Abstract
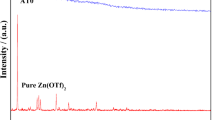
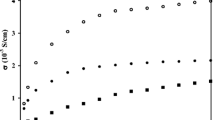
In the present work, an attempt has been made to develop nano aluminium oxide (Al2O3)-filled polyvinyl alcohol (PVA) composite gel electrolytes. Surface morphological studies, thermal behaviour, electrochemical stability and electrical characterization of these composite gel electrolytes have been performed. An increase in the concentration of Al2O3 in composite gel electrolytes increases the amorphous characteristics of pure PVA. Bulk conductivity of composite gel electrolytes increases by an order of magnitude on addition of a nano filler. Maximum conductivity of 5·81 × 10−2 S/cm is observed for 6 wt% Al2O3-filled polymer gel composite electrolytes. Temperature dependence of electrical conductivity shows a combination of Arrhenius and Vogel-Tamman-Fulcher (VTF) nature. Maximum current stability during oxidation and reduction cycle is noticed for 6 wt% Al2O3-filled PVA composite electrolyte, viz. ±1·65 V.
Similar content being viewed by others
References
Agrawal R C and Pandey G P 2008 J. Phys. D: Appl. Phys. 41 223001
Agrawal S L and Awadhia A 2004 Bull. Mater. Sci. 27 523
Agrawal S L and Shukla P K 2000 J. Pure Appl. Phys. 38 53
Awadhia A and Agrawal S L 2007 Solid State Ionics 178 951
Bhat N V and Deshmukh R R 2002 J. Pure Appl. Phys. 40 361
Bohnke O, Rousselot C, Gillet P A and Truche C 1992 J. Electrochem. Soc. 139
Caravanier C C, Montigny B C, Lemordant D and Bosser G 2003 Solid State Ionics 156 113
Cavalieri F, Chiessi E, Spagnoti C and Cowman M K 2003 J. Mater. Sci. Mater. Med. 14 687
Chandra S, Sekhon S S and Arora N 2000 Ionics 6 112
Colomban P (ed.) 1992 Proton conductors: solids, membranes and gels-materials and devices (Cambridge: Cambridge university Press)
Croce F, Gerace F, Dautzemberg G, Passerini S, Appetechi G B and Scrosati B 1994 Electrochim. Acta 39 2187
Cullity B D (ed.) 1978 Elements of X-ray diffraction (London: Addition-Wesley Publishing Company Inc.) 2nd edn, pp. 281–286
Duangkaew P and Woothikanokkhan J 2008 J. Appl. Polym. Sci. 109 452
Finch C A (ed.) 1973 Polyvinyl alcohol: properties and applications (Bristol: John Wiley & Sons)
Gong K C, Cai H S, Nazri G, Huggins R A and Shriver D F (eds) 1989 Solid State Ionics (Pittsburgh: Materials Research Society) Vol. 135, p. 377
Gray F M 1991 Solid polymer electrolytes-fundamental and technological applications (New York: VCH)
Honma I, Hirakava S, Yamada and Bae J M 1999 Solid State Ionics 118 29
Kim K M, Park N G, Ryu K S and Chang S H 2002 Polymer 43 3951
Koksbang R, Oslen I I and Shackle D 1994 Solid State Ionics 69 320
Kubota N, Watanabe H, Konaka G and Eguchi Y 2000 J. Appl. Polym. Sci. 76 12
Kubota N, Fujii S, Tatsumoto N and Sano T 2002 J. Appl. Polym. Sci. 83 2655
MacCallum J R and Vincent C A (eds) 1989 Polymer electrolyte reviews (London: Elsevier Applied Sciences Publisher) pp. 1–2
Mukherjee G S, Shukla N, Singh R K and Mathur G N 2004 J. Sci. Ind. Res. 63 596
Pandey K, Dwivedi M M, Tripathi M, Singh M and Agrawal S L 2008 Ionics 14 515
Patel S K, Patel R B, Awadhia A, Chand N and Agrawal S L 2007 Pramana 69 467
Radhucha D, Wieczorek W, Florjanczyk Z and Stevens J R 1996 J. Phys. Chem. 100 20126
Ray S S and Okamoto M 2003 Prog. Polym. Sci. 28 1539
Sato T, Bonno K, Maruo T and Nozu R 2005 J Power Sources 152 264
Scrosati B and Vincent C A 2000 M R S Bull. 28
Sekhon S S 2003 Bull. Mater. Sci. 26 321
Sekhon S S, Arora N, Singh B and Chandra S 2002 J. Mater. Sci. 37 2159
Shukla P K and Agrawal S L 2000 Ionics 6 312
Singh H P, Kumar R and Sekhon S S 2005 Bull. Mater. Sci. 128 467
Song J Y, Wang Y Y and Wan C C 2000 J. Electrochem. Soc. 147 3219
Tager A (ed.) 1978 Physical chemistry of polymers (Moscow: MIR Publishers)
Wieczorek W, Florjanczyk Z and Stevens J R 1995 Electrochim. Acta 40 2327
Zou G X, Jin P Q and Xin L Z 2008 J. Elastomers Plastics 40 303
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chand, N., Rai, N., Agrawal, S.L. et al. Morphology, thermal, electrical and electrochemical stability of nano aluminium-oxide-filled polyvinyl alcohol composite gel electrolyte. Bull Mater Sci 34, 1297–1304 (2011). https://doi.org/10.1007/s12034-011-0318-7
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12034-011-0318-7