Abstract
Purpose
The current study aimed to report cases of McCune Albright syndrome (MAS) with growth hormone (GH) hyper secretion along with a systematic review of literature to elucidate challenges and intricacies in its diagnosis and management.
Methods
It was a single centre study carried out in individuals with MAS and autonomous GH secretion (AGHS). In addition, a systematic search of literature across three databases (PubMed, Scopus and EMBASE) was performed from inception until May 31, 2021 to identify cases of MAS with AGHS in the pediatric age group (<18 years).
Results
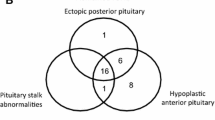
Three cases from authors centre and 42 cases identified from systematic literature review were analysed. Precocious puberty was the most common presenting endocrinopathy seen in 56.8% (25/44) cases, followed by hyperthyroidism (10/45), hypophosphatemia (4/45), and hypercortisolism (2/45). Cranio-facial fibrous dysplasia (CFFD) was seen in all while polyostotic fibrous dysplasia and Café au lait macule was seen in 40/45 (88.9%) and 35/45 (77.8%), respectively. Pituitary adenoma (58.3% microadenoma) was localized in 53.3% (24/45) cases on pituitary imaging. Biochemical and clinical remission of AGHS was achieved in 61.5% (24/45) cases with medical therapy.
Conclusion
Diagnosing AGHS in MAS is challenging because of concomitant presence of CFFD, non-GH endocrinopathies associated height spurt and elevated serum IGF-1. GH-GTT should be performed in presence of elevated growth velocity and serum IGF-1 (>1 X ULN) despite adequate control of non-GH endocrinopathies. Medical management can lead to disease control in substantial number of cases and often entails use of multiple agents.


Similar content being viewed by others
References
L.S. Weinstein, A. Shenker, P.V. Gejman, M.J. Merino, E. Friedman, A.M. Spiegel, “Activating mutations of the stimulatory G protein in the McCune-Albright syndrome,”. N. Engl. J. Med. 325(no. 24), 1688–1695 (1991). https://doi.org/10.1056/NEJM199112123252403
C.A. Landis, S.B. Masters, A. Spada, A.M. Pace, H.R. Bourne, L. Vallar, “GTPase inhibiting mutations activate the alpha chain of Gs and stimulate adenylyl cyclase in human pituitary tumours,”. Nature 340(no. 6236), 692–696 (1989). https://doi.org/10.1038/340692a0
F. Albright et al. “Syndrome characterized by osteitis Fibrosa Disseminata, areas of pigmentation and endocrine dysfunction, with precocious puberty in females,”. N. Engl. J. Med. 216(17), 727–746 (1937
A. Ramakrishnan, R. Kumar, K. Narayan, S. Kumar, “Jaffe Lichtenstein Type Polyostotic Fibrous Dysplasia with Unilateral Absent Testis,” JCDR, (2018). https://doi.org/10.7860/JCDR/2018/34680.11564
A.M. Boyce, P. Florenzano, L.F. de Castro, et al. Fibrous Dysplasia/McCune-Albright Syndrome. Seattle (WA): University of Washington, Seattle; 1993–2021.: Adam MP, Ardinger HH, Pagon RA, et al., editors. GeneReviews® [Internet]. [Online]. Available: https://www.ncbi.nlm.nih.gov/books/NBK274564/
M.T. Collins, F.R. Singer, E. Eugster, “McCune-Albright syndrome and the extraskeletal manifestations of fibrous dysplasia,”. Orphanet J. Rare Dis. 7(Suppl 1), S4 (2012). https://doi.org/10.1186/1750-1172-7-S1-S4
S. Salenave, A.M. Boyce, M.T. Collins, P. Chanson, “Acromegaly and McCune-Albright syndrome,”. J. Clin. Endocrinol. Metab. 99(no. 6), 1955–1969 (2014). https://doi.org/10.1210/jc.2013-3826
J.S. Lee et al. Normal vision despite narrowing of the optic canal in fibrous dysplasia. N. Engl. J. Med 347(no. 21), 1670–1676 (2002). https://doi.org/10.1056/NEJMoa020742
C.M. Cutler et al. Long-term outcome of optic nerve encasement and optic nerve decompression in patients with fibrous dysplasia: risk factors for blindness and safety of observation. Neurosurgery 59(no. 5), 1011–1017 (2006). https://doi.org/10.1227/01.NEU.0000254440.02736.E3
D. Tessaris et al. Growth hormone-Insulin-like growth factor 1 axis hyperactivity on bone fibrous dysplasia in McCune-Albright Syndrome. Clin. Endocrinol. 89(no. 1), 56–64 (2018). https://doi.org/10.1111/cen.13722
P. Ruggieri, F.H. Sim, J.R. Bond, K.K. Unni, “Malignancies in fibrous dysplasia,”. Cancer 73(no. 5), 1411–1424 (1994)
M. Bidlingmaier et al. Reference intervals for insulin-like growth factor-1 (IGF-I) from birth to senescence: results from a multicenter study using a new automated chemiluminescence IGF-I immunoassay conforming to recent international recommendations. J. Clin. Endocrinol. Metab. 99(no. 5), 1712–1721 (2014). https://doi.org/10.1210/jc.2013-3059
“Corrigendum to: ‘Reference Intervals for Insulin-Like Growth Factor-1 (IGF-I) From Birth to Senescence: Results From a Multicenter Study Using a New Automated Chemiluminescence IGF-I Immunoassay Conforming to Recent International Recommendations,’”. J. Clin. Endocrinol. Metab. 105(no. 12), e4983–e4983 (2020). https://doi.org/10.1210/clinem/dgaa641
S.O. Akintoye et al. “Characterization of gsp-mediated growth hormone excess in the context of McCune-Albright syndrome,”. J. Clin. Endocrinol. Metab. 87(no. 11), 5104–5112 (2002). https://doi.org/10.1210/jc.2001-012022
L. Fuyi et al. A case of McCune-Albright syndrome associated with pituitary GH adenoma: therapeutic process and autopsy. J. Pediatr. Endocrinol. Metab. 24(no.), 5–6 (2011). https://doi.org/10.1515/jpem.2011.178
P. Chanson, A. Dib, A. Visot, P.J. Derome, “McCune-Albright syndrome and acromegaly: clinical studies and responses to treatment in five cases,”. Eur. J. Endocrinol. 131(no. 3), 229–234 (1994). https://doi.org/10.1530/eje.0.1310229
L.D. Premawardhana, J.P. Vora, R. Mills, M.F. Scanlon, “Acromegaly and its treatment in the McCune-Albright syndrome,”. Clin. Endocrinol. (Oxf.) 36(no. 6), 605–608 (1992). https://doi.org/10.1111/j.1365-2265.1992.tb02272.x
C. Ihara, A. Shimatsu, H. Murabe, K. Kataoka, C. Kondo, K. Nakao, “Growth hormone-secreting pituitary adenoma associated with multiple bone cysts, skin pigmentation and aortitis syndrome,”. J. Endocrinol. Invest. 19(no. 11), 753–757 (1996). https://doi.org/10.1007/BF03347879
Y. Yao et al. Clinical characteristics and management of growth hormone excess in patients with McCune–Albright syndrome. Eur. J. Endocrinol. 176(no. 3), 295–303 (2017). https://doi.org/10.1530/EJE-16-0715
X. Wang, Q. Yu, “Management of precocious puberty in girls with McCune–Albright syndrome using letrozole,”. Endocr. Connect. 7(no. 12), 1424–1431 (2018). https://doi.org/10.1530/EC-18-0344
H. Schmidt, W. Kiess, “Secondary central precocious puberty in a girl with McCune-Albright syndrome responds to treatment with GnRH analogue. J. Pediatr. Endocrinol. Metab. 11(no. 1), 77–81 (1998). https://doi.org/10.1515/jpem.1998.11.1.77
P. Feuillan, K. Calis, S. Hill, T. Shawker, P.G. Robey, M.T. Collins, “Letrozole treatment of precocious puberty in girls with the McCune-Albright syndrome: a pilot study,”. J. Clin. Endocrinol. Metab. 92(no. 6), 2100–2106 (2007). https://doi.org/10.1210/jc.2006-2350
M.K. Javaid et al. Best practice management guidelines for fibrous dysplasia/McCune-Albright syndrome: a consensus statement from the FD/MAS international consortium. Orphanet J. Rare Dis. 14(no. 1), 139 (2019). https://doi.org/10.1186/s13023-019-1102-9
H. Plotkin, F. Rauch, L. Zeitlin, C. Munns, R. Travers, F.H. Glorieux, “Effect of pamidronate treatment in children with polyostotic fibrous dysplasia of bone,”. J. Clin. Endocrinol. Metab. 88(no. 10), 4569–4575 (2003). https://doi.org/10.1210/jc.2003-030050
E.J.P. Prada, K.H. Hassan, M.L. Brandi, A. Falchetti, “Polyostotic form of fibrous dysplasia in a 13 years old Colombian girl showing clinical and biochemical response to neridronate intravenous therapy,”. Clin. Cases Min. Bone Metab. 6(no. 3), 264–265 (2009)
K.N. Shah, “The diagnostic and clinical significance of Café-au-lait Macules,”. Pediatr. Clin. North Am. 57(no. 5), 1131–1153 (2010). https://doi.org/10.1016/j.pcl.2010.07.002
Author information
Authors and Affiliations
Contributions
S.S.J. collected data, reviewed literature and wrote the manuscript, R.P. review and editied manuscript, R.W. was involved in study design, review/edited manuscript and patients management, R.G., S.C., K.A., A.R., S.K.B., A.B.1 were involved in patients management and editied manuscript. A.B.2 was involved in nuclear bone scan and reporting.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jayant, S.S., Walia, R., Gupta, R. et al. Autonomous growth hormone secretion due to McCune Albright syndrome in paediatric age group: an ominous triad. Endocrine 81, 149–159 (2023). https://doi.org/10.1007/s12020-023-03333-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-023-03333-7