Abstract
Introduction/Background
Colorectal carcinoma (CRC) is a common malignancy, with its diverse clinical, pathological, and molecular features. The immune microenvironment of a tumor comprises of interplay between various cells and molecules, and has a significant role in deciding the tumor behavior and overall prognosis. PD-L1 (programmed cell death ligand-1) has been implicated in the regulation of the tumor immune microenvironment (TIME). There is limited data regarding the correlation of PD-L1 expression with immune cell profile in CRCs, especially in the Indian setting. The study aimed to assess the PD-L1 expression in CRC tumor cells and its association with TIME, mismatch repair (MMR), and various other clinicopathological parameters.
Methods
This is a hospital-based, cross-sectional observational study. PD-L1 expression was assessed at the protein level by immunohistochemistry and mRNA level by qRT-PCR. Immune cell markers (CD4, CD8, CD20, FOXP3, and CD163) were interpreted using the ImageJ Fiji platform.
Results
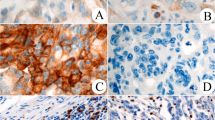
Of the 104 cases, 21% were PD-L1 positive and were more common in right-sided CRCs. PD-L1 positive cases showed significantly higher concentrations of all T-cell subsets (CD4+ , CD8+ , and FOXP3+), CD20+ B-cells, and CD163+ macrophages were noted. No statistical significance was seen between PD-L1 expression with clinical profile, pathological subtype, grade or stage, mismatch repair status (proficient vs deficient), and survival.
Conclusions
The present study showed a relatively lower frequency of PD-L1 in CRC from the Eastern Indian cohort. The immune cell concentration in the present study was calculated using image analysis-based objectivised methods. Significant correlation of PD-L1 expression in tumor cells with the tumor-infiltrating immune cells indicated its crucial role in the pathobiology of CRC especially by regulating the TIME. Considering the therapeutic implication of PD-L1 in various malignancies, it may be one of the crucial therapeutic targets in a proportion of cases.




Similar content being viewed by others
Data Availability
The master data sheet used to arrive at the conclusions of the study is available with the corresponding author, and the same may be made available, upon reasonable request.
References
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209–49.
Sinha R, Doval DC, Hussain S, Kumar K, Singh S, Basir SF, Bharadwaj M. Lifestyle and sporadic colorectal cancer in India. Asian Pac J Cancer Prev. 2015;16(17):7683–8.
Markowitz SD, Bertagnolli MM. Molecular origins of cancer: molecular basis of colorectal cancer. N Engl J Med. 2009;361(25):2449–60.
Müller MF, Ibrahim AE, Arends MJ. Molecular pathological classification of colorectal cancer. Virchows Arch. 2016;469:125–34.
Guinney J, Dienstmann R, Wang X, de Reyniès A, Schlicker A, Soneson C, et al. The consensus molecular subtypes of colorectal cancer. Nat Med. 2015;21(11):1350–6.
Akiyama M, Matsuda Y, Arai T, Saeki H. PD-L1 expression in malignant melanomas of the skin and gastrointestinal tract. Oncol Lett. 2020;19(3):2481–8.
Yuan C, Liu Z, Yu Q, Wang X, Bian M, Yu Z, Yu J. Expression of PD-1/PD-L1 in primary breast tumours and metastatic axillary lymph nodes and its correlation with clinicopathological parameters. Sci Rep. 2019;9(1):14356.
Pawelczyk K, Piotrowska A, Ciesielska U, Jablonska K, Gletzel-Plucinska N, Grzegrzolka J, et al. Role of PD-L1 expression in non-small cell lung cancer and their prognostic significance according to clinicopathological factors and diagnostic markers. Int J Mol Sci. 2019;20(4):824.
Liu X, Choi MG, Kim K, Kim KM, Kim ST, Park SH, Cristescu R, Peter S, Lee J. High PD-L1 expression in gastric cancer (GC) patients and correlation with molecular features. Pathol Res Pract. 2020;216(4): 152881.
Srivastava P, Husain N, Shukla S, Chauhan S, Pandey A, Masood S. PD-L1 Expression in colorectal carcinoma and its correlation with clinicopathological parameters, microsatellite instability and BRAF mutation. Indian J Pathol Microbiol. 2021;64(3):490–6.
Shan T, Chen S, Wu T, Yang Y, Li S, Chen X. PD-L1 expression in colon cancer and its relationship with clinical prognosis. Int J Clin Exp Pathol. 2019;12(5):1764–9 Int J Clin Exp Pathol. 2022;15(3):155–156.
Gupta M, Manjari M, Kaur H. PD-L1 Expression in colorectal carcinoma: immunohistochemical study. Annals of Pathology and Laboratory Medicine. 2020;7:A275-281.
Salemme V, Centonze G, Cavallo F, Defilippi P, Conti L. The crosstalk between tumor cells and the immune microenvironment in breast cancer: implications for immunotherapy. Front Oncol. 2021;11: 610303.
Soldevilla B, Carretero-Puche C, Gomez-Lopez G, Al-Shahrour F, Riesco MC, Gil-Calderon B, et al. The correlation between immune subtypes and consensus molecular subtypes in colorectal cancer identifies novel tumour microenvironment profiles, with prognostic and therapeutic implications. Eur J Cancer. 2019;123:118–29.
Binnewies M, Roberts EW, Kersten K, Chan V, Fearon DF, Merad M, et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med. 2018;24(5):541–50.
Jiang X, Wang J, Deng X, Xiong F, Ge J, Xiang B, et al. Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer. 2019;18(1):1–7.
Scognamiglio T, Chen YT. Beyond the percentages of PD-L1-positive tumor cells: induced versus constitutive PD-L1 expression in primary and metastatic head and neck squamous cell carcinoma. Head Neck Pathol. 2018;12(2):221–9.
Lin G, Fan X, Zhu W, Huang C, Zhuang W, Xu H, et al. Prognostic significance of PD-L1 expression and tumor infiltrating lymphocyte in surgically resectable non-small cell lung cancer. Oncotarget. 2017;8(48):83986.
Mastracci L, Grillo F, Parente P, Gullo I, Campora M, Angerilli V, et al. PD-L1 evaluation in the gastrointestinal tract: from biological rationale to its clinical application. Pathologica. 2022;114(5):352–64.
Zlobec I, Steele R, Terracciano L, Jass JR, Lugli A. Selecting immunohistochemical cut-off scores for novel biomarkers of progression and survival in colorectal cancer. J Clin Pathol. 2007;60:1112–6.
Agrawal L, Engel KB, Greytak SR, Moore HM. Understanding preanalytical variables and their effects on clinical biomarkers of oncology and immunotherapy. Semin Cancer Biol. 2018;52(Pt 2):26–38.
Huang CY, Wang Y, Luo GY, Han F, Li YQ, Zhou ZG, et al. Relationship between PD-L1 expression and CD8+ T-cell immune responses in hepatocellular carcinoma. J Immunother. 2017;40(9):323–33.
Bai J, Chen H, Bai X. Relationship between microsatellite status and immune microenvironment of colorectal cancer and its application to diagnosis and treatment. J Clin Lab Anal. 2021;35(6): e23810.
Deng M, Li SH, Fu X, Yan XP, Chen J, Qiu YD, Guo RP. Relationship between PD-L1 expression, CD8+ T-cell infiltration and prognosis in intrahepatic cholangiocarcinoma patients. Cancer Cell Int. 2021;21(1):371.
Que Y, Xiao W, Guan YX, Liang Y, Yan SM, Chen HY, et al. PD-L1 expression is associated with FOXP3+ regulatory T-cell infiltration of soft tissue sarcoma and poor patient prognosis. J Cancer. 2017;8(11):2018–25.
Sumitomo R, Hirai T, Fujita M, Murakami H, Otake Y, Huang CL. PD-L1 expression on tumor-infiltrating immune cells is highly associated with M2 TAM and aggressive malignant potential in patients with resected non-small cell lung cancer. Lung Cancer. 2019;136:136–44.
Arias-Pulido H, Cimino-Mathews A, Chaher N, Qualls C, Joste N, Colpaert C, Marotti JD, Foisey M, Prossnitz ER, Emens LA, Fiering S. The combined presence of CD20 + B cells and PD-L1 + tumor-infiltrating lymphocytes in inflammatory breast cancer is prognostic of improved patient outcome. Breast Cancer Res Treat. 2018;171(2):273–82.
Guo G, Wang Y, Zhou Y, Quan Q, Zhang Y, Wang H, Zhang B, Xia L. Immune cell concentrations among the primary tumor microenvironment in colorectal cancer patients predicted by clinicopathologic characteristics and blood indexes. J Immunother Cancer. 2019;7(1):179.
Takasu C, Nishi M, Yoshikawa K, Tokunaga T, Kashihara H, Yoshimoto T, Shimada M. Impact of sidedness of colorectal cancer on tumor immunity. PLoS ONE. 2020;15(10): e0240408.
Valentini AM, Di Pinto F, Cariola F, Guerra V, Giannelli G, Caruso ML, Pirrelli M. PD-L1 expression in colorectal cancer defines three subsets of tumor immune microenvironments. Oncotarget. 2018;9(9):8584–96.
Chen Y, Liu Q, Chen Z, Wang Y, Yang W, Hu Y, Han W, Zeng H, Ma H, Dai J, Zhang H. PD-L1 expression and tumor mutational burden status for prediction of response to chemotherapy and targeted therapy in non-small cell lung cancer. J Exp Clin Cancer Res. 2019;38(1):193.
Acknowledgements
I would like to express my sincere gratitude towards all my teachers and colleagues from All India Institute of Medical Sciences, Bhubaneswar, for their mental support during the study. I would like to express my sincere gratitude to Dr. Mukund Namdev Sable, Additional Professor, for providing the kits for mRNA analysis, and Dr. Kirtal Hansdah and Dr. Swetasmita Mishra, the scientists at the molecular lab, and Mr. Samiur Rahman, for the technical aspects. My sincere gratitude towards all the technical staff of the Histopathology Division of the Department of Pathology, AIIMS Bhubaneswar, for their help with all the technical works related to the study, and to the participants of the study. I would acknowledge the creators of the ImageJ (Fiji) software, which is made available free of cost, without which this study would not have been possible.
Author information
Authors and Affiliations
Contributions
Prefix | Author name | Name abbreviation |
|---|---|---|
Dr. | Mohammed Shahin | MS |
Dr. | Susama Patra | SP |
Dr. | Suvendu Purkait | SuP |
Dr. | Madhabananda Kar | MK |
Dr. | Saroj Kumar Das Majumdar | SKM |
Dr. | Tushar Subhadarshan Mishra | TSM |
Dr. | Subash Chandra Samal | SCS |
Dr. | Hemanta Kumar Nayak | HKN |
• All the authors contributed to the study’s conception and design.
• Material preparation, data collection, and analysis were performed by Mohammed Shahin, Susama Patra, and Suvendu Purkait.
• Case management data and specimens were provided by Madhabananda Kar, Saroj Kumar Das Majumdar, Tushar Subhadarshan Mishra, Subash Chandra Samal, and Hemanta Kumar Nayak.
• Case follow-up data were provided by Madhabananda Kar and Saroj Kumar Das Majumdar.
• The first draft of the manuscript was written by Mohammed Shahin, and initially assessed by Susama Patra and Suvendu Purkait.
• Histopathological evaluation and IHC interpretation were done by Mohammed Shahin, Susama Patra, and Suvendu Purkait.
• The master chart was prepared by Mohammed Shahin and statistical analysis was performed by Suvendu Purkait
• All other previous versions were commented upon, and approved by all the authors.
Corresponding author
Ethics declarations
Ethics Approval
Ethical approval has been taken from the AIIMS Bhubaneswar Institutional Ethics Committee in February 2020.
Consent to Participate
Informed consent was obtained from all the individual participants included in the study, before their routine diagnostic evaluation and surgical procedure.
Consent to Publish
The authors declare that the participants have provided consent for the publication of images. However, only images of the histopathology slides, IHCs, and ImageJ were included in the study, and duly acknowledged.
Competing Interests
The authors declare no competing interests.
Credits
This paper was presented at the Indian Association of Pathologists and Microbiologists Annual Conference APCON 2022, held in Bengaluru, India, in December 2022, in the “Best Award Paper” category.
The initial abstract of the study was published in the Indian Journal of Pathology and Microbiology. 65(Suppl 2): S1, November 2022.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shahin, M., Patra, S., Purkait, S. et al. PD-L1 Expression in Colorectal Carcinoma Correlates with the Immune Microenvironment. J Gastrointest Canc (2024). https://doi.org/10.1007/s12029-024-01049-z
Accepted:
Published:
DOI: https://doi.org/10.1007/s12029-024-01049-z