxidation products by countercurrent distribution methods ii methyl oleate2. J American Chem Soc 73(6):285–289. https://doi.org/10.1007/BF02648645
Herrero AM (2008) Raman spectroscopy for monitoring protein structure in muscle food systems. Crit Rev Food Sci Nutr 48(6):512–523. https://doi.org/10.1080/10408390701537385
Ji W, Yao W (2015) Rapid surface enhanced Raman scattering detection method for chloramphenicol residues. Spectrochim Acta Part A Mol Biomol Spectrosc 144:125–130. https://doi.org/10.1016/j.saa.2015.02.029
Karpińska-Tymoszczyk M (2014) The effect of antioxidants, packaging type and frozen storage time on the quality of cooked turkey meatballs. Food Chem 148:276–283. https://doi.org/10.1016/j.foodchem.2013.10.054
Lyndgaard LB, Sørensen KM, Berg F, Engelsen SB (2011) Depth profiling of porcine adipose tissue by Raman spectroscopy. J Raman Spectrosc 43(4):482–489. https://doi.org/10.1002/jrs.3067
Li-Chan, E. C. Y., Griffith, P. R., & Chalmers, J. M. 2010. Appl vibrational spectroscopy food sci: Wiley & Sons.
Lima DM, Rangel N, Moreno MB (2013) Oxidao lipdica e qualidade da carne ovina [lipid oxidation and lamb meat quality]. Acta Veterinaria Brasilica 7(1):14–28
Morris Michael D (2006) Review - modern Raman spectroscopy: a practical approach. Anal Chem 78(1):33–33. https://doi.org/10.1021/ac069338n
Min B, Ahn U (2005) Mechanism of lipid peroxidation in meat and meat products - a review. Food Sci Biotechnol 14(1):152–163
R Mozuraityte V Kristinova T Rustad 2016 Oxidation of food components Encycl Food Health 186–190https://doi.org/10.1016/B978-0-12-384947-2.00508-0
Okada Y, Okajima H, Konishi H, Terauchi M, Ishii K, Liu IM et al (1990) Antioxidant effect of naturally occurring furan fatty acids on oxidation of linoleic acid in aqueous dispersion. J Am Oil Chem Soc 67(11):858–862. https://doi.org/10.1007/BF02540506
Olsen EF, Baustad C, Egelandsdal B, Rukke EO, Isaksson T (2010) Long-term stability of a Raman instrument determining iodine value in pork adipose tissue. Meat Sci 85(1):1–6. https://doi.org/10.1016/j.meatsci.2009.12.008
Pinheiro RSB, Francisco CL, Lino DM, Borba H (2018) Meat quality of Santa Inês lamb chilled-then-frozen storage up to 12 months. Meat Sci 148:72–78. https://doi.org/10.1016/j.meatsci.2018.09.017
Ponginebbi L, Nawar WW, Chinachoti P (1999) Oxidation of linoleic acid in emulsions: effect of substrate, emulsifier, and sugar concentration. J American Oil Chem Soc 76(1):131
Qin J, Kim MS, Chao K, Schmidt WF, Huang M (2017) Subsurface inspection of food safety and quality using line-scan spatially offset raman spectroscopy technique. Food Control 75:246–254. https://doi.org/10.1016/j.foodcont.2016.12.012
Schmidt H, Sowoidnich K, Kronfeldt HD (2010) A prototype hand-held Raman sensor for the in situ characterization of meat quality. Appl Spectrosc 64(8):888–894. https://doi.org/10.1366/000370210792081028
Schmidt H, Scheier R, Hopkins DL (2013) Preliminary investigation on the relationship of Raman spectra of sheep meat with shear force and cooking loss. Meat Sci 93(1):138–143. https://doi.org/10.1016/j.meatsci.2012.08.019
Van Hecke T, Van Camp J, De Smet S (2017) Oxidation during digestion of meat: interactions with the diet and helicobacter pylori gastritis, and implications on human health. Comprehensive Rev Food Sci Food Safety 16(2):214–233. https://doi.org/10.1111/1541-4337.12248
Wan X, Guo ZY (2012) Research of factors on fat oxidation in meat products. Livestock Poult Ind 09:48–51. https://doi.org/10.19567/j.cnki.1008-0414.2012.09.031
Wang GN, Zhang L, Song YP, Liu JX, Wang JP (2017) Application of molecularly imprinted polymer based matrix solid phase dispersion for determination of fluoroquinolones, tetracyclines and sulfonamides in meat. J Chromatogr B 1065–1066:104–111. https://doi.org/10.1016/j.jchromb.2017.09.034
Warren H, Enser M, Richardson RI, Wood J, Scollan N (2003) Effect of breed and diet on total lipid and selected shelf-life parameters in beef muscle. BMJ 1(5072):691
Warren, H. E. , Enser, M. , Richardson, R. I. , Nute, G. , & Scollan, N. D. 2009. Effect of diet, breed and age at slaughter on the fatty acid composition of total lipid in beef longissimus muscle. 7th British Grassland Society (BGS) Research Conference. British Grassland Soc, pp. 25 – 26. OAI.
Weng YM, Weng RH, Tzeng CY, Chen W (2003) Structural analysis of triacylglycerols and edible oils by near-infrared fourier transform raman spectroscopy. Appl Spectrosc 57(4):413–418
Witte VC, Krause GF, Bailey ME (1970) A new extraction method for determining 2-thiobarbituric acid values for pork and beef during storage. J Food Sci 35(35):585–592. https://doi.org/10.1111/j.1365-2621.1970.tb04815.x
Wu J-B, Lin M-L, Cong X, Liu H-N, Tan P-H (2018) Raman spectroscopy of graphene-based materials and its applications in related devices. Chem Soc Rev 47(5):1822–1873. https://doi.org/10.1039/C6CS00915H
Yang D, Ying YB (2011) Applications of Raman spectroscopy in agricultural products and food analysis: a review. Appl Spec Rev 46(7):539–560. https://doi.org/10.1080/05704928.2011.593216
Zilch KT, Dutton HJ (1951) Analysis of fat acid oxidation product by countercurrent distribution methods. Anal Chem 23(5):775–778
Funding
This work has been financially supported by the “Ningxia Hui Autonomous Region Key R&D Program Major Technology Project” and funded by the Ningxia Science and Technology Department (Grant number 2017BY068).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bai, J., Zang, M., Zou, H. et al. Prediction of the Lipid Degradation and Storage Time of Chilled Beef Flank by Using Raman Spectroscopy and Chemometrics. Food Anal. Methods 15, 2213–2223 (2022). https://doi.org/10.1007/s12161-022-02276-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-022-02276-5