Abstract
Objective
Platelets have been implicated in cancer metastasis and prognosis. No population-based study has been reported as to whether preoperative platelet count directly predicts metastatic recurrence of colorectal cancer (CRC) patients.
Design
Using a well-characterized cohort of 1,513 surgically resected CRC patients, we assessed the predictive roles of preoperative platelet count in overall survival, overall recurrence, as well as locoregional and distant metastatic recurrences.
Results
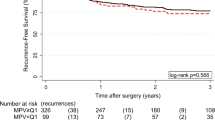
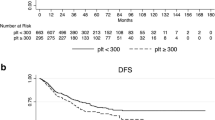
Patients with clinically high platelet count (≥400 × 109/L) measured within 1 month before surgery had a significantly unfavorable survival (hazard ratio [HR] = 1.66, 95 % confidence interval [CI] 1.34–2.05, P = 2.6 × 10−6, P log rank = 1.1 × 10−11) and recurrence (HR = 1.90, 1.24–2.93, P = 0.003, P log rank = 0.003). The association of platelet count with recurrence was evident only in patients with metastatic (HR = 2.81, 1.67–4.74, P = 1.1 × 10−4, P log rank = 2.6 × 10−6) but not locoregional recurrence (HR = 0.59, 95 % CI 0.21–1.68, P = 0.325, P log rank = 0.152). The findings were internally validated through bootstrap resampling (P < 0.01 at 98.6 % of resampling). Consistently, platelet count was significantly higher in deceased than living patients (P < 0.0001) and in patients with metastatic recurrence than locoregional (P = 0.004) or nonrecurrent patients (P < 0.0001). Time-dependent modeling indicated that the increased risks for death and metastasis associated with elevated preoperative platelet counts persisted up to 5 years after surgery.
Conclusion
Our data demonstrated that clinically high level of preoperative platelets was an independent predictor of CRC survival and metastasis. As an important component of the routinely tested complete blood count panel, platelet count may be a cost-effective and noninvasive marker for CRC prognosis and a potential intervention target to prevent metastatic recurrence.




Similar content being viewed by others
References
Siegel R, Ward E, Brawley O, Jemal A. Cancer statistics, 2011: the impact of eliminating socioeconomic and racial disparities on premature cancer deaths. CA Cancer J Clin. 2011;61:212–36.
Walther A, Johnstone E, Swanton C, Midgley R, Tomlinson I, Kerr D. Genetic prognostic and predictive markers in colorectal cancer. Nature reviews. Cancer. 2009;9:489–99.
Chau I, Allen MJ, Cunningham D, Norman AR, Brown G, Ford HE, et al. The value of routine serum carcino-embryonic antigen measurement and computed tomography in the surveillance of patients after adjuvant chemotherapy for colorectal cancer. J Clin Oncol: official j Am Soc Clin Oncol. 2004;22:1420–9.
Arriola E, Navarro M, Pares D, Munoz M, Pareja L, Figueras J, et al. Imaging techniques contribute to increased surgical rescue of relapse in the follow-up of colorectal cancer. Dis Colon Rectum. 2006;49:478–84.
Rodriguez-Moranta F, Salo J, Arcusa A, Boadas J, Pinol V, Bessa X, et al. Postoperative surveillance in patients with colorectal cancer who have undergone curative resection: a prospective, multicenter, randomized, controlled trial. J Clin Oncol: official j Am Soc Clin Oncol. 2006;24:386–93.
Safi F, Beyer HG. The value of follow-up after curative surgery of colorectal carcinoma. Cancer Detect Prev. 1993;17:417–24.
Chaffer CL, Weinberg RA. A perspective on cancer cell metastasis. Science. 2011;331:1559–64.
Chambers AF, Groom AC, MacDonald IC. Dissemination and growth of cancer cells in metastatic sites. Nature reviews. Cancer. 2002;2:563–72.
Fidler IJ. The pathogenesis of cancer metastasis: the 'seed and soil' hypothesis revisited. Nature reviews. Cancer. 2003;3:453–8.
Gupta GP, Massague J. Cancer metastasis: building a framework. Cell. 2006;127:679–95.
Joyce JA, Pollard JW. Microenvironmental regulation of metastasis. Nature reviews. Cancer. 2009;9:239–52.
Nguyen DX, Bos PD, Massague J. Metastasis: from dissemination to organ-specific colonization. Nature reviews. Cancer. 2009;9:274–84.
Nash GF, Turner LF, Scully MF, Kakkar AK. Platelets and cancer. Lancet Oncol. 2002;3:425–30.
Gupta GP, Massague J. Platelets and metastasis revisited: a novel fatty link. J Clin Invest. 2004;114:1691–3.
Erpenbeck L, Schon MP. Deadly allies: the fatal interplay between platelets and metastasizing cancer cells. Blood. 2010;115:3427–36.
Gay LJ, Felding-Habermann B. Contribution of platelets to tumour metastasis. Nature reviews. Cancer. 2011;11:123–34.
Bambace NM, Holmes CE. The platelet contribution to cancer progression. J Thromb Haemost. 2011;9:237–49.
Labelle M, Begum S, Hynes RO. Direct signaling between platelets and cancer cells induces an epithelial–mesenchymal-like transition and promotes metastasis. Cancer Cell. 2011;20:576–90.
Monreal M, Fernandez-Llamazares J, Pinol M, Julian JF, Broggi M, Escola D, et al. Platelet count and survival in patients with colorectal cancer—a preliminary study. Thromb Haemost. 1998;79:916–8.
Sasaki K, Kawai K, Tsuno NH, Sunami E, Kitayama J. Impact of preoperative thrombocytosis on the survival of patients with primary colorectal cancer. World J Surg. 2011;36:192–200.
Symbas NP, Townsend MF, El-Galley R, Keane TE, Graham SD, Petros JA. Poor prognosis associated with thrombocytosis in patients with renal cell carcinoma. BJU Int. 2000;86:203–7.
Stone RL, Nick AM, McNeish IA, Balkwill F, Han HD, Bottsford-Miller J, et al. Paraneoplastic thrombocytosis in ovarian cancer. N Engl J Med. 2012;366:610–8.
Gucer F, Moser F, Tamussino K, Reich O, Haas J, Arikan G, et al. Thrombocytosis as a prognostic factor in endometrial carcinoma. Gynecol Oncol. 1998;70:210–4.
Lopes A, Daras V, Cross PA, Robertson G, Beynon G, Monaghan JM. Thrombocytosis as a prognostic factor in women with cervical cancer. Cancer. 1994;74:90–2.
Sagman U, Maki E, Evans WK, Warr D, Shepherd FA, Sculier JP, et al. Small-cell carcinoma of the lung: derivation of a prognostic staging system. J Clin Oncol: official j Am Soc Clin Oncol. 1991;9:1639–49.
Pedersen LM, Milman N. Prognostic significance of thrombocytosis in patients with primary lung cancer. EurRespir j:official J Eur Soc Clin Respir. 1996;9:1826–30.
Henderson AR. The bootstrap: a technique for data-driven statistics. Using computer-intensive analyses to explore experimental data. Clin Chim Acta Int J Clin Chem. 2005;359:1–26.
Kaushansky K. Historical review: megakaryopoiesis and thrombopoiesis. Blood. 2008;111:981–6.
Schafer AI. Thrombocytosis. N Engl J Med. 2004;350:1211–9.
Royston P. A strategy for modelling the effect of a continuous covariate in medicine and epidemiology. Stat Med. 2000;19:1831–47.
Davi G, Patrono C. Platelet activation and atherothrombosis. N Engl J Med. 2007;357:2482–94.
Skoda RC. Thrombocytosis. Hematology Am Soc Hematol Educ Program 2009:159–67.
O'Keefe SC, Marshall FF, Issa MM, Harmon MP, Petros JA. Thrombocytosis is associated with a significant increase in the cancer specific death rate after radical nephrectomy. J Urol. 2002;168:1378–80.
Palumbo JS, Talmage KE, Massari JV, La Jeunesse CM, Flick MJ, Kombrinck KW, et al. Platelets and fibrin(ogen) increase metastatic potential by impeding natural killer cell-mediated elimination of tumor cells. Blood. 2005;105:178–85.
Nieswandt B, Hafner M, Echtenacher B, Mannel DN. Lysis of tumor cells by natural killer cells in mice is impeded by platelets. Cancer Res. 1999;59:1295–300.
Mohle R, Green D, Moore MA, Nachman RL, Rafii S. Constitutive production and thrombin-induced release of vascular endothelial growth factor by human megakaryocytes and platelets. Proc Natl Acad Sci U S A. 1997;94:663–8.
Nierodzik ML, Karpatkin S. Thrombin induces tumor growth, metastasis, and angiogenesis: evidence for a thrombin-regulated dormant tumor phenotype. Cancer Cell. 2006;10:355–62.
Boucharaba A, Serre CM, Gres S, Saulnier-Blache JS, Bordet JC, Guglielmi J, et al. Platelet-derived lysophosphatidic acid supports the progression of osteolytic bone metastases in breast cancer. J Clin Invest. 2004;114:1714–25.
Valastyan S, Weinberg RA. Tumor metastasis: molecular insights and evolving paradigms. Cell. 2011;147:275–92.
Griesshammer M, Bangerter M, Sauer T, Wennauer R, Bergmann L, Heimpel H. Aetiology and clinical significance of thrombocytosis: analysis of 732 patients with an elevated platelet count. J Intern Med. 1999;245:295–300.
Sierko E, Wojtukiewicz MZ. Platelets and angiogenesis in malignancy. Semin Thromb Hemost. 2004;30:95–108.
Acknowledgments
The work was supported by National Cancer Institute Grants CA153099, CA152703, and CA162201 and a Research Scholar Award from the V Foundation for Cancer Research.
Conflict of Interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Shaogui Wan and Yinzhi Lai contributed equally to this manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 309 kb)
Rights and permissions
About this article
Cite this article
Wan, S., Lai, Y., Myers, R.E. et al. Preoperative Platelet Count Associates with Survival and Distant Metastasis in Surgically Resected Colorectal Cancer Patients. J Gastrointest Canc 44, 293–304 (2013). https://doi.org/10.1007/s12029-013-9491-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-013-9491-9