Abstract
Maternal genetic variants of enzymes in folate–homocysteine metabolic network are significantly correlative with the risk of spina bifida. To survey the genetic causality, the genotypes of three women having spina bifida fetuses from two unrelated Chinese families were screened in candidate alleles. Polymerase chain reaction, capillary electrophoresis and Sanger sequencing were employed to recognize the allelic variation. A trinucleotide deletion (c.4_6delAGG) was identified in the first exon of MTRR. All the three women showed the novel clinical variation including one heterozygous and two homozygous. The siblings who had healthy babies from the same families did not harbor the variation. In the unaffected control individuals, the variant was also not observed. Eukaryotic expression and bioinformatics techniques were utilized to explore the molecular pathogenesis of the potential genetic risk of developing spina bifida. Exceptionally, the functional examination revealed that the Arg2del variant kept subcellular localization unaltered with catalytic activity intact, but failed to efficiently activate MTR compared with the wild type. Genetic disorder of folate and homocysteine metabolism during pregnancy is believed to be associated with folate-sensitive neural tube defects. The report highlights that the inframe deletion in MTRR exon 1 could be a high risk factor susceptibility to spina bifida.




Similar content being viewed by others
References
Blom, H. J., Shaw, G. M., den Heijer, M., & Finnell, R. H. (2006). Neural tube defects and folate: Case far from closed. Nature Reviews Neuroscience, 7(9), 724–731.
Christensen, B., Arbour, L., Tran, P., Leclerc, D., Sabbaghian, N., Platt, R., et al. (1999). Genetic polymorphisms in methylenetetrahydrofolate reductase and methionine synthase, folate levels in red blood cells, and risk of neural tube defects. American Journal of Medical Genetics, 84(2), 151–157.
Detrait, E. R., George, T. M., Etchevers, H. C., Gilbert, J. R., Vekemans, M., & Speer, M. C. (2005). Human neural tube defects: Developmental biology, epidemiology, and genetics. Neurotoxicology and Teratology, 27(3), 515–524. (Epub 2005 Mar 5).
Doolin, M. T., Barbaux, S., McDonnell, M., Hoess, K., Whitehead, A. S., & Mitchell, L. E. (2002). Maternal genetic effects, exerted by genes involved in homocysteine remethylation, influence the risk of spina bifida. American Journal of Human Genetics, 71(5), 1222–1226. (Epub 2002 Oct 9).
Froese, D. S., Wu, X., Zhang, J., Dumas, R., Schoel, W. M., Amrein, M., et al. (2008). Restricted role for methionine synthase reductase defined by subcellular localization. Molecular Genetics and Metabolism, 94(1), 68–77. doi:10.1016/j.ymgme.2007.11.019. (Epub 2008 Jan 24).
Greene, N. D., & Copp, A. J. (2014). Neural tube defects. Annual Review of Neuroscience, 37, 221–242. doi:10.1146/annurev-neuro-062012-170354.
Imbard, A., Benoist, J. F., & Blom, H. J. (2013a). Neural tube defects, folic acid and methylation. International Journal of Environmental Research and Public Health, 10(9), 4352–4389. doi:10.3390/ijerph10094352.
Imbard, A., Benoist, J. F., & Blom, H. J. (2013b). Neural tube defects, folic acid and methylation. International Journal of Environmental Research and Public Health, 10(9), 4352–4389. doi:10.3390/ijerph10094352.
Jiang, J., Zhang, Y., Wei, L., Sun, Z., & Liu, Z. (2014). Association between MTHFD1 G1958A polymorphism and neural tube defects susceptibility: A meta-analysis. PLoS ONE, 9(6), e101169. doi:10.1371/journal.pone.0101169. (eCollection 2014).
Johnson, W. G., Stenroos, E. S., Spychala, J. R., Chatkupt, S., Ming, S. X., & Buyske, S. (2004). New 19 bp deletion polymorphism in intron-1 of dihydrofolate reductase (DHFR): a risk factor for spina bifida acting in mothers during pregnancy? American Journal of Medical Genetics Part A, 124A(4), 339–345.
Relton, C. L., Wilding, C. S., Pearce, M. S., Laffling, A. J., Jonas, P. A., Lynch, S. A., et al. (2004). Gene-gene interaction in folate-related genes and risk of neural tube defects in a UK population. Journal of Medical Genetics, 41(4), 256–260.
Ross, M. E. (2010). Gene-environment interactions, folate metabolism and the embryonic nervous system. Wiley Interdisciplinary Reviews: Systems Biology and Medicine, 2(4), 471–480. doi:10.1002/wsbm.72.
van der Put, N. M., Steegers-Theunissen, R. P., Frosst, P., Trijbels, F. J., Eskes, T. K., van den Heuvel, L. P., et al. (1995). Mutated methylenetetrahydrofolate reductase as a risk factor for spina bifida. Lancet, 346(8982), 1070–1071.
Wang, Y., Liu, Y., Ji, W., Qin, H., Wu, H., Xu, D., et al. (2015). Analysis of MTR and MTRR polymorphisms for neural tube defects risk association. Medicine (Baltimore), 94(35), e1367. doi:10.1097/MD.0000000000001367.
Wilson, A., Platt, R., Wu, Q., Leclerc, D., Christensen, B., Yang, H., et al. (1999). A common variant in methionine synthase reductase combined with low cobalamin (vitamin B12) increases risk for spina bifida. Molecular Genetics and Metabolism, 67(4), 317–323.
Yamada, K., Gravel, R. A., Toraya, T., & Matthews, R. G. (2006). Human methionine synthase reductase is a molecular chaperone for human methionine synthase. Proceedings of the National Academy of Sciences, 103(25), 9476–9481. (Epub 2006 Jun 12).
Acknowledgements
We gratefully acknowledged all the patients, family members and control participants in providing specimens and clinical data for this study. The work was funded by the grants from Chongqing Municipal Education Commission (KJ080301), Chongqing Science & Technology Commission (CSTC, 2010BB5366) and National Natural Science Foundation of China (NO. 20803098).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Additional information
Xiao-lu Dai and Gui-cen Liu are co-first authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Fig. s1
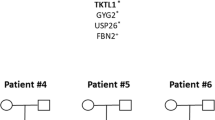
Pedigrees of the two families. In the family trees, the black circles represent the females who experienced spina bifida-affected pregnancy. The white circles signify the unaffected ones. Lack of allele information, the male or female mutation carrier could not be distinguished in the both families. The pedigree of family 1 is displayed on the left, family 2 on the right. The black arrows refer to the probands in the pedigrees. (TIFF 17592 kb)
Rights and permissions
About this article
Cite this article
Zhang, J., Dai, Xl., Liu, Gc. et al. An Inframe Trinucleotide Deletion in MTRR Exon 1 is Associated with the Risk of Spina Bifida. Neuromol Med 19, 387–394 (2017). https://doi.org/10.1007/s12017-017-8452-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-017-8452-z