Abstract
Objective
Great success has been achieved in CAR-T cell immunotherapy in the treatment of hematological tumors. However, it is particularly difficult in solid tumors, because CAR-T is difficult to enter interior and exert long-term stable immune effects. Dendritic cells (DCs) can not only present tumor antigens but also promote the infiltration of T cells. Therefore, CAR-T cells with the help of DC vaccines are a reliable approach to treat solid tumors.
Methods
To test whether DC vaccine could promote CAR-T cell therapy in solid tumors, DC vaccine was co-cultured with MSLN CAR-T cells. The in vitro effects of DC vaccine on CAR-T were assessed by measuring cell proliferation, cell differentiation, and cytokine secretion. Effects of DC vaccine on CAR-T were evaluated using mice with subcutaneous tumors in vivo. The infiltration of CAR-T was analyzed using immunofluorescence. The persistence of CAR-T in mouse blood was analyzed using real-time quantitative PCR.
Results
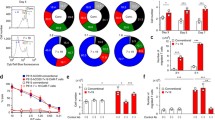
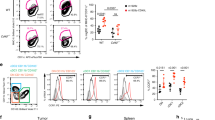
The results showed that DC vaccine significantly enhanced the proliferation potential of MSLN CAR-T cells in vitro. DC vaccines not only promoted the infiltration of CAR-T cells, but also significantly improved the persistence of CAR-T in solid tumors in vivo.
Conclusion
In conclusion, this study has demonstrated that DC vaccine can promote CAR-T therapy in solid tumors, which provides the possibility of widespread clinical application of CAR-T cells in the future.




Similar content being viewed by others
Data availability
The authors state that all data is available from the text or corresponding authors.
References
Zhang C, Liu J, Zhong JF, et al. Engineering CAR-T cells. Biomak Res. 2017. https://doi.org/10.1186/s40364-017-0102-y.
Cao J, Wang G, Cheng H, et al. Potent anti-leukemia activities of humanized CD19-targeted Chimeric antigen receptor T (CAR-T) cells in patients with relapsed/refractory acute lymphoblastic leukemia [J]. Am J Hematol. 2018;93(7):851–8.
Wei G, Ding L, Wang J, et al. Advances of CD19-directed chimeric antigen receptor-modified T cells in refractory/relapsed acute lymphoblastic leukemia. Exp Hematol Oncol. 2017. https://doi.org/10.1186/s40164-017-0070-9.
Han D, Xu Z, Zhuang Y, et al. Current progress in CAR-T cell therapy for hematological malignancies [J]. J Cancer. 2021;12(2):326–34.
Newick K, O’Brien S, Moon E, et al. CAR T Cell Therapy for Solid Tumors [M]//CASKEY C T. Annu Rev Med. 2017;68:139–52.
Dana H, Chalbatani GM, Jalali SA, et al. CAR-T cells: Early successes in blood cancer and challenges in solid tumors [J]. Acta Pharmaceutica Sinica B. 2021;11(5):1129–47.
Ma S, Li X, Wang X, et al. Current progress in CAR-T Cell therapy for solid tumors [J]. Int J Biol Sci. 2019;15(12):2548–60.
Sterner RC, Sterner RM. CAR-T cell therapy: current limitations and potential strategies. Blood Cancer J. 2021. https://doi.org/10.1038/s41408-021-00459-7.
Bol KF, Schreibelt G, Rabold K, et al. The clinical application of cancer immunotherapy based on naturally circulating dendritic cells [J]. J Immunother Cancer. 2019. https://doi.org/10.1186/s40425-019-0580-.
Jhunjhunwala S, Hammer C, Delamarre L. Antigen presentation in cancer: insights into tumour immunogenicity and immune evasion [J]. Nat Rev Cancer. 2021;21(5):298–312.
Macri C, Pang ES, Patton T, et al. Dendritic cell subsets [J]. Semin Cell Dev Biol. 2018;84:11–21.
Randolph GJ. Dendritic cells: The first step [J]. J Exp Med. 2021. https://doi.org/10.1084/jem.20202077.
Balan S, Saxena M, Bhardwaj N. Dendritic cell subsets and locations [M]//LHUILLIER C. GALLUZZI L. Immunobiology of Dendritic Cells: Pt A; 2019. p. 1–68.
Osada T, Clay TM, Woo CY, et al. Dendritic cell-based immunotherapy. Int Rev Immunol. 2006;25(5–6):377–413.
Patente TA, Pinho MP, Oliveira AA, et al. Human dendritic cells: their heterogeneity and clinical application potential in cancer immunotherapy. Front Immun. 2019. https://doi.org/10.3389/fimmu.2018.03176.
Fu C, Jiang A. Dendritic cells and CD8 T cell immunity in tumor microenvironment. Front Immunol. 2018. https://doi.org/10.3389/fimmu.2018.03059.
Spranger S, Dai D, Horton B, et al. Tumor-Residing Batf3 dendritic cells are required for effector t cell trafficking and adoptive t cell therapy. Cancer Cell. 2017. https://doi.org/10.1016/j.ccell.2017.04.003.
Gardner A, DE MINGO PULIDO A, RUFFELL B. Dendritic Cells and Their Role in Immunotherapy. Front Immunol. 2020. https://doi.org/10.3389/fimmu.2020.00924.
Filin IY, Kitaeva KV, Rutland CS, et al. Recent advances in experimental dendritic cell vaccines for cancer. Front Oncology. 2021. https://doi.org/10.3389/fonc.2021.730824.
Nava S, Lisini D, Frigerio S, et al. Dendritic cells and cancer immunotherapy: the adjuvant effect. Int J Molecular Sci. 2021;22(22):12339.
Lau SP, Klaase L, Vink M, et al. Autologous dendritic cells pulsed with allogeneic tumour cell lysate induce tumour-reactive T-cell responses in patients with pancreatic cancer: a phase I study. Eur J Cancer. 2022;169:20–31.
van Driessche A, van de Velde ALR, Nijs G, et al. Clinical-grade manufacturing of autologous mature mRNA-electroporated dendritic cells and safety testing in acute myeloid leukemia patients in a phase I dose-escalation clinical trial. Cytotherapy. 2009;11(5):653–68.
Yi DH, Appel S. Current status and future perspectives of dendritic cell-based cancer immunotherapy. Scand J Immunol. 2013;78(2):167–71.
Bol KF, Schreibelt G, Gerritsen WR, et al. Dendritic cell-based immunotherapy: state of the art and beyond. Clin Cancer Res. 2016;22(8):1897–906.
Chen J, Hu J, Gu L, et al. Anti-mesothelin CAR-T immunotherapy in patients with ovarian cancer. Cancer Immunol Immunother. 2022. https://doi.org/10.1007/s00262-022-03238-w.
Sommermeyer D, Hudecek M, Kosasih PL, et al. Chimeric antigen receptor-modified T cells derived from defined CD8(+) and CD4(+) subsets confer superior antitumor reactivity in vivo. Leukemia. 2016;30(2):492–500.
Turtle CJ, HANAFI L-A, BERGER C, et al. CD19 CAR-T cells of defined CD4(+): CD8(+) composition in adult B cell ALL patients. J Clin Invest. 2016;126(6):2123–38.
Turtle CJ, HANAFI L-A, BERGER C, et al. Immunotherapy of non-Hodgkin’s lymphoma with a defined ratio of CD8(+) and CD4(+) CD19-specific chimeric antigen receptor-modified T cells. Sci Translat Med. 2016. https://doi.org/10.1126/scitranslmed.aaf8621.
Fisher PJ, Bulur PA, Vuk-Pavlovic S, et al. Dendritic cell microvilli: a novel membrane structure associated with the multifocal synapse and T-cell clustering. Blood. 2008;112(13):5037–45.
Kim MK, Kim J. Properties of immature and mature dendritic cells: phenotype, morphology, phagocytosis, and migration. RSC Adv. 2019;9(20):11230–8.
O’Neill DW, Adams S, Bhardwaj N. Manipulating dendritic cell biology for the active immunotherapy of cancer. Blood. 2004;104(8):2235–46.
Nguyen KG, Vrabel MR, Mantooth SM, et al. Localized Interleukin-12 for Cancer Immunotherapy. Front Immunol. 2020. https://doi.org/10.3389/fimmu.2020.575597.
Wu M, Zhang L, Zhang H, et al. CD19 chimeric antigen receptor-redirected T cells combined with epidermal growth factor receptor pathway substrate 8 peptide-derived dendritic cell vaccine in leukemia. Cytotherapy. 2019;21(6):659–70.
Golubovskaya V, Wu L. Different Subsets of T Cells, memory, effector functions, and CAR-T immunotherapy. Cancers. 2016;8(3):36.
Martinez M, Moon EK. CAR T cells for solid tumors: new strategies for finding, infiltrating, and surviving in the tumor microenvironment. Front Immunol. 2019. https://doi.org/10.3389/fimmu.2019.00128.
Delgoffe GM, Xu C, Mackall CL, et al. The role of exhaustion in CAR T cell therapy. Cancer Cell. 2021;39(7):885–8.
Perez CR, de Palma M. Engineering dendritic cell vaccines to improve cancer immunotherapy. Nat Commun. 2019. https://doi.org/10.1038/s41467-019-13368-y.
Acknowledgements
Thanks to all the partners who were helpful to the article, the guidance provided by the teacher, the platform provided by the school, and the funding from the state.
Funding
National Natural Science Foundation of China, 81872284, Miaomiao Zhang
Author information
Authors and Affiliations
Contributions
MZ, YW and LH proposed the research direction and designed the experimental scheme. MZ, YW, XC, FZ, JC, and HZ organized and analyzed the data. MZ, YW, XC, FZ, JC, HZ, JL2, and ZC2 interpreted the data; MZ, YW, and XC wrote and revised the manuscript; MZ, YW, XC, FZ, JC, HZ, JL2, ZC2, AW, YX, ZC4, YD, XY, FJ, JL1, JL5, FP, ZG, and LH provided funding and platforms for the experiments. LH supervised the study.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All in vivo animal experiments were approved by the Committee on the Ethics of Animal Experiments of Nanjing Normal University (IRB#2020–0047).
Informed consent
For this type of study informed consent is not required.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, M., Wang, Y., Chen, X. et al. DC vaccine enhances CAR-T cell antitumor activity by overcoming T cell exhaustion and promoting T cell infiltration in solid tumors. Clin Transl Oncol 25, 2972–2982 (2023). https://doi.org/10.1007/s12094-023-03161-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-023-03161-1