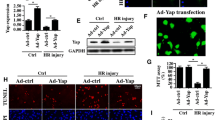
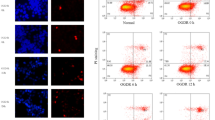
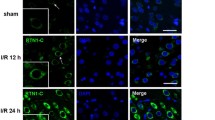
Abstract
SET domain protein 3 (SETD3) is an actin-specific methyltransferase, a rare post-translational modification with limited known biological functions. Till now, the function of SETD3 in cerebral ischemia-reperfusion (I/R)-induced injury remains unknown. Here, we show that the protein level of SETD3 is decreased in rat neurons after cerebral I/R injury. SETD3 promotes neuronal survival after both glucose and oxygen deprivation/reoxygenation (OGD/R) and cerebral I/R injury, and knockdown of SETD3 increases OGD/R-induced neuronal death. We further show that OGD/R-induced downregulation of SETD3 leads to the decrease of cellular ATP level, the reduction of mitochondrial electric potential and the increase of ROS production, thereby promoting mitochondrial dysfunction. We found that SETD3 reduction-induced mitochondrial dysfunction is mediated by the suppression of actin polymerization after OGD/R. Furthermore, we demonstrate that I/R-induced upregulation of PTEN leads to the downregulation of SETD3, and suppressing PTEN protects against ischemic neuronal death through downregulation of SETD3 and enhancement of actin polymerization. Together, this study provides the first evidence suggesting that I/R-induced downregulation of SETD3 mediates PTEN upregulation-induced ischemic neuronal death through downregulation of SETD3 and subsequent suppression of actin polymerization. Thus, upregulating SETD3 is a potential approach for the development of ischemic stroke therapy.







Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article.
References
Feigin VL, Nguyen G, Cercy K et al (2018) Global, regional, and country-specific lifetime risks of stroke, 1990 and 2016. N Engl J Med 379(25):2429–2437. https://doi.org/10.1056/NEJMoa1804492
Kochanek KD, Murphy SL, Xu J (2015) Deaths: Final Data for 2011. National vital statistics reports : from the Centers for Disease Control and Prevention. Natl Center Health Stat Natl Vital Stat Syst 63(3):1–120
Lapchak PA, Zhang JH (2017) The high cost of stroke and stroke cytoprotection research. Transl Stroke Res 8(4):307–317. https://doi.org/10.1007/s12975-016-0518-y
Fisher M, Schaebitz W (2000) An overview of acute stroke therapy: past, present, and future. Arch Intern Med 160(21):3196–3206. https://doi.org/10.1001/archinte.160.21.319
Enzmann G, Kargaran S, Engelhardt B (2018) Ischemia-reperfusion injury in stroke: impact of the brain barriers and brain immune privilege on neutrophil function. Ther Adv Neurol Disord 11:1756286418794184. https://doi.org/10.1177/1756286418794184
Liu F, Lu J, Manaenko A et al (2018) Mitochondria in ischemic stroke: new insight and implications. Aging Dis 9(5):924–937. https://doi.org/10.14336/ad.2017.1126
Hofmeijer J, van Putten MJ (2012) Ischemic cerebral damage: an appraisal of synaptic failure. Stroke. 43(2):607–615. https://doi.org/10.1161/strokeaha.111.632943
Lee JM, Grabb MC, Zipfel GJ et al (2000) Brain tissue responses to ischemia. J Clin Invest 106(6):723–731. https://doi.org/10.1172/jci11003
Dharmasaroja PA (2016) Fluid intake related to brain edema in acute middle cerebral artery infarction. Transl Stroke Res 7(1):49–53. https://doi.org/10.1007/s12975-015-0439-1
Kim DW, Kim KB, Kim JY et al (2011) Characterization of a novel histone H3K36 methyltransferase setd3 in zebrafish. Biosci Biotechnol Biochem 75(2):289–294. https://doi.org/10.1271/bbb.100648
Eom GH, Kim KB, Kim JH et al (2011) Histone methyltransferase SETD3 regulates muscle differentiation. J Biol Chem 286(40):34733–34742. https://doi.org/10.1074/jbc.M110.203307
Liao J, Luo S, Yang M et al (2020) Overexpression of CXCR5 in CD4(+) T cells of SLE patients caused by excessive SETD3. Clin Immunol (Orlando, Fla) 214:108406. https://doi.org/10.1016/j.clim.2020.108406
Diep J, Ooi YS, Wilkinson AW et al (2019) Enterovirus pathogenesis requires the host methyltransferase SETD3. Nat Microbiol 4(12):2523–2537. https://doi.org/10.1038/s41564-019-0551-1
Cheng X, Hao Y, Shu W et al (2017) Cell cycle-dependent degradation of the methyltransferase SETD3 attenuates cell proliferation and liver tumorigenesis. J Biol Chem 292(22):9022–9033. https://doi.org/10.1074/jbc.M117.778001
Abaev-Schneiderman E, Admoni-Elisha L, Levy D (2019) SETD3 is a positive regulator of DNA-damage-induced apoptosis. Cell Death Dis 10(2):74. https://doi.org/10.1038/s41419-019-1328-4
Pollard TD, Cooper JA (2009) Actin, a central player in cell shape and movement. Science (New York, NY) 326(5957):1208–1212. https://doi.org/10.1126/science.1175862
Dominguez R, Holmes KC (2011) Actin structure and function. Annu Rev Biophys 40:169–186. https://doi.org/10.1146/annurev-biophys-042910-155359
Honkura N, Matsuzaki M, Noguchi J et al (2008) The subspine organization of actin fibers regulates the structure and plasticity of dendritic spines. Neuron. 57(5):719–729. https://doi.org/10.1016/j.neuron.2008.01.013
Tian D, Diao M, Jiang Y et al (2015) Anillin regulates neuronal migration and neurite growth by linking RhoG to the actin cytoskeleton. Curr Biol : CB 25(9):1135–1145. https://doi.org/10.1016/j.cub.2015.02.072
Li GB, Zhang HW, Fu RQ et al (2018) Mitochondrial fission and mitophagy depend on cofilin-mediated actin depolymerization activity at the mitochondrial fission site. Oncogene. 37(11):1485–1502. https://doi.org/10.1038/s41388-017-0064-4
Wilkinson AW, Diep J, Dai S et al (2019) SETD3 is an actin histidine methyltransferase that prevents primary dystocia. Nature. 565(7739):372–376. https://doi.org/10.1038/s41586-018-0821-8
Kwiatkowski S, Seliga AK, Vertommen D et al (2018) SETD3 protein is the actin-specific histidine N-methyltransferase. eLife. 11:7. https://doi.org/10.7554/eLife.37921
Liu B, Li L, Zhang Q et al (2010) Preservation of GABAA receptor function by PTEN inhibition protects against neuronal death in ischemic stroke. Stroke. 41(5):1018–1026. https://doi.org/10.1161/strokeaha.110.579011
Chen SF, Pan MX, Tang JC et al (2020) Arginine is neuroprotective through suppressing HIF-1α/LDHA-mediated inflammatory response after cerebral ischemia/reperfusion injury. Mol Brain 13(1):63. https://doi.org/10.1186/s13041-020-00601-9
Shi Y, Wang C, Zhou X et al (2020) Downregulation of PTEN promotes podocyte endocytosis of lipids aggravating obesity-related glomerulopathy. Am J Physiol Ren Physiol 318(3):F589–f599. https://doi.org/10.1152/ajprenal.00392.2019
Chen Y, Luo C, Zhao M et al (2015) Administration of a PTEN inhibitor BPV(pic) attenuates early brain injury via modulating AMPA receptor subunits after subarachnoid hemorrhage in rats. Neurosci Lett 588:131–136. https://doi.org/10.1016/j.neulet.2015.01.005
He Z, Ning N, Zhou Q et al (2020) Mitochondria as a therapeutic target for ischemic stroke. Free Radic Biol Med 146:45–58. https://doi.org/10.1016/j.freeradbiomed.2019.11.005
Vosler PS, Graham SH, Wechsler LR et al (2009) Mitochondrial targets for stroke: focusing basic science research toward development of clinically translatable therapeutics. Stroke. 40(9):3149–3155. https://doi.org/10.1161/strokeaha.108.543769
Galluzzi L, Kepp O, Kroemer G (2012) Mitochondria: master regulators of danger signalling. Nat Rev Mol Cell Biol 13(12):780–788. https://doi.org/10.1038/nrm3479
Shu WJ, Du HN (2021) The methyltransferase SETD3-mediated histidine methylation: biological functions and potential implications in cancers. Biochimica et Biophysica acta Rev Cancer 1875(1):188465. https://doi.org/10.1016/j.bbcan.2020.188465
Zhou H, Wang S, Zhu P et al (2018) Empagliflozin rescues diabetic myocardial microvascular injury via AMPK-mediated inhibition of mitochondrial fission. Redox Biol 15:335–346. https://doi.org/10.1016/j.redox.2017.12.019
Alvariño R, Alonso E, Tabudravu JN et al (2021) Tavarua deoxyriboside A and jasplakinolide as potential neuroprotective agents: effects on cellular models of oxidative stress and neuroinflammation. ACS Chem Neurosci 12(1):150–162. https://doi.org/10.1021/acschemneuro.0c00626
Li J, Yen C, Liaw D et al (1997) PTEN, a putative protein tyrosine phosphatase gene mutated in human brain, breast, and prostate cancer. Science (New York, NY) 275(5308):1943–1947. https://doi.org/10.1126/science.275.5308.1943
Li C, Xu X, Wang Z et al (2020) Exercise ameliorates post-stroke depression by inhibiting PTEN elevation-mediated upregulation of TLR4/NF-κB/NLRP3 signaling in mice. Brain Res 1736:146777. https://doi.org/10.1016/j.brainres.2020.146777
Liu R, Liao XY, Pan MX et al (2019) Glycine exhibits neuroprotective effects in ischemic stroke in rats through the inhibition of M1 microglial polarization via the NF-κB p65/Hif-1α signaling pathway. J Immunol (Baltimore, Md: 1950) 202(6):1704–1714. https://doi.org/10.4049/jimmunol.1801166
Zhang Z, Wang Q, Zhao X et al (2020) YTHDC1 mitigates ischemic stroke by promoting Akt phosphorylation through destabilizing PTEN mRNA. Cell Death Dis 11(11):977. https://doi.org/10.1038/s41419-020-03186-2
Guo JM, Liu AJ, Zang P et al (2013) ALDH2 protects against stroke by clearing 4-HNE. Cell Res 23(7):915–930. https://doi.org/10.1038/cr.2013.69
Liu R, Tang JC, Pan MX et al (2018) ERK 1/2 Activation mediates the neuroprotective effect of BpV(pic) in Focal cerebral ischemia-reperfusion injury. Neurochem Res 43(7):1424–1438. https://doi.org/10.1007/s11064-018-2558-z
Cui Y, Zhang Y, Zhao X et al (2021) ACSL4 exacerbates ischemic stroke by promoting ferroptosis-induced brain injury and neuroinflammation. Brain Behav Immun 93:312–321. https://doi.org/10.1016/j.bbi.2021.01.003
Acknowledgements
This work was supported by Shandong Provincial Natural Science Foundation (ZR2020MH282), Clinical Medicine +X grant of the Affiliated Hospital of Qingdao University (Grant Number: QDFY+X2019005) to Q.W.; National Natural Science Foundation of China (31900634) to Y.C.; this work was also supported by the National Key R&D Program of China (2019YFC0120000; 2018YFC1312300), the National Natural Science Foundation of China (NSFC: 82071385), and the Key Research and Development Project of Shandong (2019JZZY021010) to Q.W. We thank Qian Wen for the technical support and the service provided by the Institute of Neurodegeneration and Neurorehabilitation at Qingdao University.
Funding
This work was supported by Shandong Provincial Natural Science Foundation (ZR2020MH282), Clinical Medicine +X grant of the Affiliated Hospital of Qingdao University (Grant Number: QDFY+X2019005) to Q.W.; National Natural Science Foundation of China (31900634) to Y.C..; this work was also supported by National Key R&D Program of China (2019YFC0120000; 2018YFC1312300), the National Natural Science Foundation of China (NSFC: 82071385), and the Key Research and Development Project of Shandong (2019JZZY021010) to Q.W.
Author information
Authors and Affiliations
Contributions
Xiangyu Xu designed and performed most experiments, analyzed data, and prepared the manuscript; Yu Cui designed and performed some experiments, analyzed data, and prepared the manuscript. Congqin Li, Yuyang Wang, Jing Cheng, Songfeng Chen performed experiments and analyzed data; Jiangdong Sun, Jinyang Ren helped with MCAO model construction; Xujin Yao, Jingchen Gao, Xiaohong Huang collected and analyzed the data. All authors have read and approved the final manuscript. Qi Wan and Qiang Wang conceptualized the research, directed the study and prepared the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All animal procedures were in accordance with the Institutional Animal Care guidelines and the Animal Care and Ethics Committee of Qingdao University (201905SD411202103019).
Consent for publication
Not applicable
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Fig. S1
Actin polymerization restored mitochondrial damage caused by SETD3 knockdown after OGD/R. (a) Flow cytometry analysis of mitochondrial potential by JC-1 staining of lentivirus-transfected neurons in the presence or absence of Jasplakinolide (2 μM). (b) ATP production was measured of lentivirus-transfected neurons in the presence or absence of Jasplakinolide (2 μM). (c) Flow cytometry analysis of intracellular ROS level by DCFH-DA staining of lentivirus-transfected neurons in the presence or absence of Jasplakinolide (2 μM). For a to c, neurons were first transfected with lentivirus for two days and then subjected to OGD/R for 1.5 h, Jasplakinolide was added 6 h after reoxygenation. All data are representative of or combined from at least three independent experiments (PNG 21480 kb)
Fig. S2
Quantification of mean fluorescence intensity of intracellular ROS level by DCFH-DA staining. (a, b) Quantification of mean fluorescence intensity (MFI) of intracellular ROS level by DCFH-DA staining of lentivirus-transfected neurons in Fig. 4 e (a) and f (b). (c) Quantification of MFI of intracellular ROS level by DCFH-DA staining of neurons-treated with 2 μM Jasplakinolide or vehicle in Fig. 5 e. (d) Quantification of MFI of intracellular ROS level by DCFH-DA staining of lentivirus-transfected neurons in the presence or absence of 2 μM Jasplakinolide in Figure 5 h. (e) Quantification of MFI of intracellular ROS level by DCFH-DA staining of lentivirus-transfected neurons in the presence or absence of Jasplakinolide (2 μM) in in Fig. S1. For Fig. S2 d to e, neurons were first transfected with lentivirus for two days and then subjected to OGD/R for 1.5 h, Jasplakinolide was added 6 h after reoxygenation. The data are means ± SD., *P < 0.05, **P < 0.01, ***P < 0.001 by one-way ANOVA analysis followed by Tukey post hoc test (a, d, e) or two-way ANOVA analysis followed by Bonferroni post hoc test (b, c) (PNG 21480 kb)
Rights and permissions
About this article
Cite this article
Xu, X., Cui, Y., Li, C. et al. SETD3 Downregulation Mediates PTEN Upregulation-Induced Ischemic Neuronal Death Through Suppression of Actin Polymerization and Mitochondrial Function. Mol Neurobiol 58, 4906–4920 (2021). https://doi.org/10.1007/s12035-021-02459-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-021-02459-x