Abstract
Background
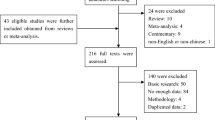
The specific association between PTEN deletion or ERG rearrangement and the recurrence of prostate cancer (PC) treated with radical prostatectomy (RP) or brachytherapy is still unelaborated. Therefore, we performed a comprehensive meta‑analysis to understand the impact of these factors on cancer recurrence.
Methods
A comprehensive literature search was performed in November 2018 based on PUBMED, EMBASE and Web of science database. Hazard ratio (HR) for biochemical recurrence free (BRF) which was defined as a PSA greater than or equal to 0.4 ng/mL after RP or another therapy for any detectable PSA and recurrence-free survival (RFS) which defined the time from the beginning of treatment to the earliest occurrence of local recurrence, distant metastasis or death. Which were extracted from eligible studies. I2 value was used to assess the pooled heterogeneity.
Result
A total of 6744 patients from 17 studies were included in this analysis Overall, The pooled results showed that PTEN loss predict pooled BRF (HR 1.79, 95% CI 1.49–2.16, P < 0.001) and RFS (HR 1.71, 95% CI 1.50–1.95, P < 0.001) in patients after radical prostatectomy or brachytherapy for prostate cancer. Subgroup analysis revealed that PTEN deletion significantly predicted poor BRF or RFS in heterozygous studies group (HR 1.70, 95% CI 1.31–2.21, P < 0.001). The PTEN deletion also significantly predicted poor BRF or PFS in homozygous studies (HR 2.54, 95% CI 1.89–3.17, P < 0.001). And we had found that there was no significant association between ERG rearrangement and cancer recurrence regardless of PTEN loss or not. In addition, we concluded that Gleason score > 6 significantly predicted the poor BRF or RFS in studies, especially in Gleason score = 4 + 3 (HR 3.16, 95% CI 2.08–4.80, P < 0.01).
Conclusion
This study presented that PTEN deletion significantly reduce time of BRF or RFS, especially based on homozygous deletion. And we also found ERG rearrangement in tumor cell could not significantly predict BRF or RFS.







Similar content being viewed by others
Abbreviations
- BRF:
-
Biochemical recurrence free
- RFS:
-
Recurrence-free survival
- RP:
-
Radical prostatectomy
- CIs:
-
Confidence intervals
- HRs:
-
Hazard ratios
- NOS:
-
Newcastle–Ottawa Scale
- PC:
-
Prostate cancer
References
Ferlay J, et al. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127(12):2893–917.
Hollander MC, Blumenthal GM, Dennis PA. PTEN loss in the continuum of common cancers, rare syndromes and mouse models. Nat Rev Cancer. 2011;11(4):289–301.
Saal LH, et al. Poor prognosis in carcinoma is associated with a gene expression signature of aberrant PTEN tumor suppressor pathway activity. Proc Natl Acad Sci. 2007;104(18):7564.
Jiang B, Liu L. PI3K/PTEN signaling in tumorigenesis and angiogenesis. Biochim et Biophys Acta Proteins Proteom. 2008;1784(1):150–8.
Yoshimoto M, et al. FISH analysis of 107 prostate cancers shows that PTEN genomic deletion is associated with poor clinical outcome. Br J Cancer. 2007;97(5):678–85.
Dehm SM. Distinct classes of chromosomal rearrangements create oncogenic ETS gene fusions in prostate cancer: Tomlins SA, Laxman B, Dhanasekaran SM, Helgeson BE, Cao X, Morris DS, Menon A, Jing X, Cao Q, Han B, Yu J, Wang L, Montie JE, Rubin MA, Pienta KJ, Roulston D, Shah RB, Varambally S, Mehra R, Chinnaiyan AM, Michigan Center for Translational Pathology, Department of Pathology, University of Michigan Medical School, Ann Arbor, MI. Urol Oncol. 2008;26(6):0–688.
Tomlins SA, Rhodes DR, Perner S, et al. Recurrent fusion of TMPRSS2 and ETS transcription factor genes in prostate cancer. J Urol. 2006;175(5):1707.
Higgins JPT, Thompson SG. Quantifying heterogeneity in a meta‐analysis. Stat Med. 2002;21(11):1539–58.
Bax L, et al. More than numbers: the power of graphs in meta-analysis. Am J Epidemiol. 2009;169(2):249–55.
Seagroatt V, Stratton I. Bias in meta-analysis detected by a simple, graphical test. Test had 10% false positive rate. BMJ. 1997;316(7129):469–71.
Troyer DA, et al. A multicenter study shows PTEN deletion is strongly associated with seminal vesicle involvement and extracapsular extension in localized prostate cancer. Prostate. 2015;75(11):1206–15.
Mehra R, et al. Association of ERG/PTEN status with biochemical recurrence after radical prostatectomy for clinically localized prostate cancer. Med Oncol. 2018;35(12):152.
Leapman MS, et al. Comparing prognostic utility of a single-marker immunohistochemistry approach with commercial gene expression profiling following radical prostatectomy. Eur Urol. 2018;74(5):668–75.
Zafarana G, et al. Copy number alterations of c-MYC and PTEN are prognostic factors for relapse after prostate cancer radiotherapy. Cancer. 2012;118(16):4053–62.
Barnett CM, et al. Genetic profiling to determine risk of relapse-free survival in high-risk localized prostate cancer. Clin Cancer Res. 2014;20(5):1306–12.
Krohn A, et al. Genomic deletion of PTEN is associated with tumor progression and early PSA recurrence in ERG fusion-positive and fusion-negative prostate cancer. Am J Pathol. 2012;181(2):401–12.
Breen KJ, et al. Investigating the role of the IGF axis as a predictor of biochemical recurrence in prostate cancer patients post-surgery. Prostate. 2017;77(12):1288–300.
Kim SH, et al. Overexpression of ERG and wild-type PTEN are associated with favorable clinical prognosis and low biochemical recurrence in prostate cancer. PLoS ONE. 2015;10(4):e0122498.
Tosoian JJ, et al. Prevalence and prognostic significance of PTEN loss in African–American and European–American men undergoing radical prostatectomy. Eur Urol. 2017;71(5):697–700.
Choucair K, et al. PTEN genomic deletion predicts prostate cancer recurrence and is associated with low AR expression and transcriptional activity. BMC cancer. 2012;12(1):543–543.
Lotan TL, et al. PTEN Loss as determined by clinical-grade immunohistochemistry assay is associated with worse recurrence-free survival in prostate cancer. Eur Urol Focus. 2016;2(2):180–8.
Mithal P, et al. PTEN loss in biopsy tissue predicts poor clinical outcomes in prostate cancer. Int J Urol. 2014;21(12):1209–14.
Geybels MS, et al. PTEN loss is associated with prostate cancer recurrence and alterations in tumor DNA methylation profiles. Oncotarget. 2017;8(48):84338.
Fontugne J, et al. Recurrent prostate cancer genomic alterations predict response to brachytherapy treatment. Cancer Epidemiol Biomark Prev. 2014;23(4):594–600.
Magi-Galluzzi C, et al. The 17-gene genomic prostate score assay predicts outcome after radical prostatectomy independent of PTEN status. Urology. 2018;121:132–8.
Bramhecha, Y.M., et al., The combination of PTEN deletion and 16p13.3 gain in prostate cancer provides additional prognostic information in patients treated with radical prostatectomy. Mod Pathol. 2019;32(1):128–38.
Ishioka J, et al. Development of a nomogram incorporating serum C-reactive protein level to predict overall survival of patients with advanced urothelial carcinoma and its evaluation by decision curve analysis. Br J Cancer. 2012;107(7):1031–6.
Stambolic V, et al. Negative regulation of PKB/Akt-dependent cell survival by the tumor suppressor PTEN. Cell. 1998;95(1):29–39.
Acknowledgements
We owe our thanks to Shuai Xia and Tao Li for their work on revising and data extracting in this manuscript.
Author information
Authors and Affiliations
Contributions
Conception and Design: JZ and DF; Extraction of Data: TL; Quality assessment: JZ and SX; Drafting the Article: JZ and DF; Revising It for Intellectual Content: RL; Final Approval of the Completed Article: JZ, RL. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Consent for publication
All authors consent this article for publication.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, R., Zhou, J., Xia, S. et al. The impact of PTEN deletion and ERG rearrangement on recurrence after treatment for prostate cancer: a systematic review and meta-analysis. Clin Transl Oncol 22, 694–702 (2020). https://doi.org/10.1007/s12094-019-02170-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-019-02170-3