Abstract
Background
Patients with prostate-specific antigen (PSA) persistence are at the increased risk of disease progression. The aim of our study was to evaluate the impact of early salvage therapy on oncological outcomes in patients with persistent PSA after radical prostatectomy (RP).
Methods
Within a single tertiary centre database, we identified men with persistent (≥ 0.1 ng/ml) versus undetectable (< 0.1 ng/ml) PSA 4–8 weeks after RP for high-risk prostate cancer (HRPCa). The cumulative incidence function was used to estimate cancer-specific survival (CSS) and clinical progression-free survival (CPFS). The Kaplan–Meier method was used to estimate overall survival (OS). The effects on oncological outcomes of salvage radiotherapy (SRT) ± androgen deprivation therapy (ADT) vs. ADT monotherapy were tested in the subgroup of patients with persistent PSA.
Results
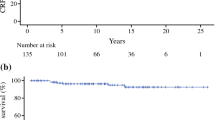
Of 414 consecutive patients who underwent RP for HRPC, 125 (30.2%) had persistent PSA. Estimated 10-year CPFS, CSS and OS for men with persistent vs. undetectable PSA were 63.8% vs. 93.5%, 78.5% vs. 98.3% and 54% vs. 83.2% (all p < 0.0001), respectively. In men with persistent PSA, ADT alone was associated with higher risk (hazard ratio (HR) for worse CSS (HR 3.9, p = 0.005) and OS (HR 4.7, p < 0.0001) but not for CP (HR 1.6, p = 0.2) when compared with SRT ± ADT.
Conclusion
In patients who underwent RP for HRPCa, persistent PSA was associated with poor oncological outcomes. Early SRT ± ADT resulted in significantly improved CSS and OS in men with persistent PSA comparing with early androgen deprivation monotherapy.


Similar content being viewed by others
References
Moris L, Cumberbatch MG, Van Den Broeck T, et al. Benefits and risks of primary treatments for high-risk localized and locally advanced prostate cancer: an international multidisciplinary systematic review. Eur Urol. 2020;77(5):614–27. https://doi.org/10.1016/j.eururo.2020.01.033.
Joniau SG, Van Baelen AA, Hsu CY, et al. Complications and functional results of surgery for locally advanced prostate cancer. Adv Urol. 2012;2012: 706309. https://doi.org/10.1155/2012/706309.
Milonas D, Baltrimavicius R, Grybas A, et al. Outcome of surgery in locally advanced pT3a prostate cancer. Cent Eur J Urol. 2011;64(4):209–12. https://doi.org/10.5173/ceju.2011.04.art4.
Mottet N, Bellmunt J, Bolla M, et al. EAU-ESTRO-SIOG guidelines on prostate cancer. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2017;71(4):618–29. https://doi.org/10.1016/j.eururo.2016.08.003.
Preisser F, Chun FKH, Pompe RS, et al. Persistent prostate-specific antigen after radical prostatectomy and its impact on oncologic outcomes. Eur Urol. 2019;76(1):106–14. https://doi.org/10.1016/j.eururo.2019.01.048.
Bianchi L, Nini A, Bianchi M, et al. The role of prostate-specific antigen persistence after radical prostatectomy for the prediction of clinical progression and cancer-specific mortality in node-positive prostate cancer patients. Eur Urol. 2016;69(6):1142–8. https://doi.org/10.1016/j.eururo.2015.12.010.
Kumar A, Samavedi S, Mouraviev V, et al. Predictive factors and oncological outcomes of persistently elevated prostate-specific antigen in patients following robot-assisted radical prostatectomy. J Robot Surg. 2017;11(1):37–45. https://doi.org/10.1007/s11701-016-0606-8.
Bartkowiak D, Siegmann A, Böhmer D, et al. The impact of prostate-specific antigen persistence after radical prostatectomy on the efficacy of salvage radiotherapy in patients with primary N0 prostate cancer. BJU Int. 2019;124(5):785–91. https://doi.org/10.1111/bju.14851.
Audenet F, Seringe E, Drouin SJ, et al. Persistently elevated prostate-specific antigen at six weeks after radical prostatectomy helps in early identification of patients who are likely to recur. World J Urol. 2012;30(2):239–44. https://doi.org/10.1007/s00345-011-0707-y.
Wiegel T, Bartkowiak D, Bottke D, et al. Prostate-specific antigen persistence after radical prostatectomy as a predictive factor of clinical relapse-free survival and overall survival: 10-year data of the ARO 96–02 trial. Int J Radiat Oncol Biol Phys. 2015;91(2):288–94. https://doi.org/10.1016/j.ijrobp.2014.09.039.
Ploussard G, Staerman F, Pierrevelcin J, et al. Predictive factors of oncologic outcomes in patients who do not achieve undetectable prostate specific antigen after radical prostatectomy. J Urol. 2013;190(5):1750–6. https://doi.org/10.1016/j.juro.2013.04.073.
Fossati N, Karnes RJ, Colicchia M, et al. Impact of early salvage radiation therapy in patients with persistently elevated or rising prostate-specific antigen after radical prostatectomy. Eur Urol. 2018;73(3):436–44. https://doi.org/10.1016/j.eururo.2017.07.026.
Gandaglia G, Boorjian SA, Parker WP, et al. Impact of postoperative radiotherapy in men with persistently elevated prostate-specific antigen after radical prostatectomy for prostate cancer: a long-term survival analysis. Eur Urol. 2017;72(6):910–7. https://doi.org/10.1016/j.eururo.2017.06.001.
McDonald ML, Howard LE, Aronson WJ, et al. First postoperative PSA is associated with outcomes in patients with node positive prostate cancer: Results from the SEARCH database. Urol Oncol. 2018;36(5):239 e217-239 e225. https://doi.org/10.1016/j.urolonc.2018.01.005.
Stephenson AJ, Scardino PT, Kattan MW, et al. Predicting the outcome of salvage radiation therapy for recurrent prostate cancer after radical prostatectomy. J Clin Oncol. 2007;25(15):2035–41. https://doi.org/10.1200/JCO.2006.08.9607.
Briganti A, Karnes RJ, Joniau S, et al. Prediction of outcome following early salvage radiotherapy among patients with biochemical recurrence after radical prostatectomy. Eur Urol. 2014;66(3):479–86. https://doi.org/10.1016/j.eururo.2013.11.045.
Tendulkar RD, Agrawal S, Gao T, et al. Contemporary update of a multi-institutional predictive nomogram for salvage radiotherapy after radical prostatectomy. J Clin Oncol. 2016;34(30):3648–54. https://doi.org/10.1200/JCO.2016.67.9647.
Fossati N, Karnes RJ, Cozzarini C, et al. Assessing the optimal timing for early salvage radiation therapy in patients with prostate-specific antigen rise after radical prostatectomy. Eur Urol. 2016;69(4):728–33. https://doi.org/10.1016/j.eururo.2015.10.009.
Schmidt-Hegemann NS, Fendler W, Ilhan H, et al. Outcome after PSMA PET/CT based radiotherapy in patients with biochemical persistence or recurrence after radical prostatectomy. Radiat Oncol. 2018;13:37. https://doi.org/10.1186/s13014-018-0983-4.
Sood A, Keeley J, Palma-Zamora I, et al. Ten-year disease progression and mortality rates in men who experience biochemical recurrence versus persistence after radical prostatectomy and undergo salvage radiation therapy: A post-hoc analysis of RTOG 9601 trial data. Urol Oncol. 2020;38(6):599 e591-599 e598. https://doi.org/10.1016/j.urolonc.2020.02.024.
Kimura S, Urabe F, Sasaki H, et al. Prognostic significance of prostate-specific antigen persistence after radical prostatectomy: a systematic review and meta-analysis. Cancers (Basel). 2021. https://doi.org/10.3390/cancers13050948.
Ploussard G, Fossati N, Wiegel T, et al. Management of persistently elevated prostate-specific antigen after radical prostatectomy: a systematic review of the literature. Eur Urol Oncol. 2021;4(2):150–69. https://doi.org/10.1016/j.euo.2021.01.001.
Van den Broeck T, van den Bergh RCN, Arfi N, et al. Prognostic value of biochemical recurrence following treatment with curative intent for prostate cancer: a systematic review. Eur Urol. 2019;75(6):967–87. https://doi.org/10.1016/j.eururo.2018.10.011.
Venclovas Z, Jievaltas M, Milonas D. Significance of time until psa recurrence after radical prostatectomy without neo- or adjuvant treatment to clinical progression and cancer-related death in high-risk prostate cancer patients. Front Oncol. 2019;9:1286. https://doi.org/10.3389/fonc.2019.01286.
Xiang C, Liu X, Chen S, et al. Prediction of biochemical recurrence following radiotherapy among patients with persistent PSA after radical prostatectomy: a single-center experience. Urol Int. 2018;101(1):47–55. https://doi.org/10.1159/000488536.
Acknowledgements
Steven Joniau is Senior Clinical Researcher of the FWO (Research Foundation Flanders)
Funding
No funding. The authors have no financial interest.
Author information
Authors and Affiliations
Contributions
DM: project development, data collection, data analysis, manuscript writing; AL: data analysis, manuscript editing; ZV: data collection, manuscript editing; LJ: data collection, manuscript editing; GD: manuscript editing; SJ: project development, manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Ethical approval
All procedures in this study involving human participants were in accordance with the ethical standards of the institutional committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Data collection and analysis was approved by the University ethical committee (BE-2–48).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Milonas, D., Laenen, A., Venclovas, Z. et al. Benefits of early salvage therapy on oncological outcomes in high-risk prostate cancer with persistent PSA after radical prostatectomy. Clin Transl Oncol 24, 371–378 (2022). https://doi.org/10.1007/s12094-021-02700-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-021-02700-y