Abstract
Background
Most existing studies have focused on the correlation between white matter lesion (WML) and baseline intraventricular hemorrhage (IVH) in patients with intracerebral hemorrhage (ICH), whereas few studies have investigated the relationship between WML severity and delayed IVH after admission. This study aimed to investigate the correlation between WML severity and delayed IVH and to verify the association between WML and baseline IVH.
Methods
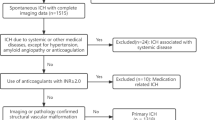
A total of 480 patients with spontaneous ICH from February 2018 to October 2020 were selected. WML was scored using the Van Swieten Scale, with scores of 0–2 representing nonslight WML and scores of 3–4 representing moderate-severe WML. We determined the presence of IVH on baseline (< 6 h) and follow-up computed tomography (< 72 h) images. Univariate analysis and multiple logistic regression were used to analyze the influencing factors of baseline and delayed IVH.
Results
Among 480 patients with ICH, 172 (35.8%) had baseline IVH, and there was a higher proportion of moderate-severe WML in patients with baseline IVH (20.3%) than in those without baseline IVH (12.7%) (P = 0.025). Among 308 patients without baseline IVH, delayed IVH was found in 40 patients (12.9%), whose proportion of moderate-severe WML (25.0%) was higher than that in patients without delayed IVH (10.8%) (P = 0.012). Multiple logistic regression results showed that moderate-severe WML was independently correlated with baseline IVH (P = 0.006, odds ratio = 2.266, 95% confidence interval = 1.270–4.042) and delayed IVH (P = 0.002, odds ratio = 7.009, 95% confidence interval = 12.086–23.552).
Conclusions
Moderate-severe WML was an independent risk factor for delayed IVH as well as baseline IVH.



Similar content being viewed by others
References
Pantoni L. Cerebral small vessel disease: from pathogenesis and clinical characteristics to therapeutic challenges. Lancet Neurol. 2010;9(7):689–701.
Paolini Paoletti F, Simoni S, Parnetti L, Gaetani L. The contribution of small vessel disease to neurodegeneration: focus on Alzheimer’s disease, Parkinson’s disease and multiple sclerosis. Int J Mol Sci. 2021;22(9):4958.
Wang Y, Yan X, Zhan J, et al. Neuroimaging markers of cerebral small vessel disease on hemorrhagic transformation and functional outcome after intravenous thrombolysis in patients with acute ischemic stroke: a systematic review and meta-analysis. Front Aging Neurosci. 2021;13:692942.
Wardlaw JM, Smith C, Dichgans M. Small vessel disease: mechanisms and clinical implications. Lancet Neurol. 2019;18(7):684–96.
Lin J, Wang D, Lan L, Fan Y. Multiple Factors Involved in the Pathogenesis of White Matter Lesions. Biomed Res Int. 2017;2017:9372050.
Etherton MR, Wu O, Rost NS. Recent advances in leukoaraiosis: white matter structural integrity and functional outcomes after acute ischemic stroke. Curr Cardiol Rep. 2016;18(12):123.
Wong SM, Jansen JFA, Zhang CE, et al. Blood-brain barrier impairment and hypoperfusion are linked in cerebral small vessel disease. Neurology. 2019;92(15):e1669–77.
Zuo S, Pan P, Li Q, Chen Y, Feng H. White matter injury and recovery after hypertensive intracerebral hemorrhage. Biomed Res Int. 2017;2017:6138424.
Chen Y, Yan S, Xu M, Zhong G, Liebeskind DS, Lou M. More extensive white matter hyperintensity is linked with higher risk of remote intracerebral hemorrhage after intravenous thrombolysis. Eur J Neurol. 2018;25(2):380-e15.
Lou M, Al-Hazzani A, Goddeau RP, Novak V, Selim M. Relationship between white-matter hyperintensities and hematoma volume and growth in patients with intracerebral hemorrhage. Stroke. 2010;41(1):34–40.
Sykora M, Herweh C, Steiner T. The association between leukoaraiosis and poor outcome in intracerebral hemorrhage is not mediated by hematoma growth. J Stroke Cerebrovasc Dis. 2017;26(6):1328–33.
Hansen BM, Ullman N, Muschelli J, et al. Relationship of white matter lesions with intracerebral hemorrhage expansion and functional outcome: MISTIE II and CLEAR III. Neurocrit Care. 2020;33(2):516–24.
Zhang Y, Wang Y, Ji R, et al. In-hospital complications affect short-term and long-term mortality in ICH: a prospective cohort study. Stroke Vasc Neurol. 2021;6(2):201–6.
Hemphill JC 3rd, Greenberg SM, Anderson CS, et al. Guidelines for the management of spontaneous intracerebral hemorrhage: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2015;46(7):2032–60.
Trifan G, Arshi B, Testai FD. Intraventricular hemorrhage severity as a predictor of outcome in intracerebral hemorrhage. Front Neurol. 2019;10:217.
Hallevi H, Albright KC, Aronowski J, et al. Intraventricular hemorrhage: anatomic relationships and clinical implications. Neurology. 2008;70(11):848–52.
Zhu DQ, Chen Q, Xiang YL, et al. Predicting intraventricular hemorrhage growth with a machine learning-based, radiomics-clinical model. Aging (Albany NY). 2021;13(9):12833–48.
Gaberel T, Magheru C, Emery E. Management of non-traumatic intraventricular hemorrhage. Neurosurg Rev. 2012;35(4):485–94 (discussion 494-5).
Kim BJ, Lee SH, Ryu WS, et al. Extents of white matter lesions and increased intraventricular extension of intracerebral hemorrhage. Crit Care Med. 2013;41(5):1325–31.
Vagal V, Venema SU, Behymer TP, et al. White matter lesion severity is associated with intraventricular hemorrhage in spontaneous intracerebral hemorrhage. J Stroke Cerebrovasc Dis. 2020;29(5):104661.
Witsch J, Bruce E, Meyers E, et al. Intraventricular hemorrhage expansion in patients with spontaneous intracerebral hemorrhage. Neurology. 2015;84(10):989–94.
Maas MB, Nemeth AJ, Rosenberg NF, Kosteva AR, Prabhakaran S, Naidech AM. Delayed intraventricular hemorrhage is common and worsens outcomes in intracerebral hemorrhage. Neurology. 2013;80(14):1295–9.
Yogendrakumar V, Ramsay T, Fergusson DA, et al. Redefining hematoma expansion with the inclusion of intraventricular hemorrhage growth. Stroke. 2020;51(4):1120–7.
van Swieten JC, Hijdra A, Koudstaal PJ, van Gijn J. Grading white matter lesions on CT and MRI: a simple scale. J Neurol Neurosurg Psychiatry. 1990;53(12):1080–3.
Wardlaw JM, Smith EE, Biessels GJ, et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 2013;12(8):822–38.
Rodrigues MA, Samarasekera N, Lerpiniere C, et al. Association between computed tomographic biomarkers of cerebral small vessel diseases and long-term outcome after spontaneous intracerebral hemorrhage. Ann Neurol. 2021;89(2):266–79.
Graeb DA, Robertson WD, Lapointe JS, Nugent RA, Harrison PB. Computed tomographic diagnosis of intraventricular hemorrhage. Etiol Progn Radiol. 1982;143(1):91–6.
Lee SH, Park KJ, Park DH, Kang SH, Park JY, Chung YG. Factors associated with clinical outcomes in patients with primary intraventricular hemorrhage. Med Sci Monit. 2017;23:1401–12.
Wan Y, Gao F, Ye F, et al. Effects of aging on hydrocephalus after intraventricular hemorrhage. Fluids Barriers CNS. 2020;17(1):8.
Mayer SA, Brun NC, Begtrup K, et al. Efficacy and safety of recombinant activated factor VII for acute intracerebral hemorrhage. N Engl J Med. 2008;358(20):2127–37.
Cusack TJ, Carhuapoma JR, Ziai WC. Update on the treatment of spontaneous intraparenchymal hemorrhage: medical and interventional management. Curr Treat Options Neurol. 2018;20(1):1.
Mayer SA, Davis SM, Skolnick BE, et al. Can a subset of intracerebral hemorrhage patients benefit from hemostatic therapy with recombinant activated factor VII? Stroke. 2009;40(3):833–40.
Li Q, Warren AD, Qureshi AI, et al. Ultra-early blood pressure reduction attenuates hematoma growth and improves outcome in intracerebral hemorrhage. Ann Neurol. 2020;88(2):388–95.
Alber J, Alladi S, Bae HJ, et al. White matter hyperintensities in vascular contributions to cognitive impairment and dementia (VCID): knowledge gaps and opportunities. Alzheimers Dement (N Y). 2019;5:107–17.
Thrippleton MJ, Backes WH, Sourbron S, et al. Quantifying blood-brain barrier leakage in small vessel disease: review and consensus recommendations. Alzheimers Dement. 2019;15(6):840–58.
Freeze WM, Jacobs HIL, de Jong JJ, et al. White matter hyperintensities mediate the association between blood-brain barrier leakage and information processing speed. Neurobiol Aging. 2020;85:113–22.
Zhang CE, Wong SM, Uiterwijk R, et al. Blood-brain barrier leakage in relation to white matter hyperintensity volume and cognition in small vessel disease and normal aging. Brain Imaging Behav. 2019;13(2):389–95.
Kerkhofs D, Wong SM, Zhang E, et al. Baseline blood-brain barrier leakage and longitudinal microstructural tissue damage in the periphery of white matter hyperintensities. Neurology. 2021;96(17):e2192–200.
Burchell SR, Tang J, Zhang JH. Hematoma expansion following intracerebral hemorrhage: mechanisms targeting the coagulation cascade and platelet activation. Curr Drug Targets. 2017;18(12):1329–44.
Yu S, Arima H, Heeley E, et al. White blood cell count and clinical outcomes after intracerebral hemorrhage: the INTERACT2 trial. J Neurol Sci. 2016;361:112–6.
Tu S, Zhao R, Fang H, Wang L, Shao A, Sheng J. Association between non-alcoholic fatty liver disease and intracerebral hemorrhage. Cell Transpl. 2019;28(8):1033–8.
Kim HC, Oh SM, Pan WH, et al. Association between alanine aminotransferase and intracerebral hemorrhage in East Asian populations. Neuroepidemiology. 2013;41(2):131–8.
Tan G, Hao Z, Lei C, et al. Subclinical change of liver function could also provide a clue on prognosis for patients with spontaneous intracerebral hemorrhage. Neurol Sci. 2016;37(10):1693–700.
Steiner T, Rosand J, Diringer M. Intracerebral hemorrhage associated with oral anticoagulant therapy: current practices and unresolved questions. Stroke. 2006;37(1):256–62.
Nurden AT. Platelets, inflammation and tissue regeneration. Thromb Haemost. 2011;105(Suppl 1):S13-33.
Acknowledgments
Thanks to all the coworkers who made a contribution to this research and to all the patients who made this research possible.
Author information
Authors and Affiliations
Contributions
MY wrote the manuscript. MY and ZL interpreted the data and prepared the tables and figures. DZ, YY, and HX revised the manuscript for intellectual content. MY and HX acquired the data. DZ, HX, and YY contributed to the conception and design of the study. ZP contributed to the database supplement and manuscript revision. All authors read and approved its publication.
Corresponding author
Ethics declarations
Source of support
This study was supported by the Scientific Research Incubation Project (FHY2019072); the Science and Technology Planning Projects of Wenzhou (grant Y2020169); the Wenzhou Major Program of Science and Technology Innovation (Grant ZY2020012); the Health Foundation for Creative Talents in Zhejiang Province, China (grant 2016); and the Project Foundation for the College Young and Middle-aged Academic Leader of Zhejiang Province, China (grant 2017).
Conflicts of Interest
All authors declare that they have no conflicts of interest.
Ethical approval/informed consent
The authors adhered to ethical guidelines, and this study was approved by the institutional review board. Written informed consent was waived because of the retrospective design.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yu, M., Zhu, D., Luo, Z. et al. Moderate-Severe White Matter Lesion Predicts Delayed Intraventricular Hemorrhage in Intracerebral Hemorrhage. Neurocrit Care 37, 714–723 (2022). https://doi.org/10.1007/s12028-022-01543-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-022-01543-x