Abstract
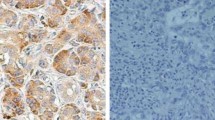
S100A11 is a member of S100 protein family, and our previous study showed that S100A11 is one of the up-regulated proteins that have not been reported to be associated with pancreatic carcinoma. The purpose of this study was to investigate the relation between S100A11 expression and the clinicopathological variables and clinical outcome in patients with pancreatic adenocarcinoma. Immunohistochemistry analysis was performed for S100A11 in 78 pairs of specimens of human pancreatic adenocarcinoma tissues and adjacent nontumorous tissues. The univariate and multivariate survival analyses were also performed to determine its prognostic significance. S100A11 expression in pancreatic adenocarcinoma (62/78) was significantly higher than that in the adjacent nontumorous tissues (19/78) (P = 0.000). High expression of S100A11 was associated with the lymph node metastasis and histological differentiation (P = 0.003 and 0.004, respectively). Univariate analysis showed that S100A11 expression was associated with poor prognosis (P = 0.0000). Multivariate analysis using the Cox regression model indicated that age ≥65 years, CA19-9 ≥1,000 U/ml and positive S100A11 were independent prognostic indicators of pancreatic adenocarcinoma (P = 0.002, 0.004 and 0.001, respectively). These results suggested that S100A11 might be a significant tumor marker for pancreatic adenocarcinoma and an unfavorable predictor for prognosis of patients who have undergone surgical resection.


Similar content being viewed by others
References
Lee SE, et al. Clinical implications of immunohistochemically demonstrated lymph node micrometastasis in resectable pancreatic cancer. J Korean Med Sci. 2011;26(7):881–5.
Jemal A, et al. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90.
Hidalgo M. Pancreatic cancer. N Engl J Med. 2010;362(17):1605–17.
Akdoqan M, et al. Extraordinarily elevated CA19-9 in benign conditions: a case report and review of the literature. Tumori. 2001;87(5):337–9.
Ni XG, et al. The clinical value of serum CEA, CA19-9, and CA242 in the diagnosis and prognosis of pancreatic cancer. Eur J Surg Oncol. 2005;31(2):164–9.
Duffy MJ. CA 19-9 as a marker for gastrointestinal cancers: a review. Ann Clin Biochem. 1998;35(Pt 3):364–70.
Donato R. S100: a multigenic family of calcium-modulated proteins of the EF-hand type with intracellular and extracellular functional roles. Int J Biochem Cell Biol. 2001;33(7):637–68.
Heizmann CW, Fritz G, Schafer BW. S100 proteins: structure, functions and pathology. Front Biosci. 2002;7:d1356–68.
Marenholz I, Heizmann CW, Fritz G. S100 proteins in mouse and man: from evolution to function and pathology (including an update of the nomenclature). Biochem Biophys Res Commun. 2004;322(4):1111–22.
Rety S, et al. Structural basis of the Ca(2+)-dependent association between S100C (S100A11) and its target, the N-terminal part of annexin I. Structure. 2000;8(2):175–84.
Dempsey AC, Walsh MP, Shaw GS. Unmasking the annexin I interaction from the structure of Apo-S100A11. Structure. 2003;11(7):887–97.
Chen JH, et al. Comparative proteomic analysis of differentially expressed proteins in human pancreatic cancer tissue. Hepatobiliary Pancreat Dis Int. 2009;8(2):193–200.
Sobin LH, Gospodarowicz MK, Wittekind C. International Union Against Cancer (UICC) TNM classification of malignant tumours. 7th ed. New York: Wiley-Liss; 2010.
Masunaga R, et al. Cyclooxygenase-2 expression correlates with tumor neovascularization and prognosis in human colorectal carcinoma patients. Clin Cancer Res. 2000;6(10):4064–8.
Ji J, et al. Differential expression of S100 gene family in human esophageal squamous cell carcinoma. J Cancer Res Clin Oncol. 2004;130(8):480–6.
Oue N, et al. Gene expression profile of gastric carcinoma: identification of genes and tags potentially involved in invasion, metastasis, and carcinogenesis by serial analysis of gene expression. Cancer Res. 2004;64(7):2397–405.
Torres-Cabala C, et al. Differential expression of S100C in thyroid lesions. Int J Surg Pathol. 2004;12(2):107–15.
Finn SP, et al. Expression microarray analysis of papillary thyroid carcinoma and benign thyroid tissue: emphasis on the follicular variant and potential markers of malignancy. Virchows Arch. 2007;450(3):249–60.
Kanamori T, et al. Increased expression of calcium-binding protein S100 in human uterine smooth muscle tumours. Mol Hum Reprod. 2004;10(10):735–42.
Rust R, et al. High expression of calcium-binding proteins, S100A10, S100A11 and CALM2 in anaplastic large cell lymphoma. Br J Haematol. 2005;131(5):596–608.
Tanaka M, et al. Human calgizzarin; one colorectal cancer-related gene selected by a large scale random cDNA sequencing and northern blot analysis. Cancer Lett. 1995;89(2):195–200.
Stulik J, et al. Protein abundance alterations in matched sets of macroscopically normal colon mucosa and colorectal carcinoma. Electrophoresis. 1999;20(18):3638–46.
Flam F, et al. Identification of distinctive protein expression patterns in colorectal adenoma. Proteomics Clin Appl. 2010;4(1):60–70.
Jung Y, et al. Clinical validation of colorectal cancer biomarkers identified from bioinformatics analysis of public expression data. Clin Cancer Res. 2011;17(4):700–9.
Schaefer KL, et al. Expression profiling of t(12; 22) positive clear cell sarcoma of soft tissue cell lines reveals characteristic up-regulation of potential new marker genes including ERBB3. Cancer Res. 2004;64(10):3395–405.
Tian T, et al. Determination of metastasis-associated proteins in non-small cell lung cancer by comparative proteomic analysis. Cancer Sci. 2007;98(8):1265–74.
Yao R, et al. The S100 proteins for screening and prognostic grading of bladder cancer. Histol Histopathol. 2007;22(9):1025–32.
Tyburczy ME, et al. Novel proteins regulated by mTOR in subependymal giant cell astrocytomas of patients with tuberous sclerosis complex and new therapeutic implications. Am J Pathol. 2010;176(4):1878–90.
Liu XG, et al. Ca2+-binding protein S100A11: a novel diagnostic marker for breast carcinoma. Oncol Rep. 2010;23(5):1301–8.
McKiernan E, et al. The role of S100 genes in breast cancer progression. Tumour Biol. 2011;32(3):441–50.
He H, et al. S100A11: diverse function and pathology corresponding to different target proteins. Cell Biochem Biophys. 2009;55(3):117–26.
Sakaguchi M, et al. Bifurcated converging pathways for high Ca2+-and TGFbeta-induced inhibition of growth of normal human keratinocytes. Proc Natl Acad Sci USA. 2005;102(39):13921–6.
Ohuchida K, et al. S100A11, a putative tumor suppressor gene, is overexpressed in pancreatic carcinogenesis. Clin Cancer Res. 2006;12(18):5417–22.
Jemal A, et al. Cancer statistics. CA Cancer J Clin. 2010;60(5):277–300.
Acknowledgments
This work was supported by grants from the Foundation for Talents in Six Fields of Jiangsu Province (No. 2006073), the Health Project of Jiangsu Province (H200923) and the Social Development Foundation of Nantong City (S2007028 and S2010012).
Author information
Authors and Affiliations
Corresponding author
Additional information
Ming-Bing Xiao and Feng Jiang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Xiao, MB., Jiang, F., Ni, WK. et al. High expression of S100A11 in pancreatic adenocarcinoma is an unfavorable prognostic marker. Med Oncol 29, 1886–1891 (2012). https://doi.org/10.1007/s12032-011-0058-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12032-011-0058-y