Abstract
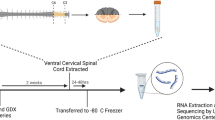
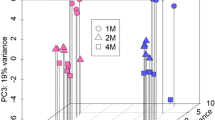
The spinal cord is essential for neuronal communication between the brain and rest of the body. To gain further insight into the molecular changes underpinning maturation of the mouse spinal cord, we analysed gene expression differences between 4 weeks of age (prior to puberty onset) and adulthood (8 weeks). We found 800 genes were significantly differentially expressed between juvenile and adult spinal cords. Gene ontology analysis revealed an overrepresentation of genes with roles in myelination and signal transduction among others. The expression of a further 19 genes was sexually dimorphic; these included both autosomal and sex-linked genes. Given the presence of steroid hormone receptors in the spinal cord, we also looked at the impact of two major steroid hormones, oestradiol and dihydrotestosterone (DHT) on spinal cord gene expression for selected genes. In gonadectomised male animals, implants with oestradiol and DHT produced significant changes to spinal cord gene expression. This study provides an overview of the global gene expression changes that occur as the spinal cord matures, over a key period of maturation. This confirms that both age and sex are important considerations in studies involving the spinal cord.






Similar content being viewed by others
References
Abreu AP, Kaiser UB (2016) Pubertal development and regulation. Lancet Diabetes Endocrinol 4:254–264. https://doi.org/10.1016/S2213-8587(15)00418-0
Akkiprik M, Feng Y, Wang H, Chen K, Hu L, Sahin A, Krishnamurthy S, Ozer A, Hao X, Zhang W (2008) Multifunctional roles of insulin-like growth factor binding protein 5 in breast cancer. Breast Cancer Res 10:212. https://doi.org/10.1186/bcr2116
Alaynick WA, Jessell TM, Pfaff SL (2011) SnapShot: spinal cord development. Cell 146:178–178 e171. https://doi.org/10.1016/j.cell.2011.06.038
Armoskus C, Moreira D, Bollinger K, Jimenez O, Taniguchi S, Tsai HW (2014) Identification of sexually dimorphic genes in the neonatal mouse cortex and hippocampus. Brain Res 1562:23–38. https://doi.org/10.1016/j.brainres.2014.03.017
Arnold AP (2009) The organizational-activational hypothesis as the foundation for a unified theory of sexual differentiation of all mammalian tissues. Horm Behav 55:570–578. https://doi.org/10.1016/j.yhbeh.2009.03.011
Arnold PM, Ma JY, Citron BA, Zoubine MN, Festoff BW (2000) Selective developmental regulation of gene expression for insulin-like growth factor-binding proteins in mouse spinal cord. Spine (Phila Pa 1976) 25:1765–1770
Barolo S, Stone T, Bang AG, Posakony JW (2002) Default repression and Notch signaling: hairless acts as an adaptor to recruit the corepressors Groucho and dCtBP to suppressor of hairless. Genes Dev 16:1964–1976. https://doi.org/10.1101/gad.987402
Bassani S, Folci A, Zapata J, Passafaro M (2013) AMPAR trafficking in synapse maturation and plasticity. Cell Mol Life Sci 70:4411–4430. https://doi.org/10.1007/s00018-013-1309-1
Berletch JB, Ma W, Yang F, Shendure J, Noble WS, Disteche CM, Deng X (2015) Escape from X inactivation varies in mouse tissues. PLoS Genet 11:e1005079. https://doi.org/10.1371/journal.pgen.1005079
Bettler B, Tiao JY (2006) Molecular diversity, trafficking and subcellular localization of GABAB receptors. Pharmacol Ther 110:533–543. https://doi.org/10.1016/j.pharmthera.2006.03.006
Byers JS, Huguenard AL, Kuruppu D, Liu NK, Xu XM, Sengelaub DR (2012) Neuroprotective effects of testosterone on motoneuron and muscle morphology following spinal cord injury. J Comp Neurol 520:2683–2696. https://doi.org/10.1002/cne.23066
Cerase A, Pintacuda G, Tattermusch A, Avner P (2015) Xist localization and function: new insights from multiple levels. Genome Biol 16:166. https://doi.org/10.1186/s13059-015-0733-y
Chen G, Fang X, Yu M (2015) Regulation of gene expression in rats with spinal cord injury based on microarray data. Mol Med Rep 12:2465–2472. https://doi.org/10.3892/mmr.2015.3670
Cheung TT, Weston MK, Wilson MJ (2017) Selection and evaluation of reference genes for analysis of mouse (Mus musculus) sex-dimorphic brain development. PeerJ 5:e2909. https://doi.org/10.7717/peerj.2909
Corre C, Shinoda G, Zhu H, Cousminer DL, Crossman C, Bellissimo C, Goldenberg A, Daley GQ, Palmert MR (2016) Sex-specific regulation of weight and puberty by the Lin28/let-7 axis. J Endocrinol 228:179–191. https://doi.org/10.1530/JOE-15-0360
Currer JM, Flurkey K, Leiter EH, Witham B (2009) The Jackson Laboratory handbook on genetically standardized mice, 6th edn. The Jackson Laboratory, Bar Harbor
Dawson MR, Polito A, Levine JM, Reynolds R (2003) NG2-expressing glial progenitor cells: an abundant and widespread population of cycling cells in the adult rat. CNS Mol Cell Neurosci 24:476–488
De Nicola AF, Gonzalez SL, Labombarda F, Gonzalez Deniselle MC, Garay L, Guennoun R, Schumacher M (2006) Progesterone treatment of spinal cord injury: effects on receptors, neurotrophins, and myelination. J Mol Neurosci 28:3–15. https://doi.org/10.1385/JMN:30:3:341
Dearolf WW 3rd, Betz RR, Vogel LC, Levin J, Clancy M, Steel HH (1990) Scoliosis in pediatric spinal cord-injured patients. J Pediatr Orthop 10:214–218
Dias TB, Yang YJ, Ogai K, Becker T, Becker CG (2012) Notch signaling controls generation of motor neurons in the lesioned spinal cord of adult zebrafish. J Neurosci 32:3245–3252. https://doi.org/10.1523/JNEUROSCI.6398-11.2012
Duncan GE, Stumpf WE (1984) Target neurons for [3H]corticosterone in the rat spinal cord. Brain Res 307:321–326
Figueroa JA, Jackson JG, McGuire WL, Krywicki RF, Yee D (1993) Expression of insulin-like growth factor binding proteins in human breast cancer correlates with estrogen receptor status. J Cell Biochem 52:196–205. https://doi.org/10.1002/jcb.240520211
Fu Y, Rusznak Z, Herculano-Houzel S, Watson C, Paxinos G (2013) Cellular composition characterizing postnatal development and maturation of the mouse brain and spinal cord. Brain Struct Funct 218:1337–1354. https://doi.org/10.1007/s00429-012-0462-x
Fukushima T, Tsuda M, Kofuji T, Hori Y (2011) Physiological properties of enkephalin-containing neurons in the spinal dorsal horn visualized by expression of green fluorescent protein in BAC transgenic mice. BMC Neurosci 12:36. https://doi.org/10.1186/1471-2202-12-36
Furukawa A, Miyatake A, Ohnishi T, Ichikawa Y (1998) Steroidogenic acute regulatory protein (StAR) transcripts constitutively expressed in the adult rat central nervous system: colocalization of StAR, cytochrome P-450SCC (CYP XIA1), and 3beta-hydroxysteroid dehydrogenase in the rat brain. J Neurochem 71:2231–2238
Furusho M, Ishii A, Bansal R (2017) Signaling by FGF receptor 2, not FGF receptor 1, regulates myelin thickness through activation of ERK1/2-MAPK, which promotes mTORC1 activity in an Akt-independent manner. J Neurosci 37:2931–2946. https://doi.org/10.1523/JNEUROSCI.3316-16.2017
Goldshmit Y, Tang J, Siegel AL, Nguyen PD, Kaslin J, Currie PD, Jusuf PR (2018) Different Fgfs have distinct roles in regulating neurogenesis after spinal cord injury in zebrafish. Neural Dev 13:24. https://doi.org/10.1186/s13064-018-0122-9
Hochman S (2007) Spinal cord. Curr Biol 17:R950–R955. https://doi.org/10.1016/j.cub.2007.10.014
Huang d W, Sherman BT, Lempicki RA (2009) Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat Protoc 4:44–57. https://doi.org/10.1038/nprot.2008.211
Huynh H (1998) Suppression of uterine insulin-like growth factor binding protein 5 by estrogen is mediated in part by insulin-like growth factor I. Int J Oncol 12:427–432
Jacobi A, Loy K, Schmalz AM, Hellsten M, Umemori H, Kerschensteiner M, Bareyre FM (2015) FGF22 signaling regulates synapse formation during post-injury remodeling of the spinal cord. EMBO J 34:1231–1243. https://doi.org/10.15252/embj.201490578
Kim HM, Hwang DH, Choi JY, Park CH, Suh-Kim H, Kim SU, Kim BG (2011) Differential and cooperative actions of Olig1 and Olig2 transcription factors on immature proliferating cells after contusive spinal cord injury. Glia 59:1094–1106. https://doi.org/10.1002/glia.21182
Kolde R (2019) pheatmap: pretty heatmaps. R package version 1.0.12. https://CRAN.R-project.org/package=pheatmap
Labombarda F, Garcia-Ovejero D (2014) Give progesterone a chance. Neural Regen Res 9:1422–1424. https://doi.org/10.4103/1673-5374.139456
Labombarda F, Meffre D, Delespierre B, Krivokapic-Blondiaux S, Chastre A, Thomas P, Pang Y, Lydon JP, Gonzalez SL, de Nicola AF, Schumacher M, Guennoun R (2010) Membrane progesterone receptors localization in the mouse spinal cord. Neuroscience 166:94–106. https://doi.org/10.1016/j.neuroscience.2009.12.012
Lancioni A, Pizzo M, Fontanella B, Ferrentino R, Napolitano LM, De Leonibus E, Meroni G (2010) Lack of Mid1, the mouse ortholog of the Opitz syndrome gene, causes abnormal development of the anterior cerebellar vermis. J Neurosci 30:2880–2887. https://doi.org/10.1523/JNEUROSCI.4196-09.2010
Levin ER (2011) Minireview: extranuclear steroid receptors: roles in modulation of cell functions. Mol Endocrinol 25:377–384. https://doi.org/10.1210/me.2010-0284
Li N, Leung GK (2015) Oligodendrocyte precursor cells in spinal cord injury: a review and update. Biomed Res Int 2015:235195. https://doi.org/10.1155/2015/235195
Li X, Floriddia EM, Toskas K, Fernandes KJL, Guerout N, Barnabe-Heider F (2016) Regenerative potential of ependymal cells for spinal cord injuries over time. EBioMedicine 13:55–65. https://doi.org/10.1016/j.ebiom.2016.10.035
Lindholm T, Cullheim S, Deckner M, Carlstedt T, Risling M (2002) Expression of neuregulin and ErbB3 and ErbB4 after a traumatic lesion in the ventral funiculus of the spinal cord and in the intact primary olfactory system. Exp Brain Res 142:81–90. https://doi.org/10.1007/s00221-001-0922-7
Liu R, Holik AZ, Su S, Jansz N, Chen K, Leong HS, Blewitt ME, Asselin-Labat ML, Smyth GK, Ritchie ME (2015) Why weight? Modelling sample and observational level variability improves power in RNA-seq analyses. Nucleic Acids Res 43:e97. https://doi.org/10.1093/nar/gkv412
Love MI, Huber W, Anders S (2014) Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol 15:550. https://doi.org/10.1186/s13059-014-0550-8
Lu QR, Yuk DI, Alberta JA, Zhu Z, Pawlitzky I, Chan J, McMahon AP, Stiles CD, Rowitch DH (2000) Sonic hedgehog—regulated oligodendrocyte lineage genes encoding bHLH proteins in the mammalian central nervous system. Neuron 25:317–329
Ludwig PE, Patil AA, Chamczuk AJ, Agrawal DK (2017) Hormonal therapy in traumatic spinal cord injury. Am J Transl Res 9:3881–3895
Ma JK, Post MW, Gorter JW, Martin Ginis KA (2016) Differences in health, participation and life satisfaction outcomes in adults following paediatric-versus adult-sustained spinal cord injury. Spinal Cord 54:1220. https://doi.org/10.1038/sc.2016.158
Maridas DE, DeMambro VE, Le PT, Nagano K, Baron R, Mohan S, Rosen CJ (2017) IGFBP-4 regulates adult skeletal growth in a sex-specific manner. J Endocrinol 233:131–144. https://doi.org/10.1530/JOE-16-0673
Meletis K, Barnabe-Heider F, Carlen M, Evergren E, Tomilin N, Shupliakov O, Frisen J (2008) Spinal cord injury reveals multilineage differentiation of ependymal cells. PLoS Biol 6:e182. https://doi.org/10.1371/journal.pbio.0060182
Mologni L, Salmikangas P, Fougerousse F, Beckmann JS, Carpen O (2001) Developmental expression of myotilin, a gene mutated in limb-girdle muscular dystrophy type 1A. Mech Dev 103:121–125
Morel V, Schweisguth F (2000) Repression by suppressor of hairless and activation by Notch are required to define a single row of single-minded expressing cells in the Drosophila embryo. Genes Dev 14:377–388
Narkis G, Ofir R, Manor E, Landau D, Elbedour K, Birk OS (2007) Lethal congenital contractural syndrome type 2 (LCCS2) is caused by a mutation in ERBB3 (Her3), a modulator of the phosphatidylinositol-3-kinase/Akt pathway. Am J Hum Genet 81:589–595. https://doi.org/10.1086/520770
Niciu MJ, Kelmendi B, Sanacora G (2012) Overview of glutamatergic neurotransmission in the nervous system. Pharmacol Biochem Behav 100:656–664. https://doi.org/10.1016/j.pbb.2011.08.008
Noble LJ, Donovan F, Igarashi T, Goussev S, Werb Z (2002) Matrix metalloproteinases limit functional recovery after spinal cord injury by modulation of early vascular events. J Neurosci 22:7526–7535
Papka RE, Mowa CN (2003) Estrogen receptors in the spinal cord, sensory ganglia, and pelvic autonomic ganglia. Int Rev Cytol 231:91–127
Parent S, Mac-Thiong JM, Roy-Beaudry M, Sosa JF, Labelle H (2011) Spinal cord injury in the pediatric population: a systematic review of the literature. J Neurotrauma 28:1515–1524. https://doi.org/10.1089/neu.2009.1153
Patte-Mensah C, Kappes V, Freund-Mercier MJ, Tsutsui K, Mensah-Nyagan AG (2003) Cellular distribution and bioactivity of the key steroidogenic enzyme, cytochrome P450side chain cleavage, in sensory neural pathways. J Neurochem 86:1233–1246
Quartier A, Chatrousse L, Redin C, Keime C, Haumesser N, Maglott-Roth A, Brino L, le Gras S, Benchoua A, Mandel JL, Piton A (2018) Genes and pathways regulated by androgens in human neural cells, potential candidates for the male excess in autism spectrum disorder. Biol Psychiatry 84:239–252. https://doi.org/10.1016/j.biopsych.2018.01.002
Riccomagno MM, Kolodkin AL (2015) Sculpting neural circuits by axon and dendrite pruning. Annu Rev Cell Dev Biol 31:779–805. https://doi.org/10.1146/annurev-cellbio-100913-013038
Robinson MD, McCarthy DJ, Smyth GK (2010) edgeR: a bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26:139–140. https://doi.org/10.1093/bioinformatics/btp616
Rouillard AD, Gundersen GW, Fernandez NF, Wang Z, Monteiro CD, McDermott MG, Ma’ayan A (2016) The harmonizome: a collection of processed datasets gathered to serve and mine knowledge about genes and proteins. Database 2016:baw100–baw100. https://doi.org/10.1093/database/baw100
Rusanescu G (2016) Adult spinal cord neurogenesis: a regulator of nociception. Neurogenesis (Austin) 3:e1256853. https://doi.org/10.1080/23262133.2016.1256853
Salih DA et al (2005) Insulin-like growth factor-binding protein-5 induces a gender-related decrease in bone mineral density in transgenic mice. Endocrinology 146:931–940. https://doi.org/10.1210/en.2004-0816
Schonemann MD, Muench MO, Tee MK, Miller WL, Mellon SH (2012) Expression of P450c17 in the human fetal nervous system. Endocrinology 153:2494–2505. https://doi.org/10.1210/en.2011-1545
Schweiger S, Foerster J, Lehmann T, Suckow V, Muller YA, Walter G, Davies T, Porter H, van Bokhoven H, Lunt PW, Traub P, Ropers HH (1999) The Opitz syndrome gene product, MID1, associates with microtubules. Proc Natl Acad Sci U S A 96:2794–2799
Segarra AC, Acosta AM, Gonzalez JL, Angulo JA, McEwen BS (1998) Sex differences in estrogenic regulation of preproenkephalin mRNA levels in the medial preoptic area of prepubertal rats. Brain Res Mol Brain Res 60:133–139
Sengul G, Watson C (2012) Spinal cord. In: Watson C, Paxinos G, Puelles L (eds) The mouse nervous system. Elsevier, London, pp 424–450
Sisk CL, Zehr JL (2005) Pubertal hormones organize the adolescent brain and behavior. Front Neuroendocrinol 26:163–174. https://doi.org/10.1016/j.yfrne.2005.10.003
Squair JW, Tigchelaar S, Moon KM, Liu J, Tetzlaff W, Kwon BK, Krassioukov AV, West CR, Foster LJ, Skinnider MA (2018) Integrated systems analysis reveals conserved gene networks underlying response to spinal cord injury. Elife 7. https://doi.org/10.7554/eLife.39188
Sribnick EA, Samantaray S, Das A, Smith J, Matzelle DD, Ray SK, Banik NL (2010) Postinjury estrogen treatment of chronic spinal cord injury improves locomotor function in rats. J Neurosci Res 88:1738–1750. https://doi.org/10.1002/jnr.22337
Sunkin SM, Ng L, Lau C, Dolbeare T, Gilbert TL, Thompson CL, Hawrylycz M, Dang C (2013) Allen Brain Atlas: an integrated spatio-temporal portal for exploring the central nervous system. Nucleic Acids Res 41:D996–D1008. https://doi.org/10.1093/nar/gks1042
Supek F, Bosnjak M, Skunca N, Smuc T (2011) REVIGO summarizes and visualizes long lists of gene ontology terms. PLoS One 6:e21800. https://doi.org/10.1371/journal.pone.0021800
Tennekes M (2017) treemap: treemap visualization. R package version 24-2. https://CRAN.R-project.org/package=treemap
Uli N, Michelen-Gomez E, Ramos EI, Druley TE (2018) Age-specific changes in genome-wide methylation enrich for Foxa2 and estrogen receptor alpha binding sites. PLoS One 13:e0203147. https://doi.org/10.1371/journal.pone.0203147
Volk B, Hettmannsperger U, Papp T, Amelizad Z, Oesch F, Knoth R (1991) Mapping of phenytoin-inducible cytochrome P450 immunoreactivity in the mouse central nervous system. Neuroscience 42:215–235
Yamamoto S, Nagao M, Sugimori M, Kosako H, Nakatomi H, Yamamoto N, Takebayashi H, Nabeshima YI, Kitamura T, Weinmaster G, Nakamura K, Nakafuku M (2001) Transcription factor expression and Notch-dependent regulation of neural progenitors in the adult rat spinal cord. J Neurosci 21:9814–9823
Yang F, Babak T, Shendure J, Disteche CM (2010) Global survey of escape from X inactivation by RNA-sequencing in mouse. Genome Res 20:614–622. https://doi.org/10.1101/gr.103200.109
Yu L, Romero DG, Gomez-Sanchez CE, Gomez-Sanchez EP (2002) Steroidogenic enzyme gene expression in the human brain. Mol Cell Endocrinol 190:9–17
Zhang YH, Song J, Wang LG, Shao J (2017) Identification of key genes and pathways associated with spinal cord injury. Mol Med Rep 15:1577–1584. https://doi.org/10.3892/mmr.2017.6192
Zhou Q, Wang S, Anderson DJ (2000) Identification of a novel family of oligodendrocyte lineage-specific basic helix-loop-helix transcription factors. Neuron 25:331–343
Acknowledgements
We thank Lyvianne Decouryte, Rebecca Clarke and Susie Szakats for comments on the manuscript, and Megan Inglis for making the oestrogen implants. This study was funded by a University of Otago Research Grant and the Wishbone Trust (New Zealand Orthopaedic society).
Author information
Authors and Affiliations
Contributions
MJW, JML and GA co-wrote the paper. JML and GA performed the gonadectomy surgeries. MJW carried out the RNA-seq and analysis. JML did the RT-qPCR validation.
Corresponding author
Ethics declarations
Ethics Statement
All procedures and tissue samples were collected under the approval of University of Otago Animal Ethics Committee.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
ESM 1
(DOCX 7086 kb)
Rights and permissions
About this article
Cite this article
McCallum-Loudeac, J., Anderson, G. & Wilson, M.J. Age and Sex-Related Changes to Gene Expression in the Mouse Spinal Cord. J Mol Neurosci 69, 419–432 (2019). https://doi.org/10.1007/s12031-019-01371-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12031-019-01371-3