Abstract
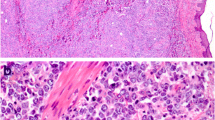
Primary lymphomas of the oral cavity are rare and the most frequent type is diffuse large B-cell lymphoma (DLBCL). Recently, several reports have highlighted the value of classifying DLBCL into prognostically important subgroups, namely germinal center B-cell like (GCB) and non-germinal center B-cell like (non-GCB) lymphomas based on gene expression profiles and by immunohistochemical expression of CD10, BCL6 and MUM-1. GCB lymphomas tend to exhibit a better prognosis than non-GCB lymphomas. Studies validating this classification have been done for DLBCL of the breast, CNS, testes and GI tract. Therefore we undertook this study to examine if primary oral DLBCLs reflect this trend. We identified 13 cases (age range 38–91 years) from our archives dating from 2003–09. IHC was performed using antibodies against germinal center markers (CD10, BCL6), activated B-cell markers (MUM1, BCL2) and Ki-67 (proliferation marker). Cases were sub-classified as GCB subgroup if CD10 and/or BCL6 were positive and MUM-1, was negative and as non-GCB subgroup if CD10 was negative and MUM-1 was positive. Immunoreactivity was noted in 2/13 cases for CD10, in 12/13 for BCL6, in 8/13 for MUM-1, and in 6/13 for BCL2. Therefore, 8/13 (58%) were sub-classified as non-GCB DLBCLs and 5/13 (42%) as GCB subgroup. All tumors showed frequent labeling with Ki-67 (range 40–95%). Four of the 8 patients with non-GCB subgroup succumbed to their disease, with the mean survival rate of 16 months. Two patients in this group are alive, one with no evidence of disease and another with disease. No information was available for the other 3 patients in this group. Four of the 5 patients in the GCB subgroup were alive with no evidence of disease and one patient succumbed to complications of therapy and recurrent disease after 18 months. In conclusion, our analysis shows that primary oral DLBCL predominantly belongs to the non-GCB subgroup, which tends to exhibit a poorer prognosis. These findings could allow pathologists to provide a more accurate insight into the potential aggressive behavior and poorer prognosis of these lymphomas.







Similar content being viewed by others
References
Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, et al. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th ed. Lyon: IARC Press; 2008.
Coiffier B. Diffuse large cell lymphoma. Curr Opin Oncol. 2001;13(5):325–34.
Kolokotronis A, Konstantinou N, Christakis I, Papadimitriou P, Matiakis A, Zaraboukas T, et al. Localized B-cell non-Hodgkin’s lymphoma of oral cavity and maxillofacial region: a clinical study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;99(3):303–10.
Solomides CC, Miller AS, Christman RA, Talwar J, Simpkins H. Lymphomas of the oral cavity: histology, immunologic type, and incidence of Epstein-Barr virus infection. Hum Pathol. 2002;33(2):153–7.
Wolvius EB, van der Valk P, van der Wal JE, van Diest PJ, Huijgens PC, van der Waal I, et al. Primary extranodal non-Hodgkin lymphoma of the oral cavity. an analysis of 34 cases. Eur J Cancer B Oral Oncol. 1994;30B(2):121–5.
Harris NL, Jaffe ES, Stein H, Banks PM, Chan JK, Cleary ML, et al. A revised European-American classification of lymphoid neoplasms: a proposal from the international lymphoma Study Group. Blood. 1994;84(5):1361–92.
Alacacioglu I, Ozcan MA, Ozkal S, Piskin O, Turgut N, Demirkan F, et al. Prognostic significance of immunohistochemical classification of diffuse large B-cell lymphoma. Hematology. 2009;14(2):84–9.
de Leval L, Harris NL. Variability in immunophenotype in diffuse large B-cell lymphoma and its clinical relevance. Histopathology. 2003;43(6):509–28.
Hans CP, Weisenburger DD, Greiner TC, Gascoyne RD, Delabie J, Ott G, et al. Confirmation of the molecular classification of diffuse large B-cell lymphoma by immunohistochemistry using a tissue microarray. Blood. 2004;103(1):275–82.
Alizadeh AA, Eisen MB, Davis RE, Ma C, Lossos IS, Rosenwald A, et al. Distinct types of diffuse large B-cell lymphoma identified by gene expression profiling. Nature. 2000;403(6769):503–11.
Lossos IS, Okada CY, Tibshirani R, Warnke R, Vose JM, Greiner TC, et al. Molecular analysis of immunoglobulin genes in diffuse large B-cell lymphomas. Blood. 2000;95(5):1797–803.
Shipp MA, Ross KN, Tamayo P, Weng AP, Kutok JL, Aguiar RC, et al. Diffuse large B-cell lymphoma outcome prediction by gene-expression profiling and supervised machine learning. Nat Med. 2002;8(1):68–74.
Rosenwald A, Wright G, Chan WC, Connors JM, Campo E, Fisher RI, et al. The use of molecular profiling to predict survival after chemotherapy for diffuse large-B-cell lymphoma. N Engl J Med. 2002;346(25):1937–47.
Barrans SL, O’Connor SJ, Evans PA, Davies FE, Owen RG, Haynes AP, et al. Rearrangement of the BCL6 locus at 3q27 is an independent poor prognostic factor in nodal diffuse large B-cell lymphoma. Br J Haematol. 2002;117(2):322–32.
Linderoth J, Jerkeman M, Cavallin-Stahl E, Kvaloy S, Torlakovic E. Immunohistochemical expression of CD23 and CD40 may identify prognostically favorable subgroups of diffuse large B-cell lymphoma: a nordic lymphoma group study. Clin Cancer Res. 2003;9(2):722–8.
Milanes-Yearsley M, Hammond ME, Pajak TF, Cooper JS, Chang C, Griffin T, et al. Tissue micro-array: a cost and time-effective method for correlative studies by regional and national cancer study groups. Mod Pathol. 2002;15(12):1366–73.
Dogan A, Bagdi E, Munson P, Isaacson PG. CD10 and BCL-6 expression in paraffin sections of normal lymphoid tissue and B-cell lymphomas. Am J Surg Pathol. 2000;24(6):846–52.
Lozada-Nur F, de Sanz S, Silverman S Jr, Miranda C, Regezi JA. Intraoral non-Hodgkin’s lymphoma in seven patients with acquired immunodeficiency syndrome. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1996;82(2):173–8.
Shima N, Kobashi Y, Tsutsui K, Ogawa K, Maetani S, Nakashima Y, et al. Extranodal non-Hodgkin’s lymphoma of the head and neck. A clinicopathologic study in the Kyoto-Nara area of Japan. Cancer. 1990;66(6):1190–7.
de Leval L, Braaten KM, Ancukiewicz M, Kiggundu E, Delaney T, Mankin HJ, et al. Diffuse large B-cell lymphoma of bone: an analysis of differentiation-associated antigens with clinical correlation. Am J Surg Pathol. 2003;27(9):1269–77.
Chang KL, Arber DA, Weiss LM. CD20: a review. Appl Immunohistochem. 1996;4(1):1–15.
Mohrmann RL, Arber DA. CD20-Positive peripheral T-cell lymphoma: report of a case after nodular sclerosis Hodgkin’s disease and review of the literature. Mod Pathol. 2000;13(11):1244–52.
Yao X, Teruya-Feldstein J, Raffeld M, Sorbara L, Jaffe ES. Peripheral T-cell lymphoma with aberrant expression of CD79a and CD20: a diagnostic pitfall. Mod Pathol. 2001;14(2):105–10.
Kaufmann O, Flath B, Spath-Schwalbe E, Possinger K, Dietel M. Immunohistochemical detection of CD10 with monoclonal antibody 56C6 on paraffin sections. Am J Clin Pathol. 1999;111(1):117–22.
Fabiani B, Delmer A, Lepage E, Guettier C, Petrella T, Briere J, et al. CD10 expression in diffuse large B-cell lymphomas does not influence survival. Virchows Arch. 2004;445(6):545–51.
Mason DY, Cordell J, Brown M, Pallesen G, Ralfkiaer E, Rothbard J, et al. Detection of T cells in paraffin wax embedded tissue using antibodies against a peptide sequence from the CD3 antigen. J Clin Pathol. 1989;42(11):1194–200.
Hall PA, Richards MA, Gregory WM, d’Ardenne AJ, Lister TA, Stansfeld AG. The prognostic value of Ki67 immunostaining in non-Hodgkin’s lymphoma. J Pathol. 1988;154(3):223–35.
Pezzella F, Tse AG, Cordell JL, Pulford KA, Gatter KC, Mason DY. Expression of the bcl-2 oncogene protein is not specific for the 14;18 chromosomal translocation. Am J Pathol. 1990;137(2):225–32.
Rantanen S, Monni O, Joensuu H, Franssila K, Knuutila S. Causes and consequences of BCL2 overexpression in diffuse large B-cell lymphoma. Leuk Lymphoma. 2001;42(5):1089–98.
Flenghi L, Bigerna B, Fizzotti M, Venturi S, Pasqualucci L, Pileri S, et al. Monoclonal antibodies PG-B6a and PG-B6p recognize, respectively, a highly conserved and a formol-resistant epitope on the human BCL-6 protein amino-terminal region. Am J Pathol. 1996;148(5):1543–55.
Ye BH, Lista F, Lo Coco F, Knowles DM, Offit K, Chaganti RS, et al. Alterations of a zinc finger-encoding gene, BCL-6, in diffuse large-cell lymphoma. Science. 1993;262(5134):747–50.
Braaten KM, Betensky RA, de Leval L, Okada Y, Hochberg FH, Louis DN, et al. BCL-6 expression predicts improved survival in patients with primary central nervous system lymphoma. Clin Cancer Res. 2003;9(3):1063–9.
Capello D, Vitolo U, Pasqualucci L, Quattrone S, Migliaretti G, Fassone L, et al. Distribution and pattern of BCL-6 mutations throughout the spectrum of B-cell neoplasia. Blood. 2000;95(2):651–9.
Falini B, Fizzotti M, Pucciarini A, Bigerna B, Marafioti T, Gambacorta M, et al. A monoclonal antibody (MUM1p) detects expression of the MUM1/IRF4 protein in a subset of germinal center B cells, plasma cells, and activated T cells. Blood. 2000;95(6):2084–92.
Al-Abbadi MA, Hattab EM, Tarawneh MS, Amr SS, Orazi A, Ulbright TM. Primary testicular diffuse large B-cell lymphoma belongs to the nongerminal center B-cell-like subgroup: a study of 18 cases. Mod Pathol. 2006;19(12):1521–7.
Yoshida S, Nakamura N, Sasaki Y, Yoshida S, Yasuda M, Sagara H, et al. Primary breast diffuse large B-cell lymphoma shows a non-germinal center B-cell phenotype. Mod Pathol. 2005;18(3):398–405.
Al-Quran SZ, Hameed O, Cardona DM, Jhala D, Li Y. Primary large B-Cell lymphomas of the breast: subclassification by immunophenotyping. Am J Clin Pathol. 2007;128(4):693–4.
Wu G, Keating A. Biomarkers of potential prognostic significance in diffuse large B-cell lymphoma. Cancer. 2006;106(2):247–57.
Jacobson JO, Wilkes BM, Kwaiatkowski DJ, Medeiros LJ, Aisenberg AC, Harris NL. bcl-2 rearrangements in de novo diffuse large cell lymphoma. association with distinctive clinical features. Cancer. 1993;72(1):231–6.
Weiss LM, Warnke RA, Sklar J, Cleary ML. Molecular analysis of the t(14;18) chromosomal translocation in malignant lymphomas. N Engl J Med. 1987;317(19):1185–9.
Shaffer AL, Yu X, He Y, Boldrick J, Chan EP, Staudt LM. BCL-6 represses genes that function in lymphocyte differentiation, inflammation, and cell cycle control. Immunity. 2000;13(2):199–212.
Bea S, Colomo L, Lopez-Guillermo A, Salaverria I, Puig X, Pinyol M, et al. Clinicopathologic significance and prognostic value of chromosomal imbalances in diffuse large B-cell lymphomas. J Clin Oncol. 2004;22(17):3498–506.
Berglund M, Enblad G, Flordal E, Lui WO, Backlin C, Thunberg U, et al. Chromosomal imbalances in diffuse large B-cell lymphoma detected by comparative genomic hybridization. Mod Pathol. 2002;15(8):807–16.
Tagawa H, Tsuzuki S, Suzuki R, Karnan S, Ota A, Kameoka Y, et al. Genome-wide array-based comparative genomic hybridization of diffuse large B-cell lymphoma: comparison between CD5-positive and CD5-negative cases. Cancer Res. 2004;64(17):5948–55.
A predictive model for aggressive non-Hodgkin’s lymphoma. The International Non-Hodgkin’s Lymphoma Prognostic Factors Project. N Engl J Med. 1993;329(14):987–94.
Shipp MA, Abeloff MD, Antman KH, Carroll G, Hagenbeek A, Loeffler M, et al. International consensus conference on high-dose therapy with hematopoietic stem cell transplantation in aggressive Non-Hodgkin’s lymphomas: report of the jury. J Clin Oncol. 1999;17(1):423–9.
Hattab EM, Martin SE, Al-Khatib SM, Kupsky WJ, Vance GH, Stohler RA, et al. Most primary central nervous system diffuse large B-cell lymphomas occurring in immunocompetent individuals belong to the nongerminal center subtype: a retrospective analysis of 31 cases. Mod Pathol. 2010;23(2):235–43.
Vose JM. Current approaches to the management of non-Hodgkin’s lymphoma. Semin Oncol. 1998;25(4):483–91.
Connors JM, Klimo P, Fairey RN, Voss N. Brief chemotherapy and involved field radiation therapy for limited-stage, histologically aggressive lymphoma. Ann Intern Med. 1987;107(1):25–30.
Cabanillas F, Hagemeister FB, McLaughlin P, Velasquez WS, Riggs S, Fuller L, et al. Results of MIME salvage regimen for recurrent or refractory lymphoma. J Clin Oncol. 1987;5(3):407–12.
Fisher RI, Gaynor ER, Dahlberg S, Oken MM, Grogan TM, Mize EM, et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin’s lymphoma. N Engl J Med. 1993;328(14):1002–6.
Natkunam Y, Farinha P, Hsi ED, Hans CP, Tibshirani R, Sehn LH, et al. LMO2 protein expression predicts survival in patients with diffuse large B-cell lymphoma treated with anthracycline-based chemotherapy with and without rituximab. J Clin Oncol. 2008;26(3):447–54.
Seki R, Ohshima K, Fujisaki T, Uike N, Kawano F, Gondo H, et al. Prognostic impact of immunohistochemical biomarkers in diffuse large B-cell lymphoma in the rituximab era. Cancer Sci. 2009;100(10):1842–7.
Acknowledgments
The authors would like to thank Ms. Elaine Dooley for her technical support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Both authors I. Bhattacharyya and H.K. Chehal have equal contribution in the study.
Rights and permissions
About this article
Cite this article
Bhattacharyya, I., Chehal, H.K., Cohen, D.M. et al. Primary Diffuse Large B-Cell Lymphoma of the Oral Cavity: Germinal Center Classification. Head and Neck Pathol 4, 181–191 (2010). https://doi.org/10.1007/s12105-010-0184-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12105-010-0184-4