Abstract
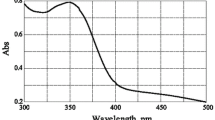
The kinetics of the reaction of HOBt with [Rh(H2O)5(OH)]2 + has been studied spectrophotometrically in aqueous medium as a function of [Rh(H2O)5OH2 + ], [HOBt], pH and temperature. At pH 4.3, the reaction proceeds via a rapid outer sphere association complex formation step followed by two consecutive steps. The first of these involves ligand-assisted anation, while the second involves chelation as the second aqua ligand is displaced. The association equilibrium constant for the outer sphere complex formation has been evaluated together with the rate constants for the two subsequent steps. The activation parameters for both steps have been evaluated using Eyrings equation. Thermodynamic parameters calculated from the temperature dependence of the outer sphere association equilibrium constants are also consistent with an associative mode of activation. The product of the reaction has been characterized by IR and ESI-mass spectroscopic analysis.

The kinetics of the reaction of HOBt with [Rh(H2O)5(OH)]2+ has been studied spectrophotometrically in aqueous medium as a function of [Rh(H2O)5OH2+], [HOBt], pH and temperature. At pH 4.3, the reaction proceeds via a rapid outer sphere association complex formation step followed by two consecutive steps. The first of these involves ligand-assisted anation, while the second involves chelation as the second aqua ligand is displaced. The association equilibrium constant for the outer sphere complex formation has been evaluated together with the rate constants for the two subsequent steps. The activation parameters for both steps have been evaluated using Eyrings equation. Thermodynamic parameters calculated from the temperature dependence of the outer sphere association equilibrium constants are also consistent with an associative mode of activation.
Similar content being viewed by others
References
Rosenberg B and Vancamp L 1970 Cancer Res. 30 1799
Davidson J P, Faber P J, Fisher IV R G, Mansy S, Peresie H J, Rosenberg B and Vancamp A L 1975 Cancer Chemother. Pt. 1 59, 287
Roberts J J, Knox R J, Friedlos F and Lydall D A, in 1986 Biochemical mechanisms of platinum antitumour drugs. eds. D. C. H. Me Brien and T. F. Slater, Oxford: IRL Press, p. 29
Sava G and Bergamo A 2007 Dalton Trans. 13 1267
Groessl M, Reisner E, Hartinger C G, Eichinger R, Semenova O, Timerbaev A R, Jakupec M A, Arion V B and Keppler B J 2007 Med. Chem. 50 2185
De Paula Q A, Batista A A, Nascimento O R, Costa-Filho A J, Schultz M S, Bonfadini M R and Oliva G J 2000 Braz. Chem. Soc. 11 530
Paula Q A, Batista A A, Castellano E E and Ellena J J 2002 Inorg. Biochem. 90 144
Vock C A, Ang W H, Scolaro C, Phillips A D, Lagopoulos L, Juillerat-Jenneret L, Sava G, Scopelliti R and Dyson P J 2007 J. Med. Chem. 50 2166
Timerbaev A R, Hartinger C G, Aleksenko S S and Keppler B K 2006 Chem. Rev. 106 2224
Alessio E 2004 Chem. Rev. 104 4203
Pieper T, Borsky K, Keppler B K 1999 In: Clarke M J and Sadler P J (eds) Metallopharmaceuticals I—DNA interactions. Springer, Europe, London/UK, pp 171
Sava G, Giraldi T, Mestroni G and Zassinovich G 1983 Chem. Biol. Interact. 45 1
(a) Ray S, Mohan R, Singh J K, Samantaray M K, Shaikh M M, Panda D and Ghosh P 2007 J. Am. Chem. Soc. 129 15042; (b) Hickey J L, Ruhayel R A, Barnard P J, Baker M V, Berners-Price S J and Filipovska A 2008 J. Am. Chem. Soc. 130 12570; (c) Teyssot M-L, Jarrousse A-S, Chevry A, De Haze A, Beaudoin C, Manin M, Nolan S P, Diez-Gonzales S, Morel L and Gautier A 2009 Chem. Eur. J. 15 314
Hindi K M, Panzner M J, Tessier C A, Cannon C L and Youngs W J 2009 Chem. Rev. 109 3859
Katsaros N and Anagnostopoulou A 2002 Crit. Rev. Oncol. Hematol. 42 297
Pruchnik F P, Jakimowicz P, Cuinik Z, Zakrzewskaczerwinska J, Opolski A, Weitrzyk J and Wojdat E 2002 Inorg.Chim. Acta. 334 59
Medvetz D A, Stakleff K D, Schreiber T, Custer P D, Hindi K, Panzner M J, Blanco D D, Taschner M J, Tessier C A and Youngs W J 2007 J. Med. Chem. 50 1703
Loganathan D and Morrison 2006 Photochem. Photobiol. 82 237
Swaminathan K and Harris G M 1996 J. Am. Chem. Soc. 88 4411
Buchacck R J and Harris G M 1976 Inorg. Chem. 15 926
Ghosh A K, Ghosh S and De G S 1996 Indian J. Chem. 35A 342
Ghosh A K, Sengupta P S and De G S 1997 Indian J. Chem. 36A 611
Mukhopadhyay S K, Ghosh A K and De G S 1999 Indian J. Chem. 38A 895
Sheehan J C and Hess G P 1955 J. Am. Chem. Soc. 77 1067
König W and Geiger R 1970 Chem. Ber. 103 788
Ayres G H and Forrester J S 1957 J. Inorg. Nucl. Chem. 3 365
Wolsey W C, Reynolds C A and Kleinberg J 1963 Inorg. Chem. 2 463
Steel D and Verhoeven P F M 2001 Vibrational Spectroscopy 25 29
Ohkubo A, Ezawa Y, Seio K and Sekine M 2002 Nucleic Acids Research Supplement No. 2, 29-30 Oxford University Press
Banyai I, Glaser J, Read M C and Stroem M S 1995 Inorg. Chem. 34 2423
Weyh J A and Hamm R E 1969 Inorg. Chem. 8 2298
Jeffrey G A 1997 An introduction to hydrogen bonding, Oxford: Oxford University Press
Desiraju G R and Steiner T T 1999 The weak hydrogen bonding in structural chemistry and biology, Oxford: Oxford University Press
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
BERA, B.K., MANDAL, A., MAITY, B. et al. Displacement of aqua ligands from the hydroxopentaaquarhodium(III) ion by 1-hydroxybenzotriazole (HOBt): A kinetic and mechanistic approach. J Chem Sci 124, 791–799 (2012). https://doi.org/10.1007/s12039-012-0287-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12039-012-0287-4