Abstract
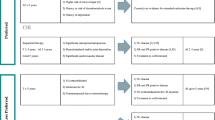
Adjuvant endocrine therapy is the most important systemic treatment for postmenopausal women with hormone-receptor-positive early breast cancer following surgery. Most trials have shown that the third-generation aromatase inhibitors, anastrozole, letrozole (LET), and exemestane, significantly prolong disease-free survival compared with tamoxifen. However, an overall survival benefit with aromatase inhibitors was observed in only three trials to date, in retrospective analyses from selected groups of patients: sequential analysis in the Austrian Breast & Colorectal Study Group 8, switch analysis in the Intergroup Exemestane Study, and analysis of initial letrozole in Breast International Group 1–98 study. Although the priming effect observed in preclinical models and breast cancer patients provides a rationale for sequencing adjuvant endocrine treatment with tamoxifen and aromatase inhibitors, the optimal strategy for adjuvant endocrine treatment has yet to be determined. This review discusses aromatase inhibitor monotherapy, sequential adjuvant treatment with tamoxifen followed by an aromatase inhibitor, and sequential adjuvant treatment with an aromatase inhibitor followed by tamoxifen. Available data in support of and against each strategy is evaluated. The safety profiles of tamoxifen and aromatase inhibitors is also examined.
Similar content being viewed by others
References
Parkin DM, Bray F, Ferlay J, Pisani P. Global cancer statistics, 2002. CA Cancer J Clin. 2005; 55:74–108.
Li CI, Daling JR, Malone KE. Incidence of invasive breast cancer by hormone receptor status from 1992 to 1998. J Clin Oncol. 2003;21:28–34.
Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet 2005;365:1687–717.
Dorssers LC, Van der Flier S, Brinkman A, et al. Tamoxifen resistance in breast cancer: elucidating mechanisms. Drugs. 2001;61(12):1721–33.
Brodie A, Jelovac D, Long BJ. Predictions from a preclinical model: studies of aromatase inhibitors and antiestrogens. Clin Cancer Res. 2003;9:455S–459S.
Forbes JF, Cuzick J, Buzdar A, et al: Effect of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: 100-month analysis of the ATAC trial. Lancet Oncol 2008;9:45–53.
Mouridsen H, Giobbie-Hurder A, Goldhirsch A, et al. Letrozole therapy alone or in sequence with tamoxifen in women with breast cancer. BIG 1-98 Collaborative Group, N Engl J Med. 2009;361:766–776.
Rea D, Hasenburg A, Seynaeve C, et al. Five Years of Exemestane as Initial Therapy Compared to 5 Years of Tamoxifen Followed by Exemestane: The TEAM Trial, a Prospective, Randomized, Phase III Trial in Postmenopausal Women with Hormone-Sensitive Early Breast Cancer. Presented at SABCS 2009. Abstract 11.
Coombes R, Kilburn L, Beare S, Snowdon C, Bliss J. Survival and safety post study treatment completion: an updated analysis of the Intergroup Exemestane Study (IES) — submitted on behalf of the IES Investigators. Eur J Cancer Supplements 2009;2:p 263.
Jakesz R, Jonat W, Gnant M, et al: Switching of postmenopausal women with endocrine-responsive early breast cancer to anastrozole after 2 years’ adjuvant tamoxifen: combined results of ABCSG trial 8 and ARNO 95 trial. Lancet 2005;366:455–462.
Boccardo F, Rubagotti A, Guglielmini P, et al: Switching to anastrozole vs continued tamoxifen treatment of early breast cancer. Updated results of the Italian tamoxifen anastrozole (ITA) trial. Ann Oncol. 2006;17:vii10–vii14.
Brodie AH, Jelovac D, Long B. The intratumoral aromatase model: studies with aromatase inhibitors and antiestrogens. J Steroid Biochem Mol Biol. 2003;86:283–288.
Mouridsen H, Gershanovich M, Sun Y, et al. Phase III study of letrozole vs tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the International Letrozole Breast Cancer Group. J Clin Oncol. 2003;21:2101–2109.
Mansell J, Monypenny IJ, Skene AI, et al Patterns and predictors of early recurrence in postmenopausal women with estrogen receptor-positive early breast cancer. Breast Cancer Res Treat. 2009;117:91–8.
Houghton J. Initial adjuvant therapy with anastrozole reduces rates of early breast cancer recurrence and adverse events compared with tamoxifen — data reported on behalf of the ATAC (Arimidex, tamoxifen, alone or in Combination) Trialists’ group. Ann Oncol. 2006; 17(Suppl. 9): ix94. Abstract 243 PD.
Mauriac L, Keshaviah A, Debled M, et al; BIG 1-98 Collaborative Group; International Breast Cancer Study Group. Predictors of early relapse in postmenopausal women with hormone receptorpositive breast cancer in the BIG 1-98 trial. Ann Oncol. 2007;18: 859–867.
Lê MG, Arriagada R, Spielmann M, et al. Prognostic factors for death after an isolated local recurrence in patients with early-stage breast carcinoma. Cancer. 2002; 94: 2813–2820.
Hudis CA, Barlow WE, Costantino JP, et al: Proposal for standardized definitions for efficacy end points in adjuvant breast cancer trials: the STEEP system. J Clin Oncol 2007; 25: 2127–2132.
Howell A, Cuzick J, Baum M, et al.; ATAC Trialists’ Group. Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years’ adjuvant treatment for breast cancer. Lancet 2005; 365: 60–62.
Breast International Group (BIG) 1-98 Collaborative Group, Thürlimann B, Keshaviah A, Coates AS, Mouridsen H, Mauriac L, Forbes JF, Paridaens R, Castiglione-Gertsch M, Gelber RD, Rabaglio M, Smith I, Wardley A, Price KN, Goldhirsch A. A comparison of letrozole and tamoxifen in postmenopausal women with early breast cancer. N Engl J Med. 2005;353: 2747–57.
Fisher B, Costantino J, Redmond C, et al. A randomized clinical trial evaluating tamoxifen in the treatment of patients with node-negative breast cancer who have estrogen-receptor-positive tumors. N Engl J Med 1989; 320: 479–484.
Rutqvist LE, Johansson H; Stockholm Breast Cancer Study Group. Long-term follow-up of the randomized Stockholm trial on adjuvant tamoxifen among postmenopausal patients with early stage breast cancer. Acta Oncol 2007; 46: 133–145.
Regan MM, Colleoni M, Giobbie-Hurder A, et al. Adjusting for Selective Crossover in Analyses of Letrozole (Let) Versus Tamoxifen (Tam) in the BIG 1-98 Trial. Presented at: San Antonio Breast Cancer Symposium, December 9–13, 2009, San Antonio, TX [abstract 16].
Jones SE, Seynaeve C, Hasenburg A, et al: Results of the first planned analysis of the TEAM (tamoxifen exemestane adjuvant multinational) prospective randomized phase III trial in hormone sensitive postmenopausal early breast cancer. Cancer Res 69(Suppl 2): Abstract no 15, 2009.
Coombes RC, Hall E, Gibson LJ, et al; Intergroup Exemestane Study. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med. 2004;350:1081–1092.
Coombes RC, Kilburn LS, Snowdon CF, et al; Intergroup Exemestane Study. Survival and safety of exemestane vs tamoxifen after 2–3 years’ tamoxifen treatment (Intergroup Exemestane Study): a randomised controlled trial. Lancet. 2007;369:559–570.
Jakesz R, Gnant M, Greil R, et al. The benefits of sequencing adjuvant tamoxifen and anastrozole in postmenopausal women with hormone-responsive early breast cancer: 5 year-analysis of ABCSG Trial 8. Breast Cancer Res Treat 2005;94(Suppl 1):S10 [abstract 13].
Dowsett M, Allred C, Knox J et al. Relationship between quantitative estrogen and progesterone receptor expression and human epidermal growth factor receptor 2 (HER2) status with recurrence in the Arimidex, Tamoxifen, alone or in combination trial. J Clin Oncol 2008; 26: 1059–1065.
Rasmussen BB, Regan MM, Lykkesfeldt AE, et al. Adjuvant letrozole vs tamoxifen according to centrally-assessed ERBB2 status for postmenopausal women with endocrine-responsive early breast cancer: supplementary results from the BIG 1-98 randomized trial. Lancet Oncol 2008; 9: 23–28.
Gradishar WJ. Safety considerations of adjuvant therapy in early breast cancer in postmenopausal women. Oncology 2005; 69: 1–9.
Conte P, Frassoldati A. Aromatase inhibitors in the adjuvant treatment of postmenopausal women with early breast cancer: Putting safety issues into perspective. Breast J 2007; 13: 28–35.
Perez EA. Safety profiles of tamoxifen and the aromatase inhibitors in adjuvant therapy of hormone-responsive early breast cancer. Ann Oncol 2007; 18(Suppl 8): viii26–35.
Thürlimann B; for the BIG 1-98 Collaborative & International Breast Cancer Study Groups. Letrozole vs tamoxifen as adjuvant endocrine therapy for postmenopausal women with receptor-positive breast cancer: update of the BIG 1–98 Primary Core Analysis (PCA). Primary Therapy of Early Breast Cancer 2009, 11th International Conference, March 11–14, St. Gallen, Switzerland [abstract 0161].
Fallowfield L. Quality of life issues in relation to the aromatase inhibitor. J Steroid Biochem Mol Biol 2007; 106:168–172.
Viale G, Giobbie-Hurder A, Regan MM, et al Breast International Group Trial 1–98. Prognostic and predictive value of centrally reviewed Ki-67 labeling index in postmenopausal women with endocrine-responsive breast cancer: results from Breast International Group Trial 1–98 comparing adjuvant tamoxifen with letrozole. J Clin Oncol 2008; 26: 5569–5575.
Arimidex, Tamoxifen, Alone or in Combination Trialists’ Group, Buzdar A, Howell A, Cuzick J, et al. Comprehensive side-effect profile of anastrozole and tamoxifen as adjuvant treatment for early-stage breast cancer: long-term safety analysis of the ATAC trial. Lancet Oncol. 2006;7:633–643.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supported by an unrestricted educational grant from MSD Oncology
Rights and permissions
About this article
Cite this article
Sánchez-Muñz, A., Ribelles, N. & Alba, E. Optimal adjuvant hormonal therapy in postmenopausal women with hormone-receptor-positive early breast cancer: have we answered the question?. Clin Transl Oncol 12, 614–620 (2010). https://doi.org/10.1007/s12094-010-0566-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12094-010-0566-9