Abstract
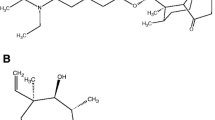
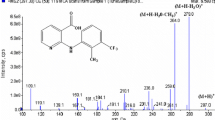
Tiamulin is a semi-synthetic derivative of the natural antibiotic pleuromutilin and is widely used as a veterinary drug for swine. Herein, we report the development of a sensitive and reliable method for determining 8α-hydroxymutilin as a marker residue for tiamulin in swine tissue using liquid chromatography-tandem mass spectrometry (LC-MS/MS). The method consists of sample extraction with acetone, defatting by acetonitrile/hexane partitioning, hydrolysis of the tiamulin metabolites to 8α-hydroxymutilin under alkaline conditions, liquid-liquid extraction with ethyl acetate, cleanup using a primary secondary amine cartridge, and LC-MS/MS analysis. The developed method was validated for 8α-hydroxymutilin in swine muscle, fat, and liver at two levels, namely 0.01 mg/kg and the maximum residue limits established in Japan (i.e., 0.1 mg/kg for swine muscle and fat, and 0.6 mg/kg for liver). The trueness ranged from 82 to 89%, and the relative standard deviations ranged from 1 to 3%. No chromatographic interference was observed near the retention time of 8α-hydroxymutilin, and matrix effects were negligible for all matrices, suggesting that the cleanup protocol was effective. The calibration curve was linear in the 0.005–0.5 μg/mL range, with a coefficient of determination greater than 0.997. The developed method enabled accurate quantification using solvent-based calibration without compensating for matrix effects and losses during sample preparation. The limit of detection of the method was 0.0005 mg/kg for each matrix. The developed method is suitable for regulatory-purpose analysis of 8α-hydroxymutilin as a marker residue for tiamulin as defined by the European Union and several other countries.




Similar content being viewed by others
References
Ben W, Qiang Z, Adams C, Zhang H, Chen L (2008) Simultaneous determination of sulfonamides, tetracyclines and tiamulin in swine wastewater by solid-phase extraction and liquid chromatography–mass spectrometry. J Chromatogr A 1202:173–180. https://doi.org/10.1016/j.chroma.2008.07.014
Bohm DA, Stachel CS, Gowik P (2011) Validated determination of eight antibiotic substance groups in cattle and pig muscle by HPLC/MS/MS. J AOAC Int 94:407–419
De Baere S, Devreese M, Maes A, De Backer P, Croubels S (2015) Quantification of 8α-hydroxymutilin as marker residue for tiamulin in rabbit tissues by high-performance liquid chromatography-mass spectrometry. Anal Bioanal Chem 407:4437–4445. https://doi.org/10.1007/s00216-014-8437-9
European Commission (2002) Commission Decision of 12 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results. 2002/657/EC. Off J Eur Union L221:8–36. https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2002:221:0008:0036:EN:PDF. Accessed 2 June 2020
European Commission (2010) Commission Regulation (EU) No 37/2010 of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin. Off J Eur Union L15:1–72. https://eur-lex.europa.eu/legal-content/EN/TXT/PDF/?uri=CELEX:32010R0037&from=EN. Accessed 2 June 2020
European Medicines Agency (EMA) (1999) Committee for veterinary medicinal products, tiamulin: summary report (1), EMEA/MRL/578/99-FINAL-corr. https://www.ema.europa.eu/en/documents/mrl-report/tiamulin-summary-report-1-committee-veterinary-medicinal-products_en.pdf. Accessed 2 June 2020
Hu F-Y, He L-M, Yang J-W, Bian K, Wang Z-N, Yang H-C, Liu Y-H (2014) Determination of 26 veterinary antibiotics residues in water matrices by lyophilization in combination with LC–MS/MS. J Chromatogr B 949–950:79–86. https://doi.org/10.1016/j.jchromb.2014.01.008
Islam KMS, Klein U, Burch DGS (2009) The activity and compatibility of the antibiotic tiamulin with other drugs in poultry medicine - a review. Poult Sci 88:2353–2359. https://doi.org/10.3382/ps.2009-00257
Lykkeberg AK, Cornett C, Halling-Sørensen B, Hansen SH (2006) Isolation and structural elucidation of tiamulin metabolites formed in liver microsomes of pigs. J Pharm Biomed Anal 42:223–231. https://doi.org/10.1016/j.jpba.2006.03.023
Markus JR, Sherma J (1993a) Method. IV. Gas Chromatographic determination of tiamulin residues in swine liver. J AOAC Int 76:451–458. https://doi.org/10.1093/jaoac/76.2.451
Markus JR, Sherma J (1993b) Method. V. Gas chromatographic/mass spectrometric confirmation of 8-hydroxymutilin, a tiamulin metabolite, in swine liver extracts. J AOAC Int 76:459–460. https://doi.org/10.1093/jaoac/76.2.459
Ministry of Health, Labour and Welfare, Japan (MHLW) (2010) Director Notice; Guideline for the validation of analytical methods for testing agricultural chemical residues in food. Syoku-An No. 1224–1. https://www.mhlw.go.jp/english/topics/foodsafety/positivelist060228/dl/181130_21.pdf. Accessed 2 June 2020
Novak R, Shlaes DM (2010) The pleuromutilin antibiotics: a new class for human use. Curr Opin Investig Drugs 11:182–191
Nozal MJ, Bernal JK, Martín MT, Jiménez JJ, Bernal J, Higes M (2006) Trace analysis of tiamulin in honey by liquid chromatography-diode array-electrospray ionization mass spectrometry detection. J Chromatogr A 1116:102–108. https://doi.org/10.1016/j.chroma.2006.03.028
Patyra E, Nebot C, Gavilán RE, Cepeda A, Kwiatek K (2018) Development and validation of an LC-MS/MS method for the quantification of tiamulin, trimethoprim, tylosin, sulfadiazine and sulfamethazine in medicated feed. Food Addit Contam A 35:882–891. https://doi.org/10.1080/19440049.2018.1426887
Pomorska-Mól M, Kowalski C (2009) Determination of tiamulin in chickens’ plasma by HPLC with UV-VIS detection. J Liq Chromatogr Relat Technol 32:1023–1031. https://doi.org/10.1080/10826070902791122
Poulsen SM, Karlsson M, Johansson LB, Vester B (2001) The pleuromutilin drugs tiamulin and valnemulin bind to the RNA at the peptidyl transferase centre on the ribosome. Mol Microbiol 41:1091–1099. https://doi.org/10.1046/j.1365-2958.2001.02595.x
Robert C, Brasseur PY, Dubois M, Delahaut P, Gillard N (2016) Development and validation of rapid multiresidue and multi-class analysis for antibiotics and anthelmintics in feed by ultra-high-performance liquid chromatography coupled to tandem mass spectrometry. Food Addit Contam A 33:1312–1323. https://doi.org/10.1080/19440049.2016.1207808
Saito-Shida S, Sakai T, Nemoto S, Akiyama H (2017) Quantitative analysis of veterinary drugs in bovine muscle and milk by liquid chromatography quadrupole time-of-flight mass spectrometry. Food Addit Contam A 34:1153–1161. https://doi.org/10.1080/19440049.2017.1333161
Schlünzen F, Pyetan E, Fucini P, Yonath A, Harms JM (2004) Inhibition of peptide bond formation by pleuromutilins: the structure of the 50S ribosomal subunit from Deinococcus radiodurans in complex with tiamulin. Mol Microbiol 54:1287–1294. https://doi.org/10.1111/j.1365-2958.2004.04346.x
Schlüsener MP, Bester K, Spiteller M (2003) Determination of antibiotics such as macrolides, ionophores and tiamulin in liquid manure by HPLC-MS/MS. Anal Bioanal Chem 375:942–947. https://doi.org/10.1007/s00216-003-1838-9
Sun F, Yang S, Zhang H, Zhou J, Li Y, Zhang J, Jin Y, Wang Z, Li Y, Shen J, Zhang S, Cao X (2017) Comprehensive analysis of tiamulin metabolites in various species of farm animals using ultra-high-performance liquid chromatography coupled to quadrupole/time-of-flight. J Agric Food Chem 65:199–207. https://doi.org/10.1021/acs.jafc.6b04377
Tan A, Gu G, Gui X, Wu Y, Bolger G, Licollari A, Fanaras JC (2018) Improving the selectivity and sensitivity for quantifying 8α-hydroxymutilin in rabbit tissues by using basic mobile phases and negative ionization. J Chromatogr B 1072:142–148. https://doi.org/10.1016/j.jchromb.2017.11.008
Tang YZ, Liu YH, Chen JX (2012) Pleuromutilin and its derivatives-the lead compounds for novel antibiotics. Mini Rev Med Chem 12:53–61. https://doi.org/10.2174/138955712798868968
van Duijkeren E, Greko C, Pringle M, Baptiste KE, Catry B, Jukes H, Moreno MA, Ferreira Pomba MCM, Pyörälä S, Rantala M, Ružauskas M, Sanders P, Teale C, Threlfall EJ, Torren-Edo J, Törneke K (2014) Pleuromutilins: use in food-producing animals in the European Union, development of resistance and impact on human and animal health. J Antimicrob Chemother 69:2022–2031. https://doi.org/10.1093/jac/dku123
Funding
This study was supported by the Ministry of Health, Labour and Welfare of Japan (grant number 19KA1002).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Shizuka Saito-Shida declares no conflict of interest. Nao Kashiwabara declares no conflict of interest. Satoru Nemoto declares no conflict of interest. Hiroshi Akiyama declares no conflict of interest.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed Consent
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 121 kb)
Rights and permissions
About this article
Cite this article
Saito-Shida, S., Kashiwabara, N., Nemoto, S. et al. Determination of 8α-hydroxymutilin as a Marker Residue for Tiamulin in Swine Tissue by Liquid Chromatography-Tandem Mass Spectrometry. Food Anal. Methods 14, 845–855 (2021). https://doi.org/10.1007/s12161-020-01950-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12161-020-01950-w