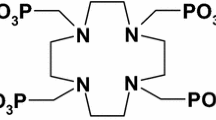
Abstract
Objective
Optimized production and quality control of gallium-68 labeled ethylenediamine tetramethylene phosphonate (68Ga-EDTMP) as an efficient PET radiotracer for bone scans have been presented.
Methods
Efforts have been made to present a fast, efficient, cost-effective and facile protocol for 68Ga-EDTMP productions for clinical trials. 68Ga-EDTMP was prepared using generator-based 68GaCl3 and EDTMP at optimized conditions for time, temperature, ligand amount, gallium content followed by proper formulation. The biodistribution of the tracer in rats was studied using tissue counting and PET/CT imaging up to 155 min.
Results
68Ga-EDTMP was prepared at optimized conditions in 5–10 min at 50–60 °C (radiochemical purity ≈99 ± 0.88 % ITLC, >99 % HPLC, specific activity: 15–18 GBq/mM). The biodistribution of the tracer demonstrated high bone uptake of the tracer in 10–20 min while yielding the best images in 2 h.
Conclusion
The whole production and quality control of 68Ga-EDTMP including labeling, purification, HPLC analysis, sterilization and LAL test took 18–20 min with significant specific activity for administration to limited number of patients in a PET center.











Similar content being viewed by others
References
Sudbrock F, Fischer T, Zimmermann B, Guliyev M, Dietlein M, Drezezga A, et al. Characterization of SnO2-based 68Ge/68Ga generators and 68Ga-DOTATATE preparations: radionuclide purity, radiochemical yield and long-term constancy. EJNMMI Res. 2014;4:36.
Fazaeli Y, Jalilian AR, Amini MM, Ardaneh K, Rahiminejad A, Bolourinovin F, et al. Development of 68Ga-fluorinated porphyrin complex as a possible PET imaging agent. Nucl Med Mol Imaging. 2012;46:20–6.
Velikyan I. Prospective of 68Ga-radiopharmaceutical development. Theranostics. 2014;4:47–80.
Serafini AN. Therapy of metastatic bone pain. J Nucl Med. 2001;42:895–906.
Bayouth JE, Macey DJ, Kasi LP, Fosselia FV. Dosimetry and toxicity of samarium-153-EDTMP administered for bone pain due to skeletal metastases. J Nucl Med. 1994;35:63–9.
Fellner M, Riss P, Loktionova N, Zhernosekov K, Thews O, Geraldes CFGC, et al. Comparison of different phosphorus-containing ligands complexing 68Ga for PET-imaging of bone metabolism. Radiochem Acta. 2011;99:43–51.
Ogawa K, Ishizaki A, Takai K, Kitamura Y, Kiwada T, Shiba K, et al. Development of novel radiogallium-labeled bone imaging agents using oligo-aspartic acid peptides as carriers. PLoS One. 2013;8:e84335.
Fellner M, Biesalski B, Bausbacher N, Kubicek V, Herman P, Rösch F, et al. 68Ga-BPAMD: PET-imaging of bone metastases with generator based positron emitter. Nucl Med Biol. 2012;39:993–9.
Fellner M, Baum RP, Kubicek V, Hermann P, Lukes I, Prasad V, et al. PET/CT imaging of osteoblastic bone metastases with 68Ga-bisphosphonates: first human study. Eur J Nucl Med Mol Imaging. 2010;37:834.
Ayati N, Aryana K, Jalilian AR, Hoseinnejad T, Samani AB, Ayati Z, et al. Treatment efficacy of 153Sm-EDTMP for painful bone metastasis. Asia Oceania J Nucl Med Biol. 2013;1:27–31.
Bahrami-Samani A, Anvari A, Jalilian AR, Shirvani-Arani S, Yousefnia H, Aghamiri MR, et al. Production, quality control and pharmacokinetic studies of 177Lu-EDTMP for human bone pain palliation therapy trials. Iran J Pharm Res. 2012;11:137–44.
Mitterhauser M, Toegei S, Wadsak W, Lanzenberger RR, Mien LK, Kunter C, et al. Pre vivo, ex vivo and in vivo evaluations of [68Ga]-EDTMP. Nucl Med Biol. 2007;34:391–7.
Mirzaei A, Jalilian AR, Aghanejad A, Mazidi M, Yousefnia H, Shabani G, et al. Preparation and evaluation of 68Ga-ECC as a PET renal imaging agent. Nucl Med Mol Imag. 2015. doi:10.1007/s13139-015-0323-7.
Goeckeler WF, Troutner DE, Volkert WA, Edwards B, Simon J, Wilson D. 153Sm radiotherapeutic bone agents. Int J Rad Appl Instrum B. 1986;13:479–82.
Breeman WA, de Jong M, de Blois E, Bernard BF, Konijnenberg M, Krenning EP. Radiolabeling DOTA-peptides with 68Ga. Eur J Nucl Med Mol Imaging. 2005;32:478–85.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mirzaei, A., Jalilian, A.R., Badbarin, A. et al. Optimized production and quality control of 68Ga-EDTMP for small clinical trials. Ann Nucl Med 29, 506–511 (2015). https://doi.org/10.1007/s12149-015-0971-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-015-0971-9