Abstract
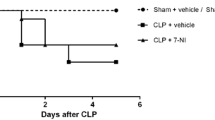
The aim of this study was to analyze the effect of IL-1ra (an Interleukin-1 receptor antagonist) on sepsis-induced alterations in vasopressin (AVP) and nitric oxide (NO) levels. In addition, IL-1ra effect on the hypothalamic nitric oxide synthase (NOS) activities and survival rate was also analyzed. After Wistar rats were intracerebroventricular injected with IL-1ra (9 pmol) or vehicle (PBS 0.01 M), sepsis was induced by cecal-ligation and puncture (CLP). Blood, CSF, and hypothalamic samples were collected from different groups of rats (n = 8/group) after 4, 6, and 24 h. AVP and NO levels were greatly increased in CLP. Both total NOS and inducible NOS (iNOS) activities were also greatly increased in CLP rats. These changes in AVP, NO, and NOS were not observed in sham-operated control rats. IL-1ra administration did not alter plasma AVP levels after 4 and 6 h as compared to vehicle in CLP animals but after 24 h were significantly (P < 0.01) higher in IL-1ra-treated animals. IL-1ra administration significantly (P < 0.01) decreased NO concentration in CSF but not in plasma. Both total NOS and iNOS activities were also significantly decreased by IL-1ra at 24 h in CLP animals. Moreover, the 24 h survival rate of IL-1ra-treated rats increased by 38 % in comparison to vehicle administered animals. The central administration of IL-1ra increased AVP secretion in the late phase of sepsis which was beneficial for survival. We believe that one of the mechanisms for this effect of IL-1ra is through reduction of NO concentration in CSF and hence lower hypothalamic iNOS activities in the septic rats.





Similar content being viewed by others

References
R.C. Bone, The pathogenesis of sepsis. Ann. Intern. Med. 115, 457–469 (1991)
R.C. Bone, W.J. Sibbald, C.L. Sprung, The ACCP-SCCM consensus conference on sepsis and organ failure. Chest 101, 1481–1483 (1992)
J.L. Vincent, H.A. Korkut, Defining sepsis. Clin. Chest Med. 29, 585–590 (2008)
D. Annane, E. Bellissant, J.M. Cavaillon, Septic shock. Lancet 365, 63–78 (2005)
K.J. Kovacs, Neurohypophyseal hormones in the integration of physiological responses to immune challenges. Prog. Brain Res. 139, 127–146 (2002)
S.M. McCann, M. Kimura, S. Karanth, W.H. Yu, C.A. Mastronardi, V. Rettori, The mechanism of action of cytokines to control the release of hypothalamic and pituitary hormones in infection. Ann. N. Y. Acad. Sci. 917, 4–18 (2000)
J.E. Parrillo, Pathogenetic mechanisms of septic shock. N. Engl. J. Med. 328, 1471–1477 (1993)
M.J.A. Rocha, G.R. Oliveira, P.B. Farias-Corrêa, Neurohypophyseal hormone secretion during septic shock, in New trends in brain research, ed. by F.J. Chen (Nova Science Publishers, New York, 2006), pp. 75–94
D.W. Landry, H.R. Levin, E.M. Gallant, R.C. Ashton Jr, S. Seo, D. D’Alessandro, M.C. Oz, J.A. Oliver, Vasopressin deficiency contributes to the vasodilation of septic shock. Circulation 95, 1122–1125 (1997)
G.R. Oliveira-Pelegrin, M.I. Ravanelli, L.G. Branco, M.J. Rocha, Thermoregulation and vasopressin secretion during polymicrobial sepsis. NeuroImmunomodulation 16, 45–53 (2009)
T. Sharshar, A. Blanchard, M. Paillard, J.C. Raphael, P. Gajdos, D. Annane, Circulating vasopressin levels in septic shock. Crit. Care Med. 31, 1752–1758 (2003)
L.A. Athayde, G.R. Oliveira-Pelegrin, A. Nomizo, L.H. Faccioli, M.J. Rocha, Blocking central leukotrienes synthesis affects vasopressin release during sepsis. Neuroscience 160, 829–836 (2009)
P.B. Correa, J.A. Pancoto, G.R. de Oliveira-Pelegrin, E.C. Carnio, M.J. Rocha, Participation of iNOS-derived NO in hypothalamic activation and vasopressin release during polymicrobial sepsis. J. Neuroimmunol. 183, 17–25 (2007)
J.A. Pancoto, P.B. Correa, G.R. Oliveira-Pelegrin, M.J. Rocha, Autonomic dysfunction in experimental sepsis induced by cecal ligation and puncture. Auton. Neurosci. 138, 57–63 (2008)
F. Wahab, B. Atika, G.R. Oliveira-Pelegrin, M.J. Rocha, Recent advances in the understanding of sepsis-induced alterations in the neuroendocrine system. Endocr. Metab. Immune Disord. Drug Targets 13, 335–347 (2013)
M.L. Wong, P.B. Bongiorno, V. Rettori, S.M. McCann, J. Licinio, Interleukin (IL) 1beta, IL-1 receptor antagonist, IL-10, and IL-13 gene expression in the central nervous system and anterior pituitary during systemic inflammation: pathophysiological implications. Proc. Natl. Acad. Sci. U.S.A. 94, 227–232 (1997)
C.A. Dinarello, Interleukin-1 in the pathogenesis and treatment of inflammatory diseases. Blood 117, 3720–3732 (2011)
G.R. Oliveira-Pelegrin, P.J. Basso, M.J. Rocha, Cellular bioenergetics changes in magnocellular neurons may affect copeptin expression in the late phase of sepsis. J. Neuroimmunol. 267, 28–34 (2014)
T. Sharshar, R. Carlier, A. Blanchard, A. Feydy, F. Gray, M. Paillard, J.C. Raphael, P. Gajdos, D. Annane, Depletion of neurohypophyseal content of vasopressin in septic shock. Crit. Care Med. 30, 497–500 (2002)
A.R. Consiglio, A.B. Lucion, Technique for collecting cerebrospinal fluid in the cisterna magna of non-anesthetized rats. Brain Res. Brain Res. Protoc. 5, 109–114 (2000)
M.J. Rocha, Y. Chen, G.R. Oliveira, M. Morris, Physiological regulation of brain angiotensin receptor mRNA in AT1a deficient mice. Exp. Neurol. 195, 229–235 (2005)
D.S. Bredt, S.H. Snyder, Isolation of nitric oxide synthetase, a calmodulin-requiring enzyme. Proc. Natl. Acad. Sci. U.S.A. 87, 682–685 (1990)
G.R. Oliveira-Pelegrin, S.V. de Azevedo, S.T. Yao, D. Murphy, M.J. Rocha, Central NOS inhibition differentially affects vasopressin gene expression in hypothalamic nuclei in septic rats. J. Neuroimmunol. 227, 80–86 (2010)
M.L. Wong, V. Rettori, A. al-Shekhlee, P.B. Bongiorno, G. Canteros, S.M. McCann, P.W. Gold, J. Licinio, Inducible nitric oxide synthase gene expression in the brain during systemic inflammation. Nat. Med. 2, 581–584 (1996)
R. Sonneville, C. Guidoux, L. Barrett, O. Viltart, V. Mattot, A. Polito, S. Siami, G.L. de la Grandmaison, A. Blanchard, M. Singer, D. Annane, F. Gray, J.P. Brouland, T. Sharshar, Vasopressin synthesis by the magnocellular neurons is different in the supraoptic nucleus and in the paraventricular nucleus in human and experimental septic shock. Brain Pathol. 20, 613–622 (2010)
G.R. Oliveira-Pelegrin, F.A. Aguila, P.J. Basso, M.J. Rocha, Role of central NO-cGMP pathway in vasopressin and oxytocin gene expression during sepsis. Peptides 31, 1847–1852 (2010)
M.M. Gabellec, R. Griffais, G. Fillion, F. Haour, Expression of interleukin 1, interleukin 1 and interleukin 1 receptor antagonist mRNA in mouse brain: regulation by bacterial lipopolysaccharide (LPS) treatment. Mol. Brain Res. 31, 122–130 (1995)
B. Beltrán, A. Mathur, M.R. Duchen, J.D. Erusalimsky, S. Moncada, The effect of nitric oxide on cell respiration: a key to understanding its role in cell survival or death. Proc. Natl. Acad. Sci. U.S.A. 97, 14602–14607 (2000)
M.L. Wong, V. Rettori, A. al-Shekhlee, P.B. Bongiorno, G. Canteros, S.M. McCann, P.W. Gold, J. Licinio, Inducible nitric oxide synthase gene expression in the brain during systemic inflammation. Nat. Med. 2, 581–584 (1996)
S. Rivest, S. Lacroix, L. Vallières, S. Nadeau, J. Zhang, N. Laflamme, How the blood talks to the brain parenchyma and the paraventricular nucleus of the hypothalamus during systemic inflammatory and infectious stimuli. Proc. Soc. Exp. Biol. Med. 223, 22–38 (2000)
L.C. Ehrlich, K.P. Phillip, H. Shuxian, Interleukin (IL)-1 [beta]-mediated apoptosis of human astrocytes. Neuroreport 10(9), 1849–1852 (1999)
C. Binder, M. Schulz, W. Hiddemann, M. Oellerich, Caspase-activation and induction of inducible nitric oxide-synthase during TNF alpha-triggered apoptosis. Anticancer Res. 19, 1715–1720 (1998)
J. Bustamante, G. Bersier, M. Romero, R.A. Badin, A. Boveris, Nitric oxide production and mitochondrial dysfunction during rat thymocyte apoptosis. Arch. Biochem. Biophys. 376, 239–247 (2000)
G.C. Brown, C.E. Cooper, Nanomolar concentrations of nitric oxide reversibly inhibit synaptosomal respiration by competing with oxygen at cytochrome oxidase. FEBS Lett. 356, 295–298 (1994)
J.D. Erusalimsky, S. Moncada, Nitric oxide and mitochondrial signaling: from physiology to pathophysiology. Arterioscler. Thromb. Vasc. Biol. 27, 2524–2531 (2007)
F.X. Guix, I. Uribesalgo, M. Coma, F.J. Munoz, The physiology and pathophysiology of nitric oxide in the brain. Prog. Neurobiol. 76, 126–152 (2005)
P. Ghafourifar, E. Cadenas, Mitochondrial nitric oxide synthase. Trends Pharmacol. Sci. 26, 190–195 (2005)
F.R. Sharp, M. Bernaudin, HIF1 and oxygen sensing in the brain. Nat. Rev. Neurosci. 5, 437–448 (2004)
R.K. Bruick, Expression of the gene encoding the proapoptotic Nip3 protein is induced by hypoxia. Proc. Natl. Acad. Sci. U.S.A. 97, 9082–9087 (2000)
G.R. Oliveira-Pelegrin, R.S. Saia, E.C. Cárnio, M.J. Rocha, Oxytocin affects nitric oxide and cytokine production by sepsis-sensitized macrophages. NeuroImmunoModulation 20, 65–71 (2013)
Acknowledgments
The authors thank Nadir Martins Fernandes and Milene Mantovani for the technical assistant. Fernando Queiróz Cunha and José Antunes Rodrigues provided the infrastructure for the NOS activity analysis. Financial support from Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) is gratefully acknowledged.
Disclosures
The authors have nothing to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wahab, F., Tazinafo, L.F., Cárnio, E.C. et al. Interleukin-1 receptor antagonist decreases cerebrospinal fluid nitric oxide levels and increases vasopressin secretion in the late phase of sepsis in rats. Endocrine 49, 215–221 (2015). https://doi.org/10.1007/s12020-014-0452-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12020-014-0452-2