Abstract
Background
With the increasing use of magnetic resonance imaging in the assessment of acute intracerebral hemorrhage, diffusion-weighted imaging hyperintense lesions have been recognized to occur at sites remote to the hematoma in up to 40% of patients. We investigated whether blood pressure reduction was associated with diffusion-weighted imaging hyperintense lesions in acute intracerebral hemorrhage and whether such lesions are associated with worse clinical outcomes by analyzing imaging data from a randomized trial.
Methods
We performed exploratory subgroup analyses in an open-label randomized trial that investigated acute blood pressure lowering in 1000 patients with intracerebral hemorrhage between May 2011 and September 2015. Eligible participants were assigned to an intensive systolic blood pressure target of 110–139 mm Hg versus 140–179 mm Hg with the use of intravenous nicardipine. Of these, 171 patients had requisite magnetic resonance imaging sequences for inclusion in these subgroup analyses. The primary outcome was the presence of diffusion-weighted imaging hyperintense lesions. Secondary outcomes included death or disability and serious adverse event at 90 days.
Results
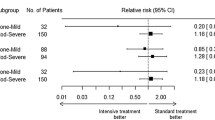
Diffusion-weighted imaging hyperintense lesions were present in 25% of patients (mean age 62 years). Hematoma volume > 30 cm3 was an adjusted predictor (adjusted relative risk 2.41, 95% confidence interval 1.00–5.80) of lesion presence. Lesions occurred in 25% of intensively treated patients and 24% of standard treatment patients (relative risk 1.01, 95% confidence interval 0.71–1.43, p = 0.97). Patients with diffusion-weighted imaging hyperintense lesions had similar frequencies of death or disability at 90 days, compared with patients without lesions.
Conclusions
Randomized assignment to intensive acute blood pressure lowering did not result in a greater frequency of diffusion-weighted imaging hyperintense lesion. Alternative mechanisms of diffusion-weighted imaging hyperintense lesion formation other than hemodynamic fluctuations need to be explored.
Clinical trial registration ClinicalTrials.gov (Ref. NCT01176565; https://clinicaltrials.gov/ct2/show/NCT01176565).


Similar content being viewed by others
References
Kimberly WT, Gilson A, Rost NS, Rosand J, Viswanathan A, Smith EE, et al. Silent ischemic infarcts are associated with hemorrhage burden in cerebral amyloid angiopathy. Neurology. 2009;72:1230–5.
Prabhakaran S, Gupta R, Ouyang B, John S, Temes RE, Mohammad Y, et al. Acute brain infarcts after spontaneous intracerebral hemorrhage: a diffusion-weighted imaging study. Stroke. 2010;41:89–94.
Gregoire SM, Charidimou A, Gadapa N, Dolan E, Antoun N, Peeters A, et al. Acute ischaemic brain lesions in intracerebral haemorrhage: multicentre cross-sectional magnetic resonance imaging study. Brain. 2011;134:2376–86.
Kang DW, Han MK, Kim HJ, Yun SC, Jeon SB, Bae HJ, et al. New ischemic lesions coexisting with acute intracerebral hemorrhage. Neurology. 2012;79:848–55.
Auriel E, Gurol ME, Ayres A, Dumas AP, Schwab KM, Vashkevich A, et al. Characteristic distributions of intracerebral hemorrhage-associated diffusion-weighted lesions. Neurology. 2012;79:2335–41.
Garg RK, Liebling SM, Maas MB, Nemeth AJ, Russell EJ, Naidech AM. Blood pressure reduction, decreased diffusion on mri, and outcomes after intracerebral hemorrhage. Stroke. 2012;43:67–71.
Menon RS, Burgess RE, Wing JJ, Gibbons MC, Shara NM, Fernandez S, et al. Predictors of highly prevalent brain ischemia in intracerebral hemorrhage. Ann Neurol. 2012;71:199–205.
Arsava EM, Kayim-Yildiz O, Oguz KK, Akpinar E, Topcuoglu MA. Elevated admission blood pressure and acute ischemic lesions in spontaneous intracerebral hemorrhage. J Stroke Cerebrovasc Dis. 2013;22:250–4.
Butcher KS, Jeerakathil T, Hill M, Demchuk AM, Dowlatshahi D, Coutts SB, et al. The intracerebral hemorrhage acutely decreasing arterial pressure trial. Stroke. 2013;44:620–6.
Gioia LC, Kate M, Choi V, Sivakumar L, Jeerakathil T, Kosior J, et al. Ischemia in intracerebral hemorrhage is associated with leukoaraiosis and hematoma volume, not blood pressure reduction. Stroke. 2015;46:1541–7.
Kidwell CS, Rosand J, Norato G, Dixon S, Worrall BB, James ML, et al. Ischemic lesions, blood pressure dysregulation, and poor outcomes in intracerebral hemorrhage. Neurology. 2017;88(8):782–8.
Tsai YH, Lee MH, Weng HH, Chang SW, Yang JT, Huang YC. Fate of diffusion restricted lesions in acute intracerebral hemorrhage. PLoS One. 2014;9:e105970.
van Veluw SJ, Shih AY, Smith EE, Chen C, Schneider JA, Wardlaw JM, et al. Detection, risk factors, and functional consequences of cerebral microinfarcts. Lancet Neurol. 2017;16:730–40.
van Veluw SJ, Lauer A, Charidimou A, Bounemia N, Xiong L, Boulouis G, et al. Evolution of dwi lesions in cerebral amyloid angiopathy: evidence for ischemia. Neurology. 2017;89:2136–42.
Wu B, Yao X, Lei C, Liu M, Selim MH. Enlarged perivascular spaces and small diffusion-weighted lesions in intracerebral hemorrhage. Neurology. 2015;85:2045–52.
Qureshi AI, Palesch YY. Antihypertensive treatment of acute cerebral hemorrhage (atach) ii: design, methods, and rationale. Neurocrit Care. 2011;15:559–76.
Qureshi AI, Palesch YY, Barsan WG, Hanley DF, Hsu CY, Martin RL, et al. Intensive blood-pressure lowering in patients with acute cerebral hemorrhage. N Engl J Med. 2016;375:1033–43.
Goldstein J, Brouwers H, Romero J, McNamara K, Schwab K, Greenberg S, et al. Score-it: the spot sign score in restricting ich growth horizontal line an atach-ii ancillary study. J Vasc Interv Neurol. 2012;5:20–5.
Greenberg SM, Vernooij MW, Cordonnier C, Viswanathan A, Al-Shahi Salman R, Warach S, et al. Cerebral microbleeds: a guide to detection and interpretation. Lancet Neurol. 2009;8:165–74.
Wardlaw JM, Smith EE, Biessels GJ, Cordonnier C, Fazekas F, Frayne R, et al. Neuroimaging standards for research into small vessel disease and its contribution to ageing and neurodegeneration. Lancet Neurol. 2013;12:822–38.
Wahlund LO, Barkhof F, Fazekas F, Bronge L, Augustin M, Sjögren M, et al. A new rating scale for age-related white matter changes applicable to mri and ct. Stroke. 2001;32:1318–22.
Oliveira-Filho J, Ay H, Shoamanesh A, Park KY, Avery R, Sorgun M, et al. Incidence and etiology of microinfarcts in patients with ischemic stroke. J Neuroimaging. 2018;28:406–11.
Murthy SB, Cho SM, Gupta A, Shoamanesh A, Navi BB, Avadhani R, et al. A pooled analysis of diffusion-weighted imaging lesions in patients with acute intracerebral hemorrhage. JAMA Neurol. 2020;77:1–9.
Ye XH, Cai XL, Nie DL, Chen YJ, Li JW, Xu XH, et al. Stress-induced hyperglycemia and remote diffusion-weighted imaging lesions in primary intracererbal hemorrhage. Neurocrit Care. 2020;32:427–36.
Zazulia AR, Diringer MN, Videen TO, Adams RE, Yundt K, Aiygari V, et al. Hypoperfusion without ischemia surrounding acute intracerebral hemorrhage. J Cereb Blood Flow Metab. 2001;21:804–10.
Shoamanesh A, Morotti A, Romero JM, Oliveira-Filho J, Schlunk F, Jessel MJ, et al. Cerebral microbleeds and the effect of intensive blood pressure reduction on hematoma expansion and functional outcomes: a secondary analysis of the atach-2 randomized clinical trial. JAMA Neurol. 2018;75:850–9.
Author information
Authors and Affiliations
Consortia
Contributions
All authorship requirements have been met, and the final manuscript was approved by all authors. Study conception and design: AS, CC, AIQ, JR, JNG. Acquisition of data: AS, CC, AM, JR, JOF, FS, MJ, AA, AV, MRA, RHM. Administrative support: MJ, AA, AV, KS. Statistical analysis and interpretation of data: AS, CC, RHM, JR, JNG. Drafting of manuscript: AS. Critical revisions: AS, CC, AM, JR, JOF, FS, MJ, KB, LG, AA, AV, MRA, KS, MRA, RHM, AIQ, SMG, JR, JNG. Study supervision/funding: JNG, JR, AIQ.
Corresponding author
Ethics declarations
Source of support
Supported by grants (National Institutes of Health - National Institute of Neurological Disorders and Stroke (NINDS) R01NS073344, to Dr. Rosand and U01-NS062091, to Dr. Qureshi). Chiesi USA and Astellas Pharma supplied intravenous nicardipine for use during the trial but had no other role in the design or conduct of the ATACH-2 trial or in this analysis/article. The National Institutes of Health, Chiesi USA, and Astellas Pharma had no involvement in the design, analyses, interpretation, or drafting of this report.
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval/informed consent
The ATACH-2 protocol and consent forms were approved by the institutional review board or equivalent ethics committee at each participating site, and all participants or their legally authorized representative provided written informed consent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shoamanesh, A., Cassarly, C., Morotti, A. et al. Intensive Blood Pressure Lowering and DWI Lesions in Intracerebral Hemorrhage: Exploratory Analysis of the ATACH-2 Randomized Trial. Neurocrit Care 36, 71–81 (2022). https://doi.org/10.1007/s12028-021-01254-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12028-021-01254-9