Abstract
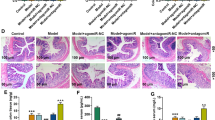
Aurantii Fructus Immaturus total flavonoids (AFIF) is the main effective fraction extracted from AFI, which has a good effect on promoting gastrointestinal motility. This study aimed to investigate AFIF which regulates miR-5100 to improve constipation symptoms in mice by targeting Frizzled-2 (Fzd2) to alleviate interstitial cells of Cajal (ICCs) calcium ion balance and autophagy apoptosis. The constipated mouse model was induced by an antibiotic suspension, and then treated with AFIF. RNA-seq sequencing, luciferase assay, immunofluorescence staining, transmission electron microscopy, ELISA, flow cytometry, quantitative polymerase chain reaction (PCR), and Western blot were applied in this study. The results showed that AFIF improved constipation symptoms in antibiotic-induced constipated mice, and decreased the autophagy-related protein Beclin1 levels and the LC3-II/I ratio in ICCs. miR-5100 and its target gene Fzd2 were screened as key miRNAs and regulator associated with autophagy. Downregulation of miR-5100 caused increased expression of Fzd2, decreased proliferation activity of ICCs, increased apoptotic cells, and enhanced calcium ion release and autophagy signals. After AFIF treatment, miR-5100 expression was upregulated and Fzd2 was downregulated, while autophagy-related protein levels and calcium ion concentration decreased. Furthermore, AFIF increased the levels of SP, 5-HT, and VIP, and increased the expression of PGP9.5, Sy, and Cx43, which alleviated constipation by improving the integrity of the enteric nervous system network. In conclusion, AFIF could attenuate constipation symptoms by upregulating the expression of miR-5100 and targeting inhibition of Fzd2, alleviating calcium overload and autophagic death of ICCs, regulating the content of neurotransmitters, and enhancing the integrity of the enteric nervous system network.








Similar content being viewed by others
Data Availability
The data used to support the current study are available from the corresponding author on reasonable request.
References
Camilleri M, Ford AC, Mawe GM, Dinning PG, Rao SS, Chey WD, Simren M, Lembo A et al (2017) Chronic constipation. Nat Rev Dis Primers 3:17095. https://doi.org/10.1038/nrdp.2017.95
Black CJ, Drossman DA, Talley NJ, Ruddy J, Ford AC (2020) Functional gastrointestinal disorders: advances in understanding and management. Lancet 396(10263):1664–1674. https://doi.org/10.1016/S0140-6736(20)32115-2
Herrington H (2017) Constipation. Clin Gastroenterol Hepatol S1542–3565(17):31416–31417. https://doi.org/10.1016/j.cgh.2017.11.043
Schiller LR (2019) Chronic constipation: new insights, better outcomes? Lancet Gastroenterol Hepatol 4(11):873–882. https://doi.org/10.1016/s2468-1253(19)30199-2
Bharucha AE, Lacy BE (2020) Mechanisms, evaluation, and management of chronic constipation. Gastroenterology 158(5):1232-1249 e1233. https://doi.org/10.1053/j.gastro.2019.12.034
Zhao Q, Chen YY, Xu DQ, Yue SJ, Fu RJ, Yang J, Xing LM, Tang YP (2021) Action mode of gut motility, fluid and electrolyte transport in chronic constipation. Front Pharmacol 12:630249. https://doi.org/10.3389/fphar.2021.630249
Wedel T, Spiegler J, Soellner S, Roblick UJ, Schiedeck TH, Bruch HP, Krammer HJ (2002) Enteric nerves and interstitial cells of Cajal are altered in patients with slow-transit constipation and megacolon. Gastroenterology 123(5):1459–1467. https://doi.org/10.1053/gast.2002.36600
Thuneberg L (1982) Interstitial cells of Cajal: intestinal pacemaker cells? Adv Anat Embryol Cell Biol 71:1–130
Kim HJ, Kim BJ (2017) Naringenin inhibits pacemaking activity in interstitial cells of Cajal from murine small intestine. Integr Med Res 6(2):149–155. https://doi.org/10.1016/j.imr.2017.02.001
Komuro T (2006) Structure and organization of interstitial cells of Cajal in the gastrointestinal tract. J Physiol 576(Pt 3):653–658. https://doi.org/10.1113/jphysiol.2006.116624
Hirst GD, Edwards FR (2004) Role of interstitial cells of Cajal in the control of gastric motility. J Pharmacol Sci 96(1):1–10. https://doi.org/10.1254/jphs.crj04002x
Cohen M, Cazals-Hatem D, Duboc H, Sabate JM, Msika S, Slove AL, Panis Y, Coffin B (2017) Evaluation of interstitial cells of Cajal in patients with severe colonic inertia requiring surgery: a clinical-pathological study. Colorectal Dis 19(5):462–467. https://doi.org/10.1111/codi.13511
He CL, Burgart L, Wang L, Pemberton J, Young-Fadok T, Szurszewski J, Farrugia G (2000) Decreased interstitial cell of cajal volume in patients with slow-transit constipation. Gastroenterology 118(1):14–21. https://doi.org/10.1016/s0016-5085(00)70409-4
Klein S, Seidler B, Kettenberger A, Sibaev A, Rohn M, Feil R, Allescher HD, Vanderwinden JM et al (2013) Interstitial cells of Cajal integrate excitatory and inhibitory neurotransmission with intestinal slow-wave activity. Nat Commun 4:1630. https://doi.org/10.1038/ncomms2626
Koh SD, Sanders KM, Ward SM (1998) Spontaneous electrical rhythmicity in cultured interstitial cells of cajal from the murine small intestine. J Physiol 513(Pt 1):203–213. https://doi.org/10.1111/j.1469-7793.1998.203by.x
Sanders KM, Koh SD, Ro S, Ward SM (2012) Regulation of gastrointestinal motility–insights from smooth muscle biology. Nat Rev Gastroenterol Hepatol 9(11):633–645. https://doi.org/10.1038/nrgastro.2012.168
Cobine CA, Hannah EE, Zhu MH, Lyle HE, Rock JR, Sanders KM, Ward SM, Keef KD (2017) ANO1 in intramuscular interstitial cells of Cajal plays a key role in the generation of slow waves and tone in the internal anal sphincter. J Physiol 595(6):2021–2041. https://doi.org/10.1113/JP273618
Tan RQ, Zhang Z, Ju J, Ling JH (2019) Effect of Chaihu Shugan powder-contained serum on glutamate-induced autophagy of interstitial cells of Cajal in the rat gastric antrum. Evid Based Complement Alternat Med 2019:7318616. https://doi.org/10.1155/2019/7318616
Zhang LM, Zeng LJ, Deng J, Zhang YQ, Wang YJ, Xie TY, Ling JH (2018) Investigation of autophagy and differentiation of myenteric interstitial cells of Cajal in the pathogenesis of gastric motility disorders in rats with functional dyspepsia. Biotechnol Appl Biochem 65(4):533–539. https://doi.org/10.1002/bab.1635
Filippi-Chiela EC, Viegas MS, Thome MP, Buffon A, Wink MR, Lenz G (2016) Modulation of autophagy by calcium signalosome in human disease. Mol Pharmacol 90(3):371–384. https://doi.org/10.1124/mol.116.105171
Cui X, Luo Y, Li C, Li Y, Wang Z (2015) Changes of intracellular Ca2+ in quercetin-induced autophagy progression. Acta Biochim Biophys Sin 47(11):908–914. https://doi.org/10.1093/abbs/gmv096. (Shanghai)
East DA, Campanella M (2013) Ca2+ in quality control: an unresolved riddle critical to autophagy and mitophagy. Autophagy 9(11):1710–1719. https://doi.org/10.4161/auto.25367
Committee CP (2020) Pharmacopoeia of the People’s Republic of China (Part I). Chinese Medical Science and Technology Press, Beijing
Zhang XJ, Zhao LY, Li JH, Zhang N, Fan ZW (2021) Research review on Fructus aurantii Immaturus. Acta Chin Med Pharmacol 49(1):94–100
Wang L, Wu F, Hong Y, Shen L, Zhao L, Lin X (2022) Research progress in the treatment of slow transit constipation by traditional Chinese medicine. J Ethnopharmacol 290:115075. https://doi.org/10.1016/j.jep.2022.115075
Xu SS, Xu J, Zhang XM, Zhang TJ, Liu CX (2018) Research progress on Citri Reticulatae Pericarpium, Aurantii Fructus Immaturus, and Aurantii Fructus and Q-marker predictive analysis. Chin Tradit Herb Drugs 49(1):35–44. https://doi.org/10.7501/j.issn.0253-2670.2018.01.004
Wang C, Ren Q, Chen XT, Song ZQ, Ning ZC, Gan JH, Ma XL, Liang DR et al (2018) System pharmacology-based strategy to decode the synergistic mechanism of Zhi-zhu Wan for functional dyspepsia. Front Pharmacol 9:841. https://doi.org/10.3389/fphar.2018.00841
Yan S, Hao M, Yang H, Sun M, Wu B, Yue Y, Wang X (2020) Metabolomics study on the therapeutic effect of the Chinese herb pair Fructus Aurantii Immaturus and Rhizoma Atractylodis Macrocephalae in constipated rats based on UPLC-Q/TOF-MS analysis. Ann Palliat Med 9(5):2837–2852. https://doi.org/10.21037/apm-20-280
Qi QH, Wang J, Liang GG, Wu XZ (2007) Da-Cheng-Qi-Tang promotes the recovery of gastrointestinal motility after abdominal surgery in humans. Dig Dis Sci 52(6):1562–1570. https://doi.org/10.1007/s10620-007-9751-2
Dou DB, Cai G, Wang SP, Ni KZ, Tang JF (2002) Clinical studies on functional dyspepsia treated with different dosage-form of Zhishi Xiaopi Pill Recipe. Chin J Integr Med 8(3):186–190
Wang SY, Liu YP, Fan YH, Zhang L, Cai LJ, Lv B (2015) Mechanism of aqueous Fructus Aurantii Immaturus extracts in neuroplexus of cathartic colons. World J Gastroenterol 21(31):9358–9366. https://doi.org/10.3748/wjg.v21.i31.9358
Yan S, Yue YZ, Sun MM, Wu BS, Wang XP (2020) Suppressive effect of Aurantii Fructus Immaturus and Atractylodis Macrocephalae Rhizoma on glutamic acid-induced autophagy of interstitial cells of Cajal. J Integr Med 18(4):334–343. https://doi.org/10.1016/j.joim.2020.04.005
Chen HF, Zhang WG, Yuan JB, Li YG, Yang SL, Yang WL (2012) Simultaneous quantification of polymethoxylated flavones and coumarins in Fructus Aurantii and Fructus Aurantii Immaturus using HPLC-ESI-MS/MS. J Pharm Biomed Anal 59:90–95. https://doi.org/10.1016/j.jpba.2011.10.013
He Y, Li Z, Wang W, Sooranna SR, Shi Y, Chen Y, Wu C, Zeng J et al (2018) Chemical profiles and simultaneous quantification of Aurantii fructus by use of HPLC-Q-TOF-MS combined with GC-MS and HPLC methods. Molecules 23(9):2189. https://doi.org/10.3390/molecules23092189
Wang LF, Liu XL, Li HT, Chen QY, Wang Y, Zou B, Yang M, Zhang XF et al (2020) Mechanism of Aurantii Fructus Immaturus volatile oil in treatment of slow transit constipation based on network pharmacology. Chin J Chin Mater Med 45(8):1909–1917
Yan L, Yu L, Zhao L, Wang D, Qin D, Fan H, Cheng L, Qiu M et al (2019) Efficacy of Weikang Pian in patients with functional dyspepsia: a double-blind, randomized, placebo-controlled clinical trial. Evid Based Complement Alternat Med 2019:4827046. https://doi.org/10.1155/2019/4827046
Tan W, Li Y, Wang Y, Zhang Z, Wang T, Zhou Q, Wang X (2017) Anti-coagulative and gastrointestinal motility regulative activities of Fructus Aurantii Immaturus and its effective fractions. Biomed Pharmacother 90:244–252. https://doi.org/10.1016/j.biopha.2017.03.060
Wang S, Bao YR, Li TJ, Yu T, Chang X, Yang GL, Meng XS (2017) Mechanism of Fructus Aurantii flavonoids promoting gastrointestinal motility: from organic and inorganic endogenous substances combination point of view. Pharmacogn Mag 13(51):372–377. https://doi.org/10.4103/pm.pm_179_16
Hong Y, Ren X, Liu W, Sun K, Chen B, Liu B, Yu X, Chen Q et al (2021) miR-128 participates in the pathogenesis of chronic constipation by regulating the p38alpha/M-CSF inflammatory signaling pathway. Am J Physiol Gastrointest Liver Physiol 321(4):G436–G447. https://doi.org/10.1152/ajpgi.00114.2021
Krishna CV, Singh J, Thangavel C, Rattan S (2016) Role of microRNAs in gastrointestinal smooth muscle fibrosis and dysfunction: novel molecular perspectives on the pathophysiology and therapeutic targeting. Am J Physiol Gastrointest Liver Physiol 310(7):G449-459. https://doi.org/10.1152/ajpgi.00445.2015
Park C, Yan W, Ward SM, Hwang SJ, Wu Q, Hatton WJ, Park JK, Sanders KM et al (2011) MicroRNAs dynamically remodel gastrointestinal smooth muscle cells. PLoS ONE 6(4):e18628. https://doi.org/10.1371/journal.pone.0018628
Deng J, Yang S, Yuan Q, Chen Y, Li D, Sun H, Tan X, Zhang F et al (2017) Acupuncture ameliorates postoperative lleus via IL-6-miR-19a-KIT axis to protect interstitial cells of Cajal. Am J Chin Med 45(4):737–755. https://doi.org/10.1142/S0192415X17500392
Zhao S, Chen Q, Kang X, Kong B, Wang Z (2018) Aberrantly expressed genes and miRNAs in slow transit constipation based on RNA-Seq analysis. Biomed Res Int 2018:2617432. https://doi.org/10.1155/2018/2617432
Inagawa H, Saika T, Nishiyama N, Nisizawa T, Kohchi C, Uenobe M, Soma GI (2017) Improvement effect of dewaxed brown rice on constipation in antibiotic-treated mice. In Vivo 31(4):573–577. https://doi.org/10.21873/invivo.11096
Hu Q, Sun DF, Sun LJ, Wang C, Hu LH, Fang ZJ, Deng Q (2020) Intervention effect of mussel and oyster complex on constipation in mice with intestinal flora disorder. Mod Food Sci Technol 36(10):16–26. https://doi.org/10.13982/j.mfst.1673-9078.2020.10.0397
Zhao Q, Xing F, Tao Y, Liu H, Huang K, Peng Y, Feng N, Liu C (2020) Xiaozhang Tie improves intestinal motility in rats with cirrhotic ascites by regulating the stem cell factor/c-kit pathway in interstitial cells of Cajal. Front Pharmacol 11:1. https://doi.org/10.3389/fphar.2020.00001
Zhou Y, Wang Z, Huang Y, Bai C, Zhang X, Fang M, Ju Z, Liu B (2022) Membrane dynamics of ATG4B and LC3 in autophagosome formation. J Mol Cell Biol 13(12):853–863. https://doi.org/10.1093/jmcb/mjab059
Niu LJ, Xu RX, Zhang P, Du MX, Jiang XD (2012) Suppression of Frizzled-2-mediated Wnt/Ca(2)(+) signaling significantly attenuates intracellular calcium accumulation in vitro and in a rat model of traumatic brain injury. Neuroscience 213:19–28. https://doi.org/10.1016/j.neuroscience.2012.03.057
Zhuang Z, Chen M, Niu J, Qu N, Ji B, Duan X, Liu Z, Liu X et al (2019) The manufacturing process of kiwifruit fruit powder with high dietary fiber and its laxative effect. Molecules 24(21):3813. https://doi.org/10.3390/molecules24213813
King SK, Sutcliffe JR, Ong SY, Lee M, Koh TL, Wong SQ, Farmer PJ, Peck CJ et al (2010) Substance P and vasoactive intestinal peptide are reduced in right transverse colon in pediatric slow-transit constipation. Neurogastroenterol Motil 22(8):883–892, e234. https://doi.org/10.1111/j.1365-2982.2010.01524.x
Wang Y, Wang Q, Kuerban K, Dong M, Qi F, Li G, Ling J, Qiu W et al (2019) Colonic electrical stimulation promotes colonic motility through regeneration of myenteric plexus neurons in slow transit constipation beagles. Biosci Rep 39(5). 10.1042/BSR20182405
Li S, He Y, Zhang H, Zheng R, Xu R, Liu Q, Tang S, Ke X et al (2020) Formulation of traditional Chinese medicine and its application on intestinal flora of constipated rats. Microb Cell Fact 19(1):212. https://doi.org/10.1186/s12934-020-01473-3
Wu M, Li Y, Gu Y (2020) Hesperidin improves colonic motility in loeramide-induced constipation rat model via 5-hydroxytryptamine 4R/cAMP signaling pathway. Digestion 101(6):692–705. https://doi.org/10.1159/000501959
Jang Y, Kim TK, Shim WS (2013) Naringin exhibits in vivo prokinetic activity via activation of ghrelin receptor in gastrointestinal motility dysfunction rats. Pharmacology 92(3–4):191–197. https://doi.org/10.1159/000354579
Acknowledgements
We are grateful for the support from the Affiliated Hospital of Southwest Medical University and the North Sichuan Medical College.
Funding
This study was supported by the Youth Fund of the National Natural Science Foundation of China (No. 82004173), Science Fund of the National Natural Science Foundation of China (No. 82074429), and the Science and Technology Research Special Project of Sichuan Provincial Administration of Traditional Chinese Medicine (No. 2020JC0063).
Author information
Authors and Affiliations
Contributions
All authors have contributed significantly to this work. YZ and YW contributed equally to this work. YZ, YW, and XGT conceived and designed the experiments; TYC, JL, and SYT performed most of the experiments; FZ and QL contributed to data collection and analysis; YZ and YW wrote the manuscript; XGT revised and edited the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
Animal experiments were approved and conducted in accordance with the guidelines of the Experimental Animal Ethics Committee of West China Hospital of Sichuan University (approval No. 20221111003).
Consent to Participate
Not applicable.
Consent to Publish
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wen, Y., Zhan, Y., Chen, T. et al. Total Flavonoids of Aurantii Fructus Immaturus Regulate miR-5100 to Improve Constipation by Targeting Fzd2 to Alleviate Calcium Balance and Autophagy in Interstitial Cells of Cajal. Mol Neurobiol (2024). https://doi.org/10.1007/s12035-024-03958-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12035-024-03958-3